 Found 37 hits of Enzyme Inhibition Constant Data
Found 37 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50192770
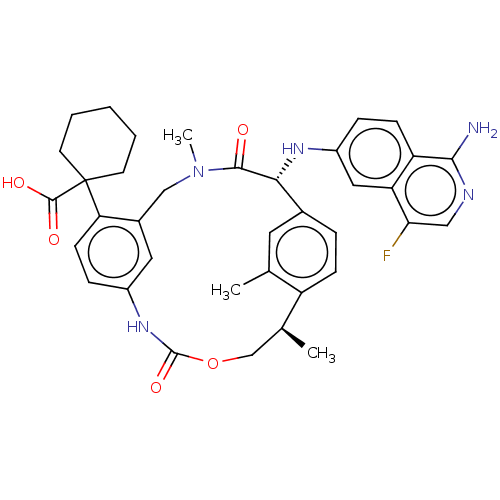
(CHEMBL3956096)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCCCC1)C(O)=O |r| Show InChI InChI=1S/C37H40FN5O5/c1-21-15-23-7-10-27(21)22(2)20-48-36(47)42-25-9-12-30(37(35(45)46)13-5-4-6-14-37)24(16-25)19-43(3)34(44)32(23)41-26-8-11-28-29(17-26)31(38)18-40-33(28)39/h7-12,15-18,22,32,41H,4-6,13-14,19-20H2,1-3H3,(H2,39,40)(H,42,47)(H,45,46)/t22-,32+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
| PDB
Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TF-factor 7a using factor 10 as substrate at 37 degC |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50192768
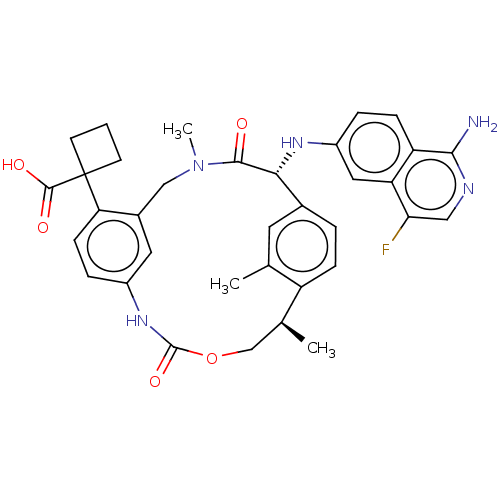
(CHEMBL3900166)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCC1)C(O)=O |r| Show InChI InChI=1S/C35H36FN5O5/c1-19-13-21-5-8-25(19)20(2)18-46-34(45)40-23-7-10-28(35(33(43)44)11-4-12-35)22(14-23)17-41(3)32(42)30(21)39-24-6-9-26-27(15-24)29(36)16-38-31(26)37/h5-10,13-16,20,30,39H,4,11-12,17-18H2,1-3H3,(H2,37,38)(H,40,45)(H,43,44)/t20-,30+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TF-factor 7a using factor 10 as substrate at 37 degC |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM189445
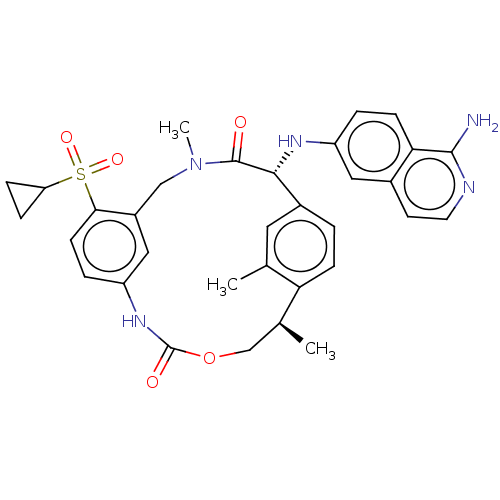
(US9174974, Example 31)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)nccc4c3)c3ccc1c(C)c3)c2)S(=O)(=O)C1CC1 |r| Show InChI InChI=1S/C33H35N5O5S/c1-19-14-22-4-9-27(19)20(2)18-43-33(40)37-25-6-11-29(44(41,42)26-7-8-26)23(16-25)17-38(3)32(39)30(22)36-24-5-10-28-21(15-24)12-13-35-31(28)34/h4-6,9-16,20,26,30,36H,7-8,17-18H2,1-3H3,(H2,34,35)(H,37,40)/t20-,30+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of human TF-factor 7a (366 to 11 residues) using factor 10 as substrate after 60 mins |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50192767
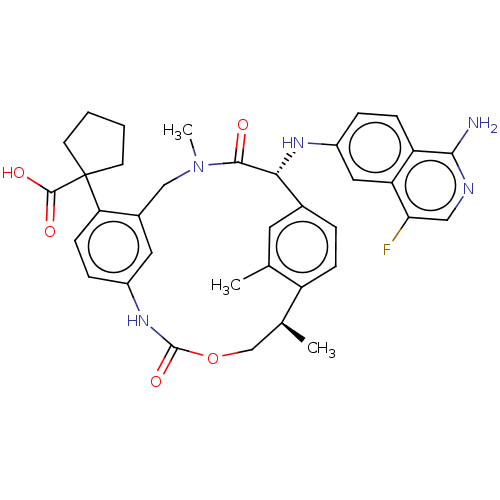
(CHEMBL3984725)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCCC1)C(O)=O |r| Show InChI InChI=1S/C36H38FN5O5/c1-20-14-22-6-9-26(20)21(2)19-47-35(46)41-24-8-11-29(36(34(44)45)12-4-5-13-36)23(15-24)18-42(3)33(43)31(22)40-25-7-10-27-28(16-25)30(37)17-39-32(27)38/h6-11,14-17,21,31,40H,4-5,12-13,18-19H2,1-3H3,(H2,38,39)(H,41,46)(H,44,45)/t21-,31+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 0.220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TF-factor 7a using factor 10 as substrate at 37 degC |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50192771

(CHEMBL3898371)Show SMILES [H][C@]1(C)COC(=O)Nc2ccc(c(CN(C)C(=O)[C@]([H])(Nc3ccc4c(c3)c(F)c[nH]c4=N)c3ccc1c(C)c3)c2)C1(CC1)C(O)=O |r| Show InChI InChI=1S/C34H34FN5O5/c1-18-12-20-4-7-24(18)19(2)17-45-33(44)39-22-6-9-27(34(10-11-34)32(42)43)21(13-22)16-40(3)31(41)29(20)38-23-5-8-25-26(14-23)28(35)15-37-30(25)36/h4-9,12-15,19,29,38H,10-11,16-17H2,1-3H3,(H2,36,37)(H,39,44)(H,42,43)/t19-,29+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 0.550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TF-factor 7a using factor 10 as substrate at 37 degC |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50192766
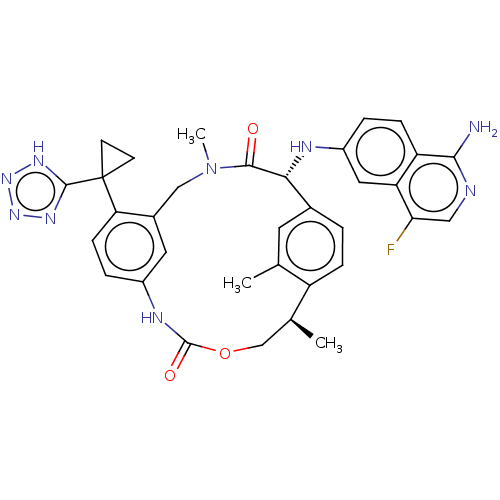
(CHEMBL3967204)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CC1)c1nnn[nH]1 |r| Show InChI InChI=1S/C34H34FN9O3/c1-18-12-20-4-7-24(18)19(2)17-47-33(46)39-22-6-9-27(34(10-11-34)32-40-42-43-41-32)21(13-22)16-44(3)31(45)29(20)38-23-5-8-25-26(14-23)28(35)15-37-30(25)36/h4-9,12-15,19,29,38H,10-11,16-17H2,1-3H3,(H2,36,37)(H,39,46)(H,40,41,42,43)/t19-,29+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 0.720 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TF-factor 7a using factor 10 as substrate at 37 degC |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50192769
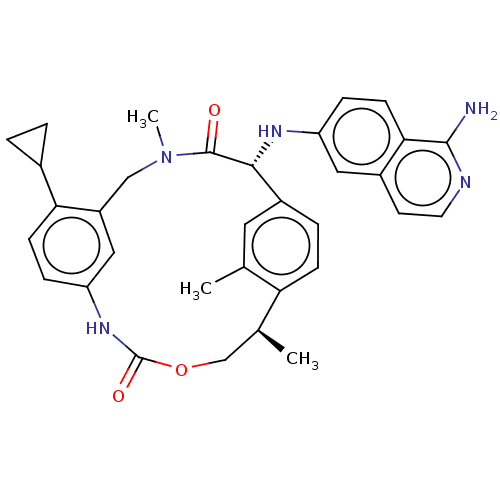
(CHEMBL3976650)Show SMILES C[C@H]1COC(=O)Nc2ccc(C3CC3)c(CN(C)C(=O)[C@H](Nc3ccc4c(N)nccc4c3)c3ccc1c(C)c3)c2 |r| Show InChI InChI=1S/C33H35N5O3/c1-19-14-23-6-9-27(19)20(2)18-41-33(40)37-26-7-10-28(21-4-5-21)24(16-26)17-38(3)32(39)30(23)36-25-8-11-29-22(15-25)12-13-35-31(29)34/h6-16,20-21,30,36H,4-5,17-18H2,1-3H3,(H2,34,35)(H,37,40)/t20-,30+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of human TF-factor 7a (366 to 11 residues) using factor 10 as substrate after 60 mins |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50192765
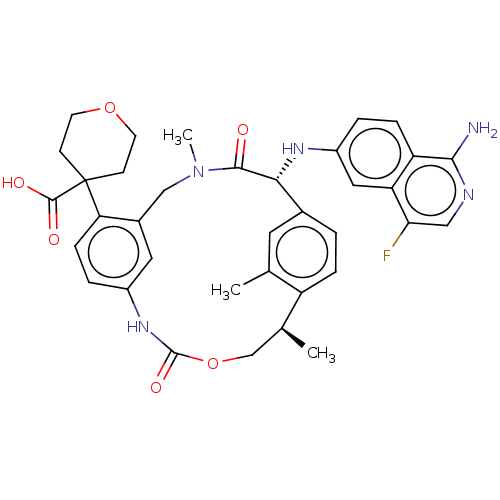
(CHEMBL3895930)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCOCC1)C(O)=O |r| Show InChI InChI=1S/C36H38FN5O6/c1-20-14-22-4-7-26(20)21(2)19-48-35(46)41-24-6-9-29(36(34(44)45)10-12-47-13-11-36)23(15-24)18-42(3)33(43)31(22)40-25-5-8-27-28(16-25)30(37)17-39-32(27)38/h4-9,14-17,21,31,40H,10-13,18-19H2,1-3H3,(H2,38,39)(H,41,46)(H,44,45)/t21-,31+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TF-factor 7a using factor 10 as substrate at 37 degC |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Kallikrein-1
(Homo sapiens (Human)) | BDBM50192764
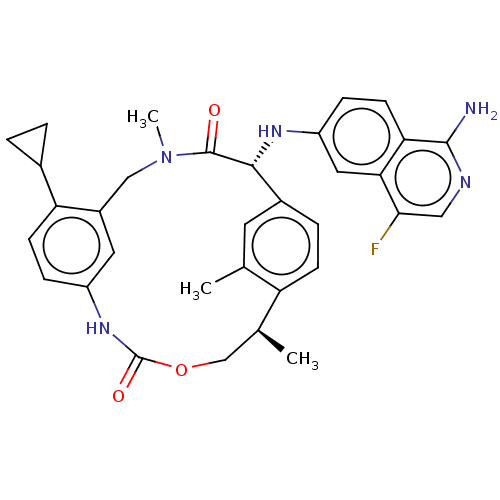
(CHEMBL3934882)Show SMILES C[C@H]1COC(=O)Nc2ccc(C3CC3)c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2 |r| Show InChI InChI=1S/C33H34FN5O3/c1-18-12-21-6-9-25(18)19(2)17-42-33(41)38-23-7-10-26(20-4-5-20)22(13-23)16-39(3)32(40)30(21)37-24-8-11-27-28(14-24)29(34)15-36-31(27)35/h6-15,19-20,30,37H,4-5,16-17H2,1-3H3,(H2,35,36)(H,38,41)/t19-,30+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of human kallikrein1 at 37 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50192764

(CHEMBL3934882)Show SMILES C[C@H]1COC(=O)Nc2ccc(C3CC3)c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2 |r| Show InChI InChI=1S/C33H34FN5O3/c1-18-12-21-6-9-25(18)19(2)17-42-33(41)38-23-7-10-26(20-4-5-20)22(13-23)16-39(3)32(40)30(21)37-24-8-11-27-28(14-24)29(34)15-36-31(27)35/h6-15,19-20,30,37H,4-5,16-17H2,1-3H3,(H2,35,36)(H,38,41)/t19-,30+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TF-factor 7a using factor 10 as substrate at 37 degC |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Vitamin K-dependent protein C
(Homo sapiens (Human)) | BDBM50192771

(CHEMBL3898371)Show SMILES [H][C@]1(C)COC(=O)Nc2ccc(c(CN(C)C(=O)[C@]([H])(Nc3ccc4c(c3)c(F)c[nH]c4=N)c3ccc1c(C)c3)c2)C1(CC1)C(O)=O |r| Show InChI InChI=1S/C34H34FN5O5/c1-18-12-20-4-7-24(18)19(2)17-45-33(44)39-22-6-9-27(34(10-11-34)32(42)43)21(13-22)16-40(3)31(41)29(20)38-23-5-8-25-26(14-23)28(35)15-37-30(25)36/h4-9,12-15,19,29,38H,10-11,16-17H2,1-3H3,(H2,36,37)(H,39,44)(H,42,43)/t19-,29+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of activated protein C (unknown origin) at 37 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Vitamin K-dependent protein C
(Homo sapiens (Human)) | BDBM50192770

(CHEMBL3956096)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCCCC1)C(O)=O |r| Show InChI InChI=1S/C37H40FN5O5/c1-21-15-23-7-10-27(21)22(2)20-48-36(47)42-25-9-12-30(37(35(45)46)13-5-4-6-14-37)24(16-25)19-43(3)34(44)32(23)41-26-8-11-28-29(17-26)31(38)18-40-33(28)39/h7-12,15-18,22,32,41H,4-6,13-14,19-20H2,1-3H3,(H2,39,40)(H,42,47)(H,45,46)/t22-,32+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
| Article
PubMed
| 155 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of activated protein C (unknown origin) at 37 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Vitamin K-dependent protein C
(Homo sapiens (Human)) | BDBM50192766

(CHEMBL3967204)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CC1)c1nnn[nH]1 |r| Show InChI InChI=1S/C34H34FN9O3/c1-18-12-20-4-7-24(18)19(2)17-47-33(46)39-22-6-9-27(34(10-11-34)32-40-42-43-41-32)21(13-22)16-44(3)31(45)29(20)38-23-5-8-25-26(14-23)28(35)15-37-30(25)36/h4-9,12-15,19,29,38H,10-11,16-17H2,1-3H3,(H2,36,37)(H,39,46)(H,40,41,42,43)/t19-,29+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 175 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of activated protein C (unknown origin) at 37 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Vitamin K-dependent protein C
(Homo sapiens (Human)) | BDBM50192764

(CHEMBL3934882)Show SMILES C[C@H]1COC(=O)Nc2ccc(C3CC3)c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2 |r| Show InChI InChI=1S/C33H34FN5O3/c1-18-12-21-6-9-25(18)19(2)17-42-33(41)38-23-7-10-26(20-4-5-20)22(13-23)16-39(3)32(40)30(21)37-24-8-11-27-28(14-24)29(34)15-36-31(27)35/h6-15,19-20,30,37H,4-5,16-17H2,1-3H3,(H2,35,36)(H,38,41)/t19-,30+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of activated protein C (unknown origin) at 37 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Vitamin K-dependent protein C
(Homo sapiens (Human)) | BDBM50192767

(CHEMBL3984725)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCCC1)C(O)=O |r| Show InChI InChI=1S/C36H38FN5O5/c1-20-14-22-6-9-26(20)21(2)19-47-35(46)41-24-8-11-29(36(34(44)45)12-4-5-13-36)23(15-24)18-42(3)33(43)31(22)40-25-7-10-27-28(16-25)30(37)17-39-32(27)38/h6-11,14-17,21,31,40H,4-5,12-13,18-19H2,1-3H3,(H2,38,39)(H,41,46)(H,44,45)/t21-,31+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of activated protein C (unknown origin) at 37 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Vitamin K-dependent protein C
(Homo sapiens (Human)) | BDBM50192768

(CHEMBL3900166)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCC1)C(O)=O |r| Show InChI InChI=1S/C35H36FN5O5/c1-19-13-21-5-8-25(19)20(2)18-46-34(45)40-23-7-10-28(35(33(43)44)11-4-12-35)22(14-23)17-41(3)32(42)30(21)39-24-6-9-26-27(15-24)29(36)16-38-31(26)37/h5-10,13-16,20,30,39H,4,11-12,17-18H2,1-3H3,(H2,37,38)(H,40,45)(H,43,44)/t20-,30+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 725 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of activated protein C (unknown origin) at 37 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50192768

(CHEMBL3900166)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCC1)C(O)=O |r| Show InChI InChI=1S/C35H36FN5O5/c1-19-13-21-5-8-25(19)20(2)18-46-34(45)40-23-7-10-28(35(33(43)44)11-4-12-35)22(14-23)17-41(3)32(42)30(21)39-24-6-9-26-27(15-24)29(36)16-38-31(26)37/h5-10,13-16,20,30,39H,4,11-12,17-18H2,1-3H3,(H2,37,38)(H,40,45)(H,43,44)/t20-,30+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 850 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a (unknown origin) at 25 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50192770

(CHEMBL3956096)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCCCC1)C(O)=O |r| Show InChI InChI=1S/C37H40FN5O5/c1-21-15-23-7-10-27(21)22(2)20-48-36(47)42-25-9-12-30(37(35(45)46)13-5-4-6-14-37)24(16-25)19-43(3)34(44)32(23)41-26-8-11-28-29(17-26)31(38)18-40-33(28)39/h7-12,15-18,22,32,41H,4-6,13-14,19-20H2,1-3H3,(H2,39,40)(H,42,47)(H,45,46)/t22-,32+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
| Article
PubMed
| 900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a (unknown origin) at 25 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Kallikrein-1
(Homo sapiens (Human)) | BDBM50192766

(CHEMBL3967204)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CC1)c1nnn[nH]1 |r| Show InChI InChI=1S/C34H34FN9O3/c1-18-12-20-4-7-24(18)19(2)17-47-33(46)39-22-6-9-27(34(10-11-34)32-40-42-43-41-32)21(13-22)16-44(3)31(45)29(20)38-23-5-8-25-26(14-23)28(35)15-37-30(25)36/h4-9,12-15,19,29,38H,10-11,16-17H2,1-3H3,(H2,36,37)(H,39,46)(H,40,41,42,43)/t19-,29+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 960 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of human kallikrein1 at 37 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50192766

(CHEMBL3967204)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CC1)c1nnn[nH]1 |r| Show InChI InChI=1S/C34H34FN9O3/c1-18-12-20-4-7-24(18)19(2)17-47-33(46)39-22-6-9-27(34(10-11-34)32-40-42-43-41-32)21(13-22)16-44(3)31(45)29(20)38-23-5-8-25-26(14-23)28(35)15-37-30(25)36/h4-9,12-15,19,29,38H,10-11,16-17H2,1-3H3,(H2,36,37)(H,39,46)(H,40,41,42,43)/t19-,29+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 1.05E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a (unknown origin) at 25 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Kallikrein-1
(Homo sapiens (Human)) | BDBM50192771

(CHEMBL3898371)Show SMILES [H][C@]1(C)COC(=O)Nc2ccc(c(CN(C)C(=O)[C@]([H])(Nc3ccc4c(c3)c(F)c[nH]c4=N)c3ccc1c(C)c3)c2)C1(CC1)C(O)=O |r| Show InChI InChI=1S/C34H34FN5O5/c1-18-12-20-4-7-24(18)19(2)17-45-33(44)39-22-6-9-27(34(10-11-34)32(42)43)21(13-22)16-40(3)31(41)29(20)38-23-5-8-25-26(14-23)28(35)15-37-30(25)36/h4-9,12-15,19,29,38H,10-11,16-17H2,1-3H3,(H2,36,37)(H,39,44)(H,42,43)/t19-,29+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of human kallikrein1 at 37 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Kallikrein-1
(Homo sapiens (Human)) | BDBM50192768

(CHEMBL3900166)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCC1)C(O)=O |r| Show InChI InChI=1S/C35H36FN5O5/c1-19-13-21-5-8-25(19)20(2)18-46-34(45)40-23-7-10-28(35(33(43)44)11-4-12-35)22(14-23)17-41(3)32(42)30(21)39-24-6-9-26-27(15-24)29(36)16-38-31(26)37/h5-10,13-16,20,30,39H,4,11-12,17-18H2,1-3H3,(H2,37,38)(H,40,45)(H,43,44)/t20-,30+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 1.15E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of human kallikrein1 at 37 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Kallikrein-1
(Homo sapiens (Human)) | BDBM50192770

(CHEMBL3956096)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCCCC1)C(O)=O |r| Show InChI InChI=1S/C37H40FN5O5/c1-21-15-23-7-10-27(21)22(2)20-48-36(47)42-25-9-12-30(37(35(45)46)13-5-4-6-14-37)24(16-25)19-43(3)34(44)32(23)41-26-8-11-28-29(17-26)31(38)18-40-33(28)39/h7-12,15-18,22,32,41H,4-6,13-14,19-20H2,1-3H3,(H2,39,40)(H,42,47)(H,45,46)/t22-,32+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
| Article
PubMed
| 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of human kallikrein1 at 37 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50192765

(CHEMBL3895930)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCOCC1)C(O)=O |r| Show InChI InChI=1S/C36H38FN5O6/c1-20-14-22-4-7-26(20)21(2)19-48-35(46)41-24-6-9-29(36(34(44)45)10-12-47-13-11-36)23(15-24)18-42(3)33(43)31(22)40-25-5-8-27-28(16-25)30(37)17-39-32(27)38/h4-9,14-17,21,31,40H,10-13,18-19H2,1-3H3,(H2,38,39)(H,41,46)(H,44,45)/t21-,31+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a (unknown origin) at 25 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Kallikrein-1
(Homo sapiens (Human)) | BDBM50192767

(CHEMBL3984725)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCCC1)C(O)=O |r| Show InChI InChI=1S/C36H38FN5O5/c1-20-14-22-6-9-26(20)21(2)19-47-35(46)41-24-8-11-29(36(34(44)45)12-4-5-13-36)23(15-24)18-42(3)33(43)31(22)40-25-7-10-27-28(16-25)30(37)17-39-32(27)38/h6-11,14-17,21,31,40H,4-5,12-13,18-19H2,1-3H3,(H2,38,39)(H,41,46)(H,44,45)/t21-,31+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of human kallikrein1 at 37 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Vitamin K-dependent protein C
(Homo sapiens (Human)) | BDBM50192765

(CHEMBL3895930)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCOCC1)C(O)=O |r| Show InChI InChI=1S/C36H38FN5O6/c1-20-14-22-4-7-26(20)21(2)19-48-35(46)41-24-6-9-29(36(34(44)45)10-12-47-13-11-36)23(15-24)18-42(3)33(43)31(22)40-25-5-8-27-28(16-25)30(37)17-39-32(27)38/h4-9,14-17,21,31,40H,10-13,18-19H2,1-3H3,(H2,38,39)(H,41,46)(H,44,45)/t21-,31+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of activated protein C (unknown origin) at 37 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50192767

(CHEMBL3984725)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCCC1)C(O)=O |r| Show InChI InChI=1S/C36H38FN5O5/c1-20-14-22-6-9-26(20)21(2)19-47-35(46)41-24-8-11-29(36(34(44)45)12-4-5-13-36)23(15-24)18-42(3)33(43)31(22)40-25-7-10-27-28(16-25)30(37)17-39-32(27)38/h6-11,14-17,21,31,40H,4-5,12-13,18-19H2,1-3H3,(H2,38,39)(H,41,46)(H,44,45)/t21-,31+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a (unknown origin) at 25 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50192764

(CHEMBL3934882)Show SMILES C[C@H]1COC(=O)Nc2ccc(C3CC3)c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2 |r| Show InChI InChI=1S/C33H34FN5O3/c1-18-12-21-6-9-25(18)19(2)17-42-33(41)38-23-7-10-26(20-4-5-20)22(13-23)16-39(3)32(40)30(21)37-24-8-11-27-28(14-24)29(34)15-36-31(27)35/h6-15,19-20,30,37H,4-5,16-17H2,1-3H3,(H2,35,36)(H,38,41)/t19-,30+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a (unknown origin) at 25 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Kallikrein-1
(Homo sapiens (Human)) | BDBM50192765

(CHEMBL3895930)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCOCC1)C(O)=O |r| Show InChI InChI=1S/C36H38FN5O6/c1-20-14-22-4-7-26(20)21(2)19-48-35(46)41-24-6-9-29(36(34(44)45)10-12-47-13-11-36)23(15-24)18-42(3)33(43)31(22)40-25-5-8-27-28(16-25)30(37)17-39-32(27)38/h4-9,14-17,21,31,40H,10-13,18-19H2,1-3H3,(H2,38,39)(H,41,46)(H,44,45)/t21-,31+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 5.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of human kallikrein1 at 37 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50192771

(CHEMBL3898371)Show SMILES [H][C@]1(C)COC(=O)Nc2ccc(c(CN(C)C(=O)[C@]([H])(Nc3ccc4c(c3)c(F)c[nH]c4=N)c3ccc1c(C)c3)c2)C1(CC1)C(O)=O |r| Show InChI InChI=1S/C34H34FN5O5/c1-18-12-20-4-7-24(18)19(2)17-45-33(44)39-22-6-9-27(34(10-11-34)32(42)43)21(13-22)16-40(3)31(41)29(20)38-23-5-8-25-26(14-23)28(35)15-37-30(25)36/h4-9,12-15,19,29,38H,10-11,16-17H2,1-3H3,(H2,36,37)(H,39,44)(H,42,43)/t19-,29+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 6.65E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of factor 10a (unknown origin) at 25 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50192768

(CHEMBL3900166)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCC1)C(O)=O |r| Show InChI InChI=1S/C35H36FN5O5/c1-19-13-21-5-8-25(19)20(2)18-46-34(45)40-23-7-10-28(35(33(43)44)11-4-12-35)22(14-23)17-41(3)32(42)30(21)39-24-6-9-26-27(15-24)29(36)16-38-31(26)37/h5-10,13-16,20,30,39H,4,11-12,17-18H2,1-3H3,(H2,37,38)(H,40,45)(H,43,44)/t20-,30+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| >1.33E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of thrombin (unknown origin) at 25 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50192771

(CHEMBL3898371)Show SMILES [H][C@]1(C)COC(=O)Nc2ccc(c(CN(C)C(=O)[C@]([H])(Nc3ccc4c(c3)c(F)c[nH]c4=N)c3ccc1c(C)c3)c2)C1(CC1)C(O)=O |r| Show InChI InChI=1S/C34H34FN5O5/c1-18-12-20-4-7-24(18)19(2)17-45-33(44)39-22-6-9-27(34(10-11-34)32(42)43)21(13-22)16-40(3)31(41)29(20)38-23-5-8-25-26(14-23)28(35)15-37-30(25)36/h4-9,12-15,19,29,38H,10-11,16-17H2,1-3H3,(H2,36,37)(H,39,44)(H,42,43)/t19-,29+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| >1.33E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of thrombin (unknown origin) at 25 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50192770

(CHEMBL3956096)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCCCC1)C(O)=O |r| Show InChI InChI=1S/C37H40FN5O5/c1-21-15-23-7-10-27(21)22(2)20-48-36(47)42-25-9-12-30(37(35(45)46)13-5-4-6-14-37)24(16-25)19-43(3)34(44)32(23)41-26-8-11-28-29(17-26)31(38)18-40-33(28)39/h7-12,15-18,22,32,41H,4-6,13-14,19-20H2,1-3H3,(H2,39,40)(H,42,47)(H,45,46)/t22-,32+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
| Article
PubMed
| >1.33E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of thrombin (unknown origin) at 25 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50192765

(CHEMBL3895930)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCOCC1)C(O)=O |r| Show InChI InChI=1S/C36H38FN5O6/c1-20-14-22-4-7-26(20)21(2)19-48-35(46)41-24-6-9-29(36(34(44)45)10-12-47-13-11-36)23(15-24)18-42(3)33(43)31(22)40-25-5-8-27-28(16-25)30(37)17-39-32(27)38/h4-9,14-17,21,31,40H,10-13,18-19H2,1-3H3,(H2,38,39)(H,41,46)(H,44,45)/t21-,31+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.33E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of thrombin (unknown origin) at 25 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50192767

(CHEMBL3984725)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCCC1)C(O)=O |r| Show InChI InChI=1S/C36H38FN5O5/c1-20-14-22-6-9-26(20)21(2)19-47-35(46)41-24-8-11-29(36(34(44)45)12-4-5-13-36)23(15-24)18-42(3)33(43)31(22)40-25-7-10-27-28(16-25)30(37)17-39-32(27)38/h6-11,14-17,21,31,40H,4-5,12-13,18-19H2,1-3H3,(H2,38,39)(H,41,46)(H,44,45)/t21-,31+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| >1.33E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of thrombin (unknown origin) at 25 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50192766

(CHEMBL3967204)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CC1)c1nnn[nH]1 |r| Show InChI InChI=1S/C34H34FN9O3/c1-18-12-20-4-7-24(18)19(2)17-47-33(46)39-22-6-9-27(34(10-11-34)32-40-42-43-41-32)21(13-22)16-44(3)31(45)29(20)38-23-5-8-25-26(14-23)28(35)15-37-30(25)36/h4-9,12-15,19,29,38H,10-11,16-17H2,1-3H3,(H2,36,37)(H,39,46)(H,40,41,42,43)/t19-,29+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| >1.33E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of thrombin (unknown origin) at 25 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50192764

(CHEMBL3934882)Show SMILES C[C@H]1COC(=O)Nc2ccc(C3CC3)c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2 |r| Show InChI InChI=1S/C33H34FN5O3/c1-18-12-21-6-9-25(18)19(2)17-42-33(41)38-23-7-10-26(20-4-5-20)22(13-23)16-39(3)32(40)30(21)37-24-8-11-27-28(14-24)29(34)15-36-31(27)35/h6-15,19-20,30,37H,4-5,16-17H2,1-3H3,(H2,35,36)(H,38,41)/t19-,30+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of thrombin (unknown origin) at 25 degC by chromogenic substrate assay |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data