 Found 65 hits of Enzyme Inhibition Constant Data
Found 65 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Aurora kinase A
(Homo sapiens (Human)) | BDBM50206864

(CHEMBL3903916)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccc3F)n2)n[nH]1 Show InChI InChI=1S/C19H19FN6O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(14(12)11-21)22-7-8-27-16-6-4-3-5-15(16)20/h3-6,9-10H,7-8H2,1-2H3,(H3,22,23,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Aurora A kinase derived from human Hela cells using kemptide as substrate in presence of [c-32P]ATP |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50343939
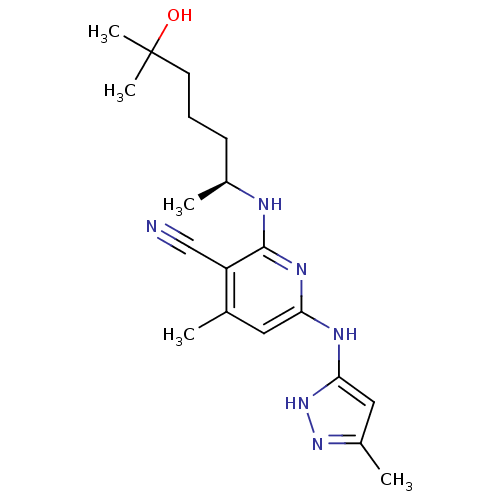
((S)-2-(6-hydroxy-6-methylheptan-2-ylamino)-4-methy...)Show SMILES C[C@@H](CCCC(C)(C)O)Nc1nc(Nc2cc(C)n[nH]2)cc(C)c1C#N |r| Show InChI InChI=1S/C19H28N6O/c1-12-9-16(22-17-10-14(3)24-25-17)23-18(15(12)11-20)21-13(2)7-6-8-19(4,5)26/h9-10,13,26H,6-8H2,1-5H3,(H3,21,22,23,24,25)/t13-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Aurora A kinase derived from human Hela cells using kemptide as substrate in presence of [c-32P]ATP |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50206860

(CHEMBL3974799)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccc3Cl)n2)n[nH]1 Show InChI InChI=1S/C19H19ClN6O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(14(12)11-21)22-7-8-27-16-6-4-3-5-15(16)20/h3-6,9-10H,7-8H2,1-2H3,(H3,22,23,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Aurora A kinase derived from human Hela cells using kemptide as substrate in presence of [c-32P]ATP |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50206865
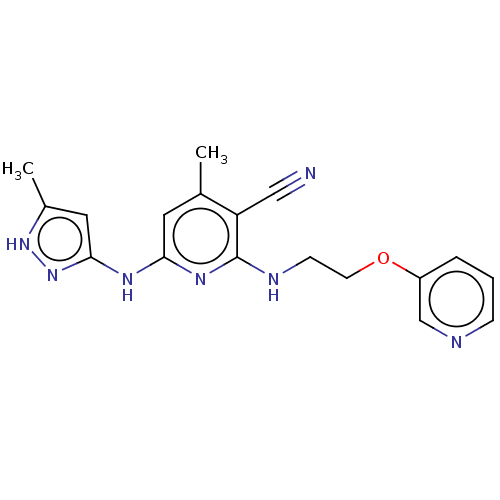
(CHEMBL3912923)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3cccnc3)n2)n[nH]1 Show InChI InChI=1S/C18H19N7O/c1-12-8-16(22-17-9-13(2)24-25-17)23-18(15(12)10-19)21-6-7-26-14-4-3-5-20-11-14/h3-5,8-9,11H,6-7H2,1-2H3,(H3,21,22,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Aurora A kinase derived from human Hela cells using kemptide as substrate in presence of [c-32P]ATP |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50206863
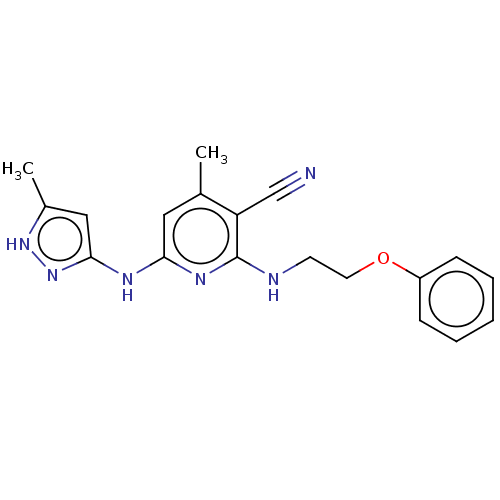
(CHEMBL3966303)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccc3)n2)n[nH]1 Show InChI InChI=1S/C19H20N6O/c1-13-10-17(22-18-11-14(2)24-25-18)23-19(16(13)12-20)21-8-9-26-15-6-4-3-5-7-15/h3-7,10-11H,8-9H2,1-2H3,(H3,21,22,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Aurora A kinase derived from human Hela cells using kemptide as substrate in presence of [c-32P]ATP |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50004205
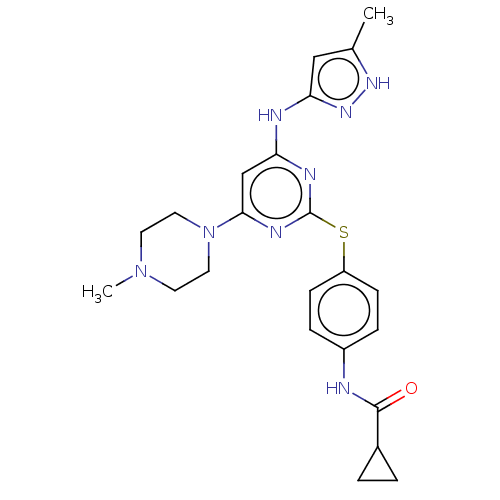
(MK-045 | MK-0457 | TOZASERTIB | US9249124, VX680 |...)Show SMILES CN1CCN(CC1)c1cc(Nc2cc(C)[nH]n2)nc(Sc2ccc(NC(=O)C3CC3)cc2)n1 Show InChI InChI=1S/C23H28N8OS/c1-15-13-20(29-28-15)25-19-14-21(31-11-9-30(2)10-12-31)27-23(26-19)33-18-7-5-17(6-8-18)24-22(32)16-3-4-16/h5-8,13-14,16H,3-4,9-12H2,1-2H3,(H,24,32)(H2,25,26,27,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Aurora A kinase derived from human Hela cells using kemptide as substrate in presence of [c-32P]ATP |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50206819
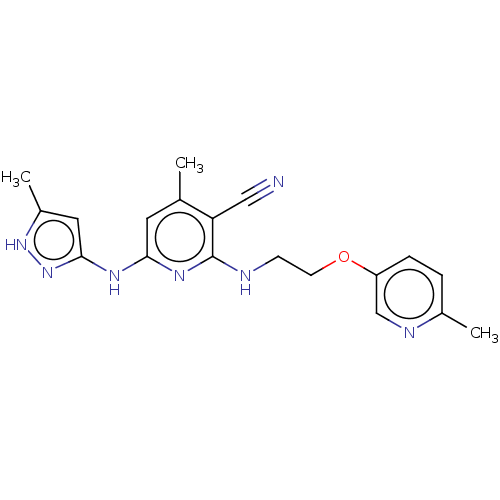
(CHEMBL3893914)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-8-17(23-18-9-14(3)25-26-18)24-19(16(12)10-20)21-6-7-27-15-5-4-13(2)22-11-15/h4-5,8-9,11H,6-7H2,1-3H3,(H3,21,23,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Aurora A kinase derived from human Hela cells using kemptide as substrate in presence of [c-32P]ATP |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50206859
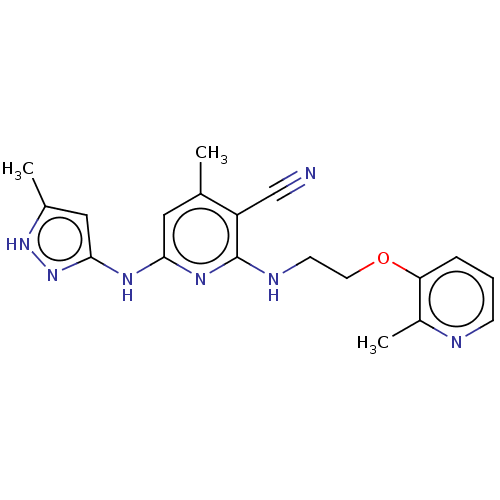
(CHEMBL3937431)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3cccnc3C)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(15(12)11-20)22-7-8-27-16-5-4-6-21-14(16)3/h4-6,9-10H,7-8H2,1-3H3,(H3,22,23,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Aurora A kinase derived from human Hela cells using kemptide as substrate in presence of [c-32P]ATP |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Ras-related protein Rab-7L1
(Homo sapiens (Human)) | BDBM50206819

(CHEMBL3893914)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-8-17(23-18-9-14(3)25-26-18)24-19(16(12)10-20)21-6-7-27-15-5-4-13(2)22-11-15/h4-5,8-9,11H,6-7H2,1-3H3,(H3,21,23,24,25,26) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of Aurora 2 kinase (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50206861

(CHEMBL3928420)Show SMILES CCc1ccc(OCCNc2nc(Nc3cc(C)[nH]n3)cc(C)c2C#N)cn1 Show InChI InChI=1S/C20H23N7O/c1-4-15-5-6-16(12-23-15)28-8-7-22-20-17(11-21)13(2)9-18(25-20)24-19-10-14(3)26-27-19/h5-6,9-10,12H,4,7-8H2,1-3H3,(H3,22,24,25,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Aurora A kinase derived from human Hela cells using kemptide as substrate in presence of [c-32P]ATP |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50206857
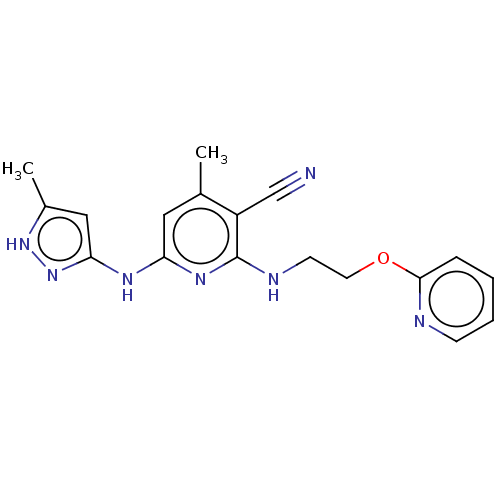
(CHEMBL3922893)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccn3)n2)n[nH]1 Show InChI InChI=1S/C18H19N7O/c1-12-9-15(22-16-10-13(2)24-25-16)23-18(14(12)11-19)21-7-8-26-17-5-3-4-6-20-17/h3-6,9-10H,7-8H2,1-2H3,(H3,21,22,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Aurora A kinase derived from human Hela cells using kemptide as substrate in presence of [c-32P]ATP |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Aurora kinase B
(Homo sapiens (Human)) | BDBM50206819

(CHEMBL3893914)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-8-17(23-18-9-14(3)25-26-18)24-19(16(12)10-20)21-6-7-27-15-5-4-13(2)22-11-15/h4-5,8-9,11H,6-7H2,1-3H3,(H3,21,23,24,25,26) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of Aurora 1 kinase (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50206862
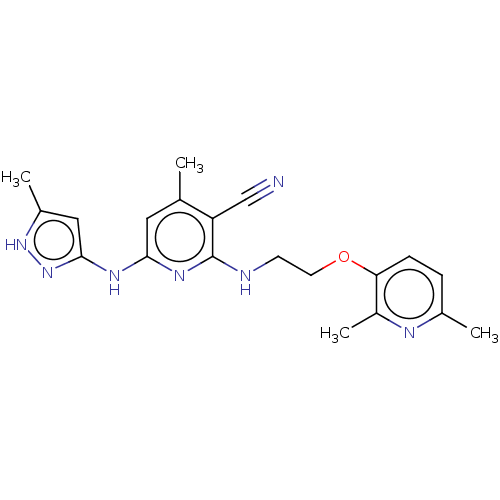
(CHEMBL3967136)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3C)n2)n[nH]1 Show InChI InChI=1S/C20H23N7O/c1-12-9-18(24-19-10-14(3)26-27-19)25-20(16(12)11-21)22-7-8-28-17-6-5-13(2)23-15(17)4/h5-6,9-10H,7-8H2,1-4H3,(H3,22,24,25,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Aurora A kinase derived from human Hela cells using kemptide as substrate in presence of [c-32P]ATP |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50206858
(CHEMBL3921905)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccncc3)n2)n[nH]1 Show InChI InChI=1S/C18H19N7O/c1-12-9-16(22-17-10-13(2)24-25-17)23-18(15(12)11-19)21-7-8-26-14-3-5-20-6-4-14/h3-6,9-10H,7-8H2,1-2H3,(H3,21,22,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 29 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Aurora A kinase derived from human Hela cells using kemptide as substrate in presence of [c-32P]ATP |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Yes
(Homo sapiens (Human)) | BDBM50206819

(CHEMBL3893914)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-8-17(23-18-9-14(3)25-26-18)24-19(16(12)10-20)21-6-7-27-15-5-4-13(2)22-11-15/h4-5,8-9,11H,6-7H2,1-3H3,(H3,21,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 79 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of YES (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Fyn
(Homo sapiens (Human)) | BDBM50206819

(CHEMBL3893914)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-8-17(23-18-9-14(3)25-26-18)24-19(16(12)10-20)21-6-7-27-15-5-4-13(2)22-11-15/h4-5,8-9,11H,6-7H2,1-3H3,(H3,21,23,24,25,26) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of FYN (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 1
(Homo sapiens (Human)) | BDBM50206819

(CHEMBL3893914)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-8-17(23-18-9-14(3)25-26-18)24-19(16(12)10-20)21-6-7-27-15-5-4-13(2)22-11-15/h4-5,8-9,11H,6-7H2,1-3H3,(H3,21,23,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of FGFR1 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50206819

(CHEMBL3893914)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-8-17(23-18-9-14(3)25-26-18)24-19(16(12)10-20)21-6-7-27-15-5-4-13(2)22-11-15/h4-5,8-9,11H,6-7H2,1-3H3,(H3,21,23,24,25,26) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of cSRC (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lyn
(Homo sapiens (Human)) | BDBM50206819

(CHEMBL3893914)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-8-17(23-18-9-14(3)25-26-18)24-19(16(12)10-20)21-6-7-27-15-5-4-13(2)22-11-15/h4-5,8-9,11H,6-7H2,1-3H3,(H3,21,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of LYN (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50206819

(CHEMBL3893914)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-8-17(23-18-9-14(3)25-26-18)24-19(16(12)10-20)21-6-7-27-15-5-4-13(2)22-11-15/h4-5,8-9,11H,6-7H2,1-3H3,(H3,21,23,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of KDR (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50206860

(CHEMBL3974799)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccc3Cl)n2)n[nH]1 Show InChI InChI=1S/C19H19ClN6O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(14(12)11-21)22-7-8-27-16-6-4-3-5-15(16)20/h3-6,9-10H,7-8H2,1-2H3,(H3,22,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <200 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50206863

(CHEMBL3966303)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccc3)n2)n[nH]1 Show InChI InChI=1S/C19H20N6O/c1-13-10-17(22-18-11-14(2)24-25-18)23-19(16(13)12-20)21-8-9-26-15-6-4-3-5-7-15/h3-7,10-11H,8-9H2,1-2H3,(H3,21,22,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <400 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50343939

((S)-2-(6-hydroxy-6-methylheptan-2-ylamino)-4-methy...)Show SMILES C[C@@H](CCCC(C)(C)O)Nc1nc(Nc2cc(C)n[nH]2)cc(C)c1C#N |r| Show InChI InChI=1S/C19H28N6O/c1-12-9-16(22-17-10-14(3)24-25-17)23-18(15(12)11-20)21-13(2)7-6-8-19(4,5)26/h9-10,13,26H,6-8H2,1-5H3,(H3,21,22,23,24,25)/t13-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <400 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50206858

(CHEMBL3921905)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccncc3)n2)n[nH]1 Show InChI InChI=1S/C18H19N7O/c1-12-9-16(22-17-10-13(2)24-25-17)23-18(15(12)11-19)21-7-8-26-14-3-5-20-6-4-14/h3-6,9-10H,7-8H2,1-2H3,(H3,21,22,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <400 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50206857

(CHEMBL3922893)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccn3)n2)n[nH]1 Show InChI InChI=1S/C18H19N7O/c1-12-9-15(22-16-10-13(2)24-25-16)23-18(14(12)11-19)21-7-8-26-17-5-3-4-6-20-17/h3-6,9-10H,7-8H2,1-2H3,(H3,21,22,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <400 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50206864

(CHEMBL3903916)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccc3F)n2)n[nH]1 Show InChI InChI=1S/C19H19FN6O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(14(12)11-21)22-7-8-27-16-6-4-3-5-15(16)20/h3-6,9-10H,7-8H2,1-2H3,(H3,22,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <400 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50206858

(CHEMBL3921905)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccncc3)n2)n[nH]1 Show InChI InChI=1S/C18H19N7O/c1-12-9-16(22-17-10-13(2)24-25-17)23-18(15(12)11-19)21-7-8-26-14-3-5-20-6-4-14/h3-6,9-10H,7-8H2,1-2H3,(H3,21,22,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <400 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50206865

(CHEMBL3912923)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3cccnc3)n2)n[nH]1 Show InChI InChI=1S/C18H19N7O/c1-12-8-16(22-17-9-13(2)24-25-17)23-18(15(12)10-19)21-6-7-26-14-4-3-5-20-11-14/h3-5,8-9,11H,6-7H2,1-2H3,(H3,21,22,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <400 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50206860

(CHEMBL3974799)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccc3Cl)n2)n[nH]1 Show InChI InChI=1S/C19H19ClN6O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(14(12)11-21)22-7-8-27-16-6-4-3-5-15(16)20/h3-6,9-10H,7-8H2,1-2H3,(H3,22,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <400 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50206860

(CHEMBL3974799)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccc3Cl)n2)n[nH]1 Show InChI InChI=1S/C19H19ClN6O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(14(12)11-21)22-7-8-27-16-6-4-3-5-15(16)20/h3-6,9-10H,7-8H2,1-2H3,(H3,22,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50206865

(CHEMBL3912923)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3cccnc3)n2)n[nH]1 Show InChI InChI=1S/C18H19N7O/c1-12-8-16(22-17-9-13(2)24-25-17)23-18(15(12)10-19)21-6-7-26-14-4-3-5-20-11-14/h3-5,8-9,11H,6-7H2,1-2H3,(H3,21,22,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50206861

(CHEMBL3928420)Show SMILES CCc1ccc(OCCNc2nc(Nc3cc(C)[nH]n3)cc(C)c2C#N)cn1 Show InChI InChI=1S/C20H23N7O/c1-4-15-5-6-16(12-23-15)28-8-7-22-20-17(11-21)13(2)9-18(25-20)24-19-10-14(3)26-27-19/h5-6,9-10,12H,4,7-8H2,1-3H3,(H3,22,24,25,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50206859

(CHEMBL3937431)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3cccnc3C)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(15(12)11-20)22-7-8-27-16-5-4-6-21-14(16)3/h4-6,9-10H,7-8H2,1-3H3,(H3,22,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50206860

(CHEMBL3974799)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccc3Cl)n2)n[nH]1 Show InChI InChI=1S/C19H19ClN6O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(14(12)11-21)22-7-8-27-16-6-4-3-5-15(16)20/h3-6,9-10H,7-8H2,1-2H3,(H3,22,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50206861

(CHEMBL3928420)Show SMILES CCc1ccc(OCCNc2nc(Nc3cc(C)[nH]n3)cc(C)c2C#N)cn1 Show InChI InChI=1S/C20H23N7O/c1-4-15-5-6-16(12-23-15)28-8-7-22-20-17(11-21)13(2)9-18(25-20)24-19-10-14(3)26-27-19/h5-6,9-10,12H,4,7-8H2,1-3H3,(H3,22,24,25,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50206861

(CHEMBL3928420)Show SMILES CCc1ccc(OCCNc2nc(Nc3cc(C)[nH]n3)cc(C)c2C#N)cn1 Show InChI InChI=1S/C20H23N7O/c1-4-15-5-6-16(12-23-15)28-8-7-22-20-17(11-21)13(2)9-18(25-20)24-19-10-14(3)26-27-19/h5-6,9-10,12H,4,7-8H2,1-3H3,(H3,22,24,25,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50206863

(CHEMBL3966303)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccc3)n2)n[nH]1 Show InChI InChI=1S/C19H20N6O/c1-13-10-17(22-18-11-14(2)24-25-18)23-19(16(13)12-20)21-8-9-26-15-6-4-3-5-7-15/h3-7,10-11H,8-9H2,1-2H3,(H3,21,22,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50206861

(CHEMBL3928420)Show SMILES CCc1ccc(OCCNc2nc(Nc3cc(C)[nH]n3)cc(C)c2C#N)cn1 Show InChI InChI=1S/C20H23N7O/c1-4-15-5-6-16(12-23-15)28-8-7-22-20-17(11-21)13(2)9-18(25-20)24-19-10-14(3)26-27-19/h5-6,9-10,12H,4,7-8H2,1-3H3,(H3,22,24,25,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50206864

(CHEMBL3903916)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccc3F)n2)n[nH]1 Show InChI InChI=1S/C19H19FN6O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(14(12)11-21)22-7-8-27-16-6-4-3-5-15(16)20/h3-6,9-10H,7-8H2,1-2H3,(H3,22,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50206862

(CHEMBL3967136)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3C)n2)n[nH]1 Show InChI InChI=1S/C20H23N7O/c1-12-9-18(24-19-10-14(3)26-27-19)25-20(16(12)11-21)22-7-8-28-17-6-5-13(2)23-15(17)4/h5-6,9-10H,7-8H2,1-4H3,(H3,22,24,25,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50343939

((S)-2-(6-hydroxy-6-methylheptan-2-ylamino)-4-methy...)Show SMILES C[C@@H](CCCC(C)(C)O)Nc1nc(Nc2cc(C)n[nH]2)cc(C)c1C#N |r| Show InChI InChI=1S/C19H28N6O/c1-12-9-16(22-17-10-14(3)24-25-17)23-18(15(12)11-20)21-13(2)7-6-8-19(4,5)26/h9-10,13,26H,6-8H2,1-5H3,(H3,21,22,23,24,25)/t13-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50206862

(CHEMBL3967136)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3C)n2)n[nH]1 Show InChI InChI=1S/C20H23N7O/c1-12-9-18(24-19-10-14(3)26-27-19)25-20(16(12)11-21)22-7-8-28-17-6-5-13(2)23-15(17)4/h5-6,9-10H,7-8H2,1-4H3,(H3,22,24,25,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50206859

(CHEMBL3937431)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3cccnc3C)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(15(12)11-20)22-7-8-27-16-5-4-6-21-14(16)3/h4-6,9-10H,7-8H2,1-3H3,(H3,22,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50206858

(CHEMBL3921905)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccncc3)n2)n[nH]1 Show InChI InChI=1S/C18H19N7O/c1-12-9-16(22-17-10-13(2)24-25-17)23-18(15(12)11-19)21-7-8-26-14-3-5-20-6-4-14/h3-6,9-10H,7-8H2,1-2H3,(H3,21,22,23,24,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50206819

(CHEMBL3893914)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-8-17(23-18-9-14(3)25-26-18)24-19(16(12)10-20)21-6-7-27-15-5-4-13(2)22-11-15/h4-5,8-9,11H,6-7H2,1-3H3,(H3,21,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50206859

(CHEMBL3937431)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3cccnc3C)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(15(12)11-20)22-7-8-27-16-5-4-6-21-14(16)3/h4-6,9-10H,7-8H2,1-3H3,(H3,22,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50206862

(CHEMBL3967136)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3C)n2)n[nH]1 Show InChI InChI=1S/C20H23N7O/c1-12-9-18(24-19-10-14(3)26-27-19)25-20(16(12)11-21)22-7-8-28-17-6-5-13(2)23-15(17)4/h5-6,9-10H,7-8H2,1-4H3,(H3,22,24,25,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50206819

(CHEMBL3893914)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-8-17(23-18-9-14(3)25-26-18)24-19(16(12)10-20)21-6-7-27-15-5-4-13(2)22-11-15/h4-5,8-9,11H,6-7H2,1-3H3,(H3,21,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50206819

(CHEMBL3893914)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-8-17(23-18-9-14(3)25-26-18)24-19(16(12)10-20)21-6-7-27-15-5-4-13(2)22-11-15/h4-5,8-9,11H,6-7H2,1-3H3,(H3,21,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50206819

(CHEMBL3893914)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-8-17(23-18-9-14(3)25-26-18)24-19(16(12)10-20)21-6-7-27-15-5-4-13(2)22-11-15/h4-5,8-9,11H,6-7H2,1-3H3,(H3,21,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50206859

(CHEMBL3937431)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3cccnc3C)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(15(12)11-20)22-7-8-27-16-5-4-6-21-14(16)3/h4-6,9-10H,7-8H2,1-3H3,(H3,22,23,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50206860

(CHEMBL3974799)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccc3Cl)n2)n[nH]1 Show InChI InChI=1S/C19H19ClN6O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(14(12)11-21)22-7-8-27-16-6-4-3-5-15(16)20/h3-6,9-10H,7-8H2,1-2H3,(H3,22,23,24,25,26) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50206857

(CHEMBL3922893)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccn3)n2)n[nH]1 Show InChI InChI=1S/C18H19N7O/c1-12-9-15(22-16-10-13(2)24-25-16)23-18(14(12)11-19)21-7-8-26-17-5-3-4-6-20-17/h3-6,9-10H,7-8H2,1-2H3,(H3,21,22,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50206863

(CHEMBL3966303)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccc3)n2)n[nH]1 Show InChI InChI=1S/C19H20N6O/c1-13-10-17(22-18-11-14(2)24-25-18)23-19(16(13)12-20)21-8-9-26-15-6-4-3-5-7-15/h3-7,10-11H,8-9H2,1-2H3,(H3,21,22,23,24,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50206864

(CHEMBL3903916)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccc3F)n2)n[nH]1 Show InChI InChI=1S/C19H19FN6O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(14(12)11-21)22-7-8-27-16-6-4-3-5-15(16)20/h3-6,9-10H,7-8H2,1-2H3,(H3,22,23,24,25,26) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50343939

((S)-2-(6-hydroxy-6-methylheptan-2-ylamino)-4-methy...)Show SMILES C[C@@H](CCCC(C)(C)O)Nc1nc(Nc2cc(C)n[nH]2)cc(C)c1C#N |r| Show InChI InChI=1S/C19H28N6O/c1-12-9-16(22-17-10-14(3)24-25-17)23-18(15(12)11-20)21-13(2)7-6-8-19(4,5)26/h9-10,13,26H,6-8H2,1-5H3,(H3,21,22,23,24,25)/t13-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50206865

(CHEMBL3912923)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3cccnc3)n2)n[nH]1 Show InChI InChI=1S/C18H19N7O/c1-12-8-16(22-17-9-13(2)24-25-17)23-18(15(12)10-19)21-6-7-26-14-4-3-5-20-11-14/h3-5,8-9,11H,6-7H2,1-2H3,(H3,21,22,23,24,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50206857

(CHEMBL3922893)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccccn3)n2)n[nH]1 Show InChI InChI=1S/C18H19N7O/c1-12-9-15(22-16-10-13(2)24-25-16)23-18(14(12)11-19)21-7-8-26-17-5-3-4-6-20-17/h3-6,9-10H,7-8H2,1-2H3,(H3,21,22,23,24,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50206861

(CHEMBL3928420)Show SMILES CCc1ccc(OCCNc2nc(Nc3cc(C)[nH]n3)cc(C)c2C#N)cn1 Show InChI InChI=1S/C20H23N7O/c1-4-15-5-6-16(12-23-15)28-8-7-22-20-17(11-21)13(2)9-18(25-20)24-19-10-14(3)26-27-19/h5-6,9-10,12H,4,7-8H2,1-3H3,(H3,22,24,25,26,27) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50206862

(CHEMBL3967136)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3C)n2)n[nH]1 Show InChI InChI=1S/C20H23N7O/c1-12-9-18(24-19-10-14(3)26-27-19)25-20(16(12)11-21)22-7-8-28-17-6-5-13(2)23-15(17)4/h5-6,9-10H,7-8H2,1-4H3,(H3,22,24,25,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50206819

(CHEMBL3893914)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-8-17(23-18-9-14(3)25-26-18)24-19(16(12)10-20)21-6-7-27-15-5-4-13(2)22-11-15/h4-5,8-9,11H,6-7H2,1-3H3,(H3,21,23,24,25,26) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50206862

(CHEMBL3967136)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3ccc(C)nc3C)n2)n[nH]1 Show InChI InChI=1S/C20H23N7O/c1-12-9-18(24-19-10-14(3)26-27-19)25-20(16(12)11-21)22-7-8-28-17-6-5-13(2)23-15(17)4/h5-6,9-10H,7-8H2,1-4H3,(H3,22,24,25,26,27) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50343939

((S)-2-(6-hydroxy-6-methylheptan-2-ylamino)-4-methy...)Show SMILES C[C@@H](CCCC(C)(C)O)Nc1nc(Nc2cc(C)n[nH]2)cc(C)c1C#N |r| Show InChI InChI=1S/C19H28N6O/c1-12-9-16(22-17-10-14(3)24-25-17)23-18(15(12)11-20)21-13(2)7-6-8-19(4,5)26/h9-10,13,26H,6-8H2,1-5H3,(H3,21,22,23,24,25)/t13-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50206859

(CHEMBL3937431)Show SMILES Cc1cc(Nc2cc(C)c(C#N)c(NCCOc3cccnc3C)n2)n[nH]1 Show InChI InChI=1S/C19H21N7O/c1-12-9-17(23-18-10-13(2)25-26-18)24-19(15(12)11-20)22-7-8-27-16-5-4-6-21-14(16)3/h4-6,9-10H,7-8H2,1-3H3,(H3,22,23,24,25,26) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50343939

((S)-2-(6-hydroxy-6-methylheptan-2-ylamino)-4-methy...)Show SMILES C[C@@H](CCCC(C)(C)O)Nc1nc(Nc2cc(C)n[nH]2)cc(C)c1C#N |r| Show InChI InChI=1S/C19H28N6O/c1-12-9-16(22-17-10-14(3)24-25-17)23-18(15(12)11-20)21-13(2)7-6-8-19(4,5)26/h9-10,13,26H,6-8H2,1-5H3,(H3,21,22,23,24,25)/t13-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
CXS Corporation
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
Bioorg Med Chem Lett 26: 5860-5862 (2016)
Article DOI: 10.1016/j.bmcl.2016.11.020
BindingDB Entry DOI: 10.7270/Q2WS8W80 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data