 Found 45 hits of Enzyme Inhibition Constant Data
Found 45 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kbeta in PTEN-deficient human MDA-MB-468 cells assessed as decrease in AKT phosphorylation at Ser473 measured after 2 hrs |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50239123
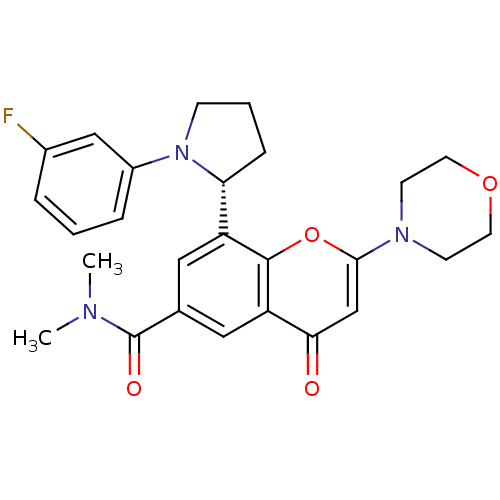
(CHEMBL4078233)Show SMILES CN(C)C(=O)c1cc([C@H]2CCCN2c2cccc(F)c2)c2oc(cc(=O)c2c1)N1CCOCC1 |r| Show InChI InChI=1S/C26H28FN3O4/c1-28(2)26(32)17-13-20(22-7-4-8-30(22)19-6-3-5-18(27)15-19)25-21(14-17)23(31)16-24(34-25)29-9-11-33-12-10-29/h3,5-6,13-16,22H,4,7-12H2,1-2H3/t22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kbeta in PTEN-deficient human MDA-MB-468 cells assessed as decrease in AKT phosphorylation at Ser473 measured after 2 hrs |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50239122

(CHEMBL4094771)Show SMILES CN(C)C(=O)c1cc(C2CCCN2c2cc(F)cc(F)c2)c2oc(cc(=O)c2c1)N1CCOCC1 Show InChI InChI=1S/C26H27F2N3O4/c1-29(2)26(33)16-10-20(22-4-3-5-31(22)19-13-17(27)12-18(28)14-19)25-21(11-16)23(32)15-24(35-25)30-6-8-34-9-7-30/h10-15,22H,3-9H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kbeta in PTEN-deficient human MDA-MB-468 cells assessed as decrease in AKT phosphorylation at Ser473 measured after 2 hrs |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50239120
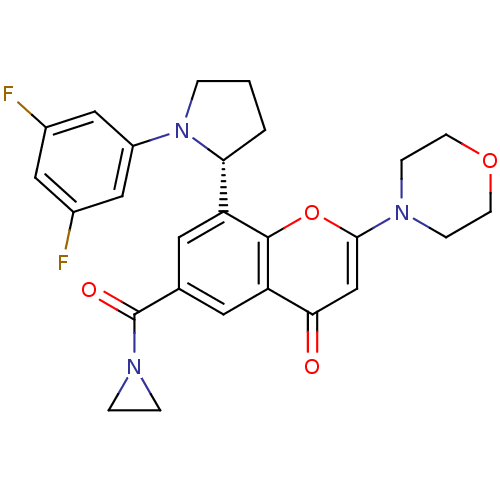
(CHEMBL4101005)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CC1 |r| Show InChI InChI=1S/C26H25F2N3O4/c27-17-12-18(28)14-19(13-17)31-3-1-2-22(31)20-10-16(26(33)30-4-5-30)11-21-23(32)15-24(35-25(20)21)29-6-8-34-9-7-29/h10-15,22H,1-9H2/t22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kbeta in PTEN-deficient human MDA-MB-468 cells assessed as decrease in AKT phosphorylation at Ser473 measured after 2 hrs |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50239126
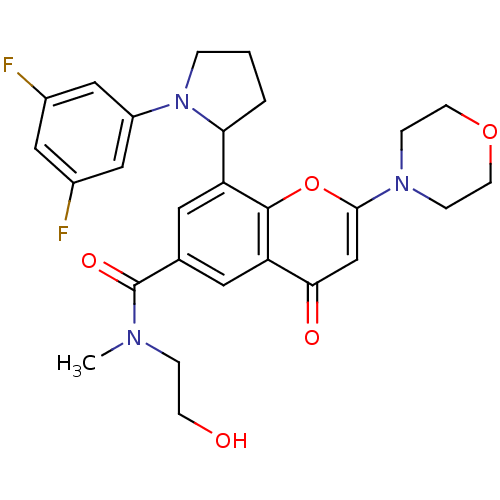
(CHEMBL4067312)Show SMILES CN(CCO)C(=O)c1cc(C2CCCN2c2cc(F)cc(F)c2)c2oc(cc(=O)c2c1)N1CCOCC1 Show InChI InChI=1S/C27H29F2N3O5/c1-30(5-8-33)27(35)17-11-21(23-3-2-4-32(23)20-14-18(28)13-19(29)15-20)26-22(12-17)24(34)16-25(37-26)31-6-9-36-10-7-31/h11-16,23,33H,2-10H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kbeta in PTEN-deficient human MDA-MB-468 cells assessed as decrease in AKT phosphorylation at Ser473 measured after 2 hrs |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50239121
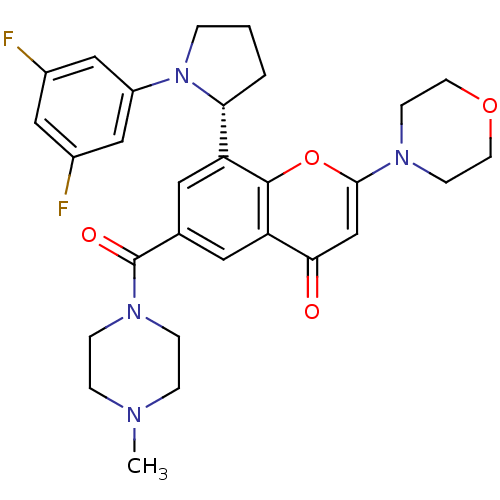
(CHEMBL4082650)Show SMILES CN1CCN(CC1)C(=O)c1cc([C@H]2CCCN2c2cc(F)cc(F)c2)c2oc(cc(=O)c2c1)N1CCOCC1 |r| Show InChI InChI=1S/C29H32F2N4O4/c1-32-5-7-34(8-6-32)29(37)19-13-23(25-3-2-4-35(25)22-16-20(30)15-21(31)17-22)28-24(14-19)26(36)18-27(39-28)33-9-11-38-12-10-33/h13-18,25H,2-12H2,1H3/t25-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kbeta in PTEN-deficient human MDA-MB-468 cells assessed as decrease in AKT phosphorylation at Ser473 measured after 2 hrs |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50239118
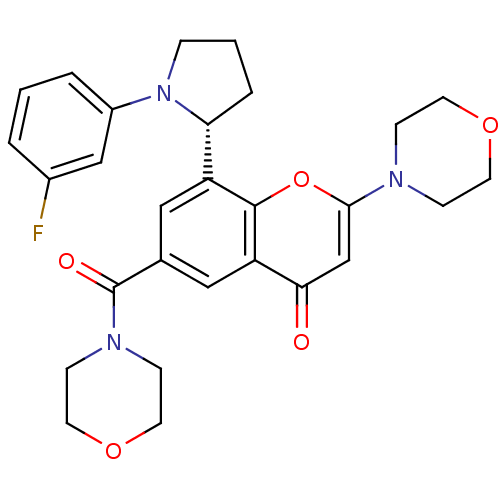
(CHEMBL4090811)Show SMILES Fc1cccc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H30FN3O5/c29-20-3-1-4-21(17-20)32-6-2-5-24(32)22-15-19(28(34)31-9-13-36-14-10-31)16-23-25(33)18-26(37-27(22)23)30-7-11-35-12-8-30/h1,3-4,15-18,24H,2,5-14H2/t24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 2.80 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kbeta in PTEN-deficient human MDA-MB-468 cells assessed as decrease in AKT phosphorylation at Ser473 measured after 2 hrs |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50239125
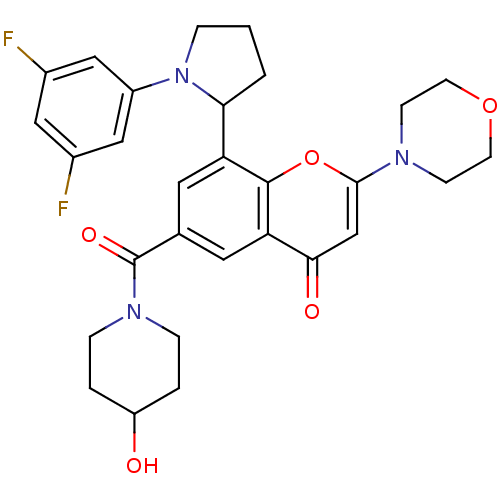
(CHEMBL4083021)Show SMILES OC1CCN(CC1)C(=O)c1cc(C2CCCN2c2cc(F)cc(F)c2)c2oc(cc(=O)c2c1)N1CCOCC1 Show InChI InChI=1S/C29H31F2N3O5/c30-19-14-20(31)16-21(15-19)34-5-1-2-25(34)23-12-18(29(37)33-6-3-22(35)4-7-33)13-24-26(36)17-27(39-28(23)24)32-8-10-38-11-9-32/h12-17,22,25,35H,1-11H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kbeta in PTEN-deficient human MDA-MB-468 cells assessed as decrease in AKT phosphorylation at Ser473 measured after 2 hrs |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50239127
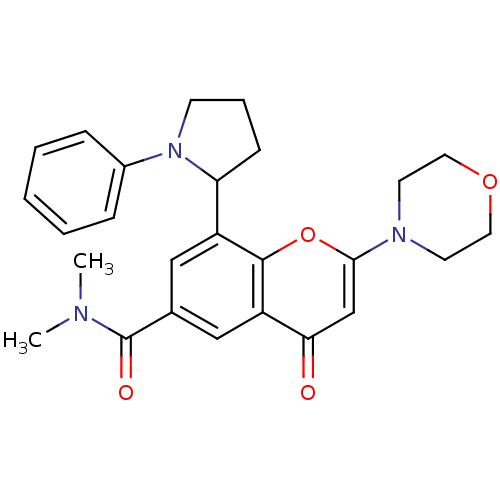
(CHEMBL4086125)Show SMILES CN(C)C(=O)c1cc(C2CCCN2c2ccccc2)c2oc(cc(=O)c2c1)N1CCOCC1 Show InChI InChI=1S/C26H29N3O4/c1-27(2)26(31)18-15-20(22-9-6-10-29(22)19-7-4-3-5-8-19)25-21(16-18)23(30)17-24(33-25)28-11-13-32-14-12-28/h3-5,7-8,15-17,22H,6,9-14H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kbeta in PTEN-deficient human MDA-MB-468 cells assessed as decrease in AKT phosphorylation at Ser473 measured after 2 hrs |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50239124
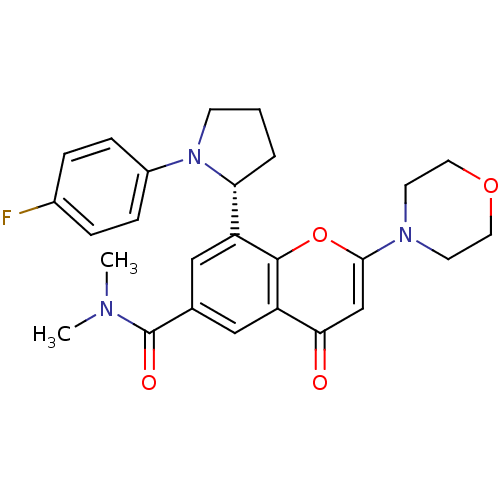
(CHEMBL4102511)Show SMILES CN(C)C(=O)c1cc([C@H]2CCCN2c2ccc(F)cc2)c2oc(cc(=O)c2c1)N1CCOCC1 |r| Show InChI InChI=1S/C26H28FN3O4/c1-28(2)26(32)17-14-20(22-4-3-9-30(22)19-7-5-18(27)6-8-19)25-21(15-17)23(31)16-24(34-25)29-10-12-33-13-11-29/h5-8,14-16,22H,3-4,9-13H2,1-2H3/t22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.40 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kbeta in PTEN-deficient human MDA-MB-468 cells assessed as decrease in AKT phosphorylation at Ser473 measured after 2 hrs |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50239119
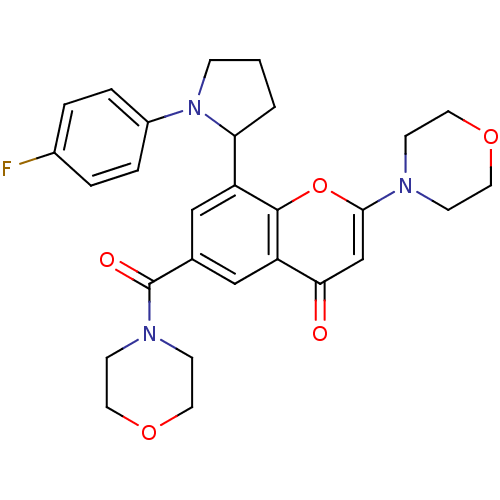
(CHEMBL4062879)Show SMILES Fc1ccc(cc1)N1CCCC1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 Show InChI InChI=1S/C28H30FN3O5/c29-20-3-5-21(6-4-20)32-7-1-2-24(32)22-16-19(28(34)31-10-14-36-15-11-31)17-23-25(33)18-26(37-27(22)23)30-8-12-35-13-9-30/h3-6,16-18,24H,1-2,7-15H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kbeta in PTEN-deficient human MDA-MB-468 cells assessed as decrease in AKT phosphorylation at Ser473 measured after 2 hrs |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50239118

(CHEMBL4090811)Show SMILES Fc1cccc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H30FN3O5/c29-20-3-1-4-21(17-20)32-6-2-5-24(32)22-15-19(28(34)31-9-13-36-14-10-31)16-23-25(33)18-26(37-27(22)23)30-7-11-35-12-8-30/h1,3-4,15-18,24H,2,5-14H2/t24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 5.10 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Displacement of [3H]baclofen from Gamma-aminobutyric acid type B receptor of rat brain membranes |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50239118

(CHEMBL4090811)Show SMILES Fc1cccc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H30FN3O5/c29-20-3-1-4-21(17-20)32-6-2-5-24(32)22-15-19(28(34)31-9-13-36-14-10-31)16-23-25(33)18-26(37-27(22)23)30-7-11-35-12-8-30/h1,3-4,15-18,24H,2,5-14H2/t24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 6.10 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PI3Kbeta using PIP2 as substrate preincubated for 20 mins followed by substrate addition measured after 80 mins in pr... |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50239124

(CHEMBL4102511)Show SMILES CN(C)C(=O)c1cc([C@H]2CCCN2c2ccc(F)cc2)c2oc(cc(=O)c2c1)N1CCOCC1 |r| Show InChI InChI=1S/C26H28FN3O4/c1-28(2)26(32)17-14-20(22-4-3-9-30(22)19-7-5-18(27)6-8-19)25-21(15-17)23(31)16-24(34-25)29-10-12-33-13-11-29/h5-8,14-16,22H,3-4,9-13H2,1-2H3/t22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.70 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PI3Kbeta using PIP2 as substrate preincubated for 20 mins followed by substrate addition measured after 80 mins in pr... |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 7.10 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PI3Kbeta using PIP2 as substrate preincubated for 20 mins followed by substrate addition measured after 80 mins in pr... |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50239124

(CHEMBL4102511)Show SMILES CN(C)C(=O)c1cc([C@H]2CCCN2c2ccc(F)cc2)c2oc(cc(=O)c2c1)N1CCOCC1 |r| Show InChI InChI=1S/C26H28FN3O4/c1-28(2)26(32)17-14-20(22-4-3-9-30(22)19-7-5-18(27)6-8-19)25-21(15-17)23(31)16-24(34-25)29-10-12-33-13-11-29/h5-8,14-16,22H,3-4,9-13H2,1-2H3/t22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8.20 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PI3Kdelta using PIP2 as substrate preincubated for 20 mins followed by substrate addition measured after 80 mins in p... |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 8.60 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PI3Kdelta using PIP2 as substrate preincubated for 20 mins followed by substrate addition measured after 80 mins in p... |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PI3Kalpha using PIP2 as substrate preincubated for 20 mins followed by substrate addition measured after 80 mins in p... |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta in human JeKo1 B cells assessed as decrease in AKT phosphorylation at Ser473 measured after 1 hr |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50239118

(CHEMBL4090811)Show SMILES Fc1cccc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H30FN3O5/c29-20-3-1-4-21(17-20)32-6-2-5-24(32)22-15-19(28(34)31-9-13-36-14-10-31)16-23-25(33)18-26(37-27(22)23)30-7-11-35-12-8-30/h1,3-4,15-18,24H,2,5-14H2/t24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PI3Kalpha using PIP2 as substrate preincubated for 20 mins followed by substrate addition measured after 80 mins in p... |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50239124

(CHEMBL4102511)Show SMILES CN(C)C(=O)c1cc([C@H]2CCCN2c2ccc(F)cc2)c2oc(cc(=O)c2c1)N1CCOCC1 |r| Show InChI InChI=1S/C26H28FN3O4/c1-28(2)26(32)17-14-20(22-4-3-9-30(22)19-7-5-18(27)6-8-19)25-21(15-17)23(31)16-24(34-25)29-10-12-33-13-11-29/h5-8,14-16,22H,3-4,9-13H2,1-2H3/t22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PI3Kalpha using PIP2 as substrate preincubated for 20 mins followed by substrate addition measured after 80 mins in p... |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Compound was evaluated for Kinetic constant for viral thymidine kinase of Herpes simplex virus (HSV) -2 |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50239124

(CHEMBL4102511)Show SMILES CN(C)C(=O)c1cc([C@H]2CCCN2c2ccc(F)cc2)c2oc(cc(=O)c2c1)N1CCOCC1 |r| Show InChI InChI=1S/C26H28FN3O4/c1-28(2)26(32)17-14-20(22-4-3-9-30(22)19-7-5-18(27)6-8-19)25-21(15-17)23(31)16-24(34-25)29-10-12-33-13-11-29/h5-8,14-16,22H,3-4,9-13H2,1-2H3/t22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 330 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PI3Kgamma using PIP2 as substrate preincubated for 20 mins followed by substrate addition measured after 80 mins in p... |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50239118

(CHEMBL4090811)Show SMILES Fc1cccc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H30FN3O5/c29-20-3-1-4-21(17-20)32-6-2-5-24(32)22-15-19(28(34)31-9-13-36-14-10-31)16-23-25(33)18-26(37-27(22)23)30-7-11-35-12-8-30/h1,3-4,15-18,24H,2,5-14H2/t24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PI3Kgamma using PIP2 as substrate preincubated for 20 mins followed by substrate addition measured after 80 mins in p... |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Mus musculus (Mouse)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Displacement of [3H]baclofen from Gamma-aminobutyric acid type B receptor of rat brain membranes |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Sphingosine kinase 1
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 2.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human SphK1 (1 to 384 residues) by ATP-Glo HTS assay |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Displacement of [3H]baclofen from Gamma-aminobutyric acid type B receptor of rat brain membranes |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Displacement of [3H]baclofen from Gamma-aminobutyric acid type B receptor of rat brain membranes |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase mTOR
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 4.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of mTOR (unknown origin) |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 4.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Displacement of [3H]baclofen from Gamma-aminobutyric acid type B receptor of rat brain membranes |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4-kinase beta
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human PIK4Cbeta (1 to 801 residues) by ADP Glo HTS assay |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Diacylglycerol kinase gamma
(Homo sapiens) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Displacement of [3H]baclofen from Gamma-aminobutyric acid type B receptor of rat brain membranes |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Choline/ethanolamine kinase
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Displacement of [3H]baclofen from Gamma-aminobutyric acid type B receptor of rat brain membranes |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Displacement of [3H]baclofen from Gamma-aminobutyric acid type B receptor of rat brain membranes |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Diacylglycerol kinase zeta
(Homo sapiens) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Displacement of [3H]baclofen from Gamma-aminobutyric acid type B receptor of rat brain membranes |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Diacylglycerol kinase beta
(Homo sapiens) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Displacement of [3H]baclofen from Gamma-aminobutyric acid type B receptor of rat brain membranes |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit beta
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human PI3K p85beta (1 to 724 residues) by ADP Glo HTS assay |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma (2 to 1102 residues) by ADP Glo HTS assay |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human PI4K2alpha (1 to 479 residues) by ADP Glo HTS assay |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Choline kinase alpha
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human CHKalpha (1 to 457 residues) by ADP Glo HTS assay |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Sphingosine kinase 2
(Homo sapiens (Human)) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human SPHK 2 (1 to 654 residues) by ADP Glo HTS assay |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50239117

(CHEMBL4060768)Show SMILES Fc1cc(F)cc(c1)N1CCC[C@@H]1c1cc(cc2c1oc(cc2=O)N1CCOCC1)C(=O)N1CCOCC1 |r| Show InChI InChI=1S/C28H29F2N3O5/c29-19-14-20(30)16-21(15-19)33-3-1-2-24(33)22-12-18(28(35)32-6-10-37-11-7-32)13-23-25(34)17-26(38-27(22)23)31-4-8-36-9-5-31/h12-17,24H,1-11H2/t24-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human PIP5K2alpha (1 to 406 residues) by ADP Glo HTS assay |
Bioorg Med Chem Lett 27: 1949-1954 (2017)
Article DOI: 10.1016/j.bmcl.2017.03.027
BindingDB Entry DOI: 10.7270/Q2SB47W8 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data