 Found 76 hits of Enzyme Inhibition Constant Data
Found 76 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283703
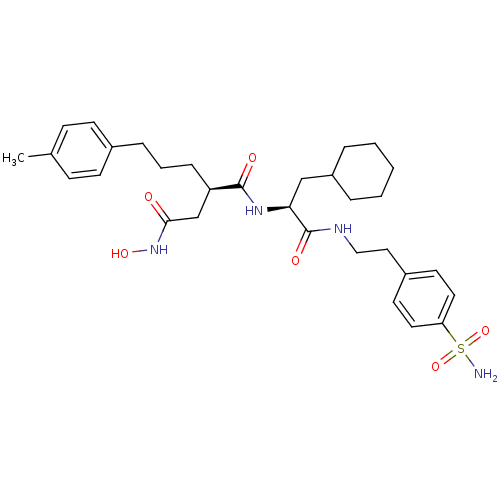
((R)-N*1*-{(S)-2-Cyclohexyl-1-[2-(4-sulfamoyl-pheny...)Show SMILES Cc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCc2ccc(cc2)S(N)(=O)=O)cc1 Show InChI InChI=1S/C31H44N4O6S/c1-22-10-12-23(13-11-22)8-5-9-26(21-29(36)35-39)30(37)34-28(20-25-6-3-2-4-7-25)31(38)33-19-18-24-14-16-27(17-15-24)42(32,40)41/h10-17,25-26,28,39H,2-9,18-21H2,1H3,(H,33,38)(H,34,37)(H,35,36)(H2,32,40,41)/t26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283705
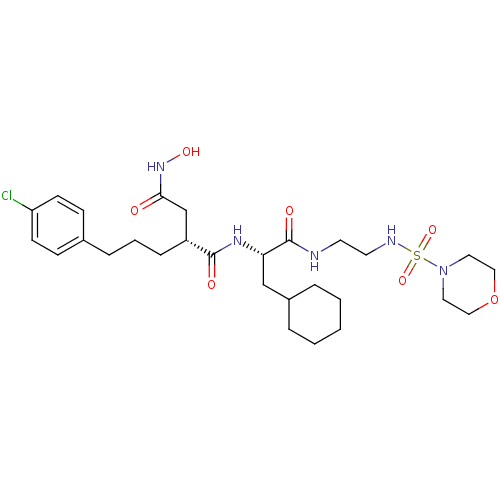
((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-{(S)-2-cyc...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(Cl)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCNS(=O)(=O)N1CCOCC1 Show InChI InChI=1S/C28H44ClN5O7S/c29-24-11-9-21(10-12-24)7-4-8-23(20-26(35)33-38)27(36)32-25(19-22-5-2-1-3-6-22)28(37)30-13-14-31-42(39,40)34-15-17-41-18-16-34/h9-12,22-23,25,31,38H,1-8,13-20H2,(H,30,37)(H,32,36)(H,33,35)/t23-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283715
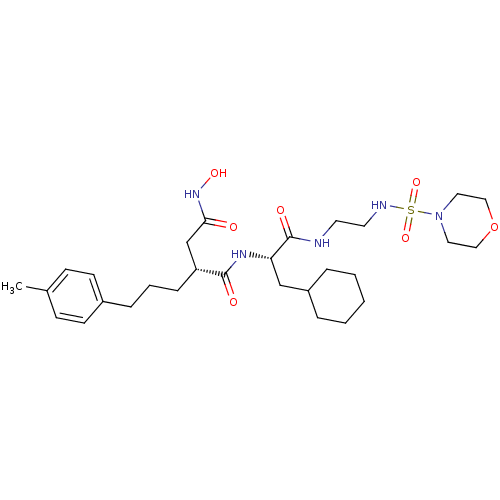
((R)-N*1*-{(S)-2-Cyclohexyl-1-[2-(morpholine-4-sulf...)Show SMILES Cc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCNS(=O)(=O)N2CCOCC2)cc1 Show InChI InChI=1S/C29H47N5O7S/c1-22-10-12-23(13-11-22)8-5-9-25(21-27(35)33-38)28(36)32-26(20-24-6-3-2-4-7-24)29(37)30-14-15-31-42(39,40)34-16-18-41-19-17-34/h10-13,24-26,31,38H,2-9,14-21H2,1H3,(H,30,37)(H,32,36)(H,33,35)/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283704
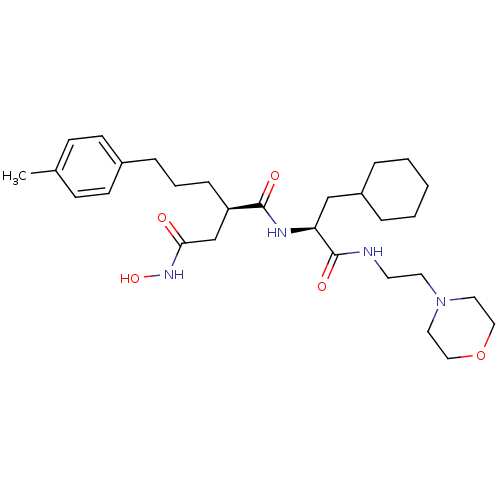
((R)-N*1*-[(S)-2-Cyclohexyl-1-(2-morpholin-4-yl-eth...)Show SMILES Cc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCN2CCOCC2)cc1 Show InChI InChI=1S/C29H46N4O5/c1-22-10-12-23(13-11-22)8-5-9-25(21-27(34)32-37)28(35)31-26(20-24-6-3-2-4-7-24)29(36)30-14-15-33-16-18-38-19-17-33/h10-13,24-26,37H,2-9,14-21H2,1H3,(H,30,36)(H,31,35)(H,32,34)/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283708
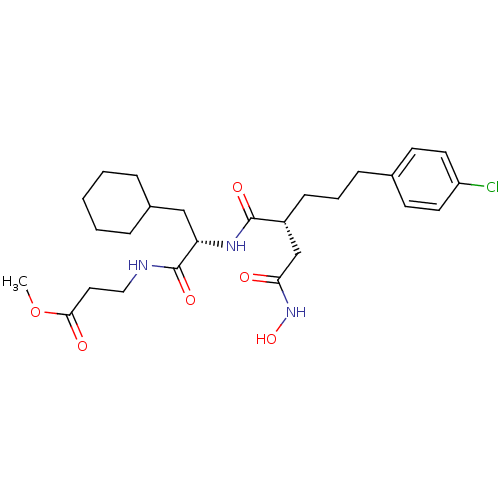
(3-{(S)-2-[(R)-5-(4-Chloro-phenyl)-2-hydroxycarbamo...)Show SMILES COC(=O)CCNC(=O)[C@H](CC1CCCCC1)NC(=O)[C@H](CCCc1ccc(Cl)cc1)CC(=O)NO Show InChI InChI=1S/C26H38ClN3O6/c1-36-24(32)14-15-28-26(34)22(16-19-6-3-2-4-7-19)29-25(33)20(17-23(31)30-35)9-5-8-18-10-12-21(27)13-11-18/h10-13,19-20,22,35H,2-9,14-17H2,1H3,(H,28,34)(H,29,33)(H,30,31)/t20-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283701
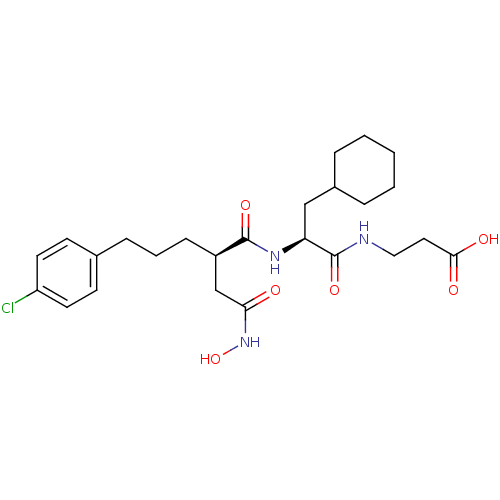
(3-{(S)-2-[(R)-5-(4-Chloro-phenyl)-2-hydroxycarbamo...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(Cl)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCC(O)=O Show InChI InChI=1S/C25H36ClN3O6/c26-20-11-9-17(10-12-20)7-4-8-19(16-22(30)29-35)24(33)28-21(15-18-5-2-1-3-6-18)25(34)27-14-13-23(31)32/h9-12,18-19,21,35H,1-8,13-16H2,(H,27,34)(H,28,33)(H,29,30)(H,31,32)/t19-,21+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283711
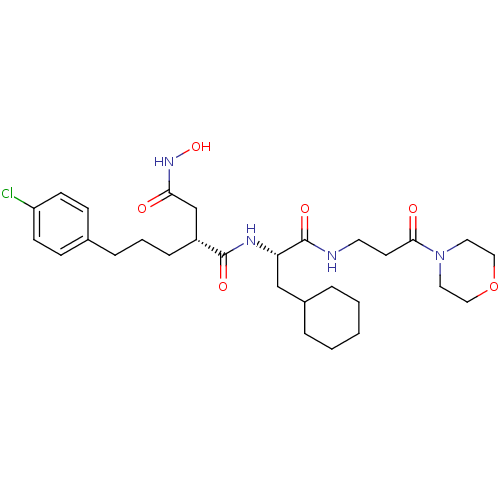
((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-[(S)-2-cyc...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(Cl)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCC(=O)N1CCOCC1 Show InChI InChI=1S/C29H43ClN4O6/c30-24-11-9-21(10-12-24)7-4-8-23(20-26(35)33-39)28(37)32-25(19-22-5-2-1-3-6-22)29(38)31-14-13-27(36)34-15-17-40-18-16-34/h9-12,22-23,25,39H,1-8,13-20H2,(H,31,38)(H,32,37)(H,33,35)/t23-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283710
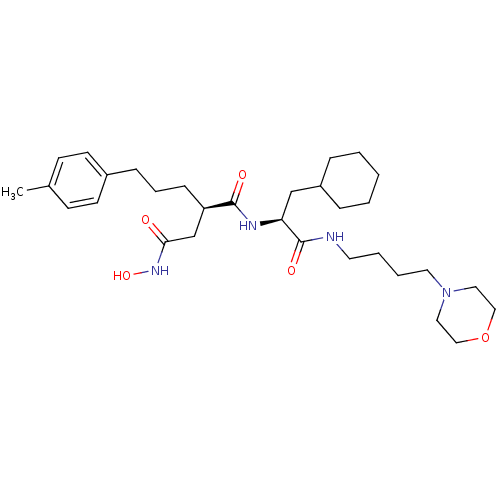
((R)-N*1*-[(S)-2-Cyclohexyl-1-(4-morpholin-4-yl-but...)Show SMILES Cc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCCCN2CCOCC2)cc1 Show InChI InChI=1S/C31H50N4O5/c1-24-12-14-25(15-13-24)10-7-11-27(23-29(36)34-39)30(37)33-28(22-26-8-3-2-4-9-26)31(38)32-16-5-6-17-35-18-20-40-21-19-35/h12-15,26-28,39H,2-11,16-23H2,1H3,(H,32,38)(H,33,37)(H,34,36)/t27-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283713
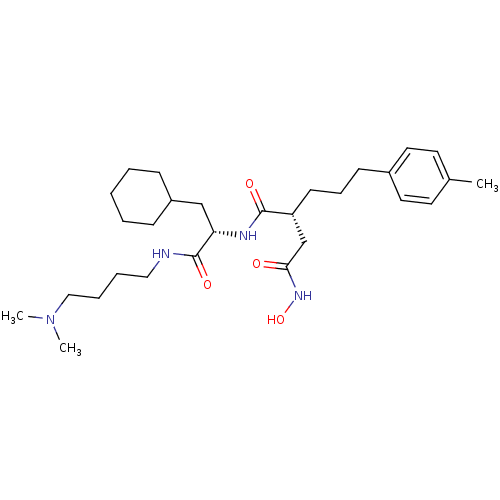
((R)-N*1*-[(S)-2-Cyclohexyl-1-(4-dimethylamino-buty...)Show SMILES CN(C)CCCCNC(=O)[C@H](CC1CCCCC1)NC(=O)[C@H](CCCc1ccc(C)cc1)CC(=O)NO Show InChI InChI=1S/C29H48N4O4/c1-22-14-16-23(17-15-22)12-9-13-25(21-27(34)32-37)28(35)31-26(20-24-10-5-4-6-11-24)29(36)30-18-7-8-19-33(2)3/h14-17,24-26,37H,4-13,18-21H2,1-3H3,(H,30,36)(H,31,35)(H,32,34)/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283702
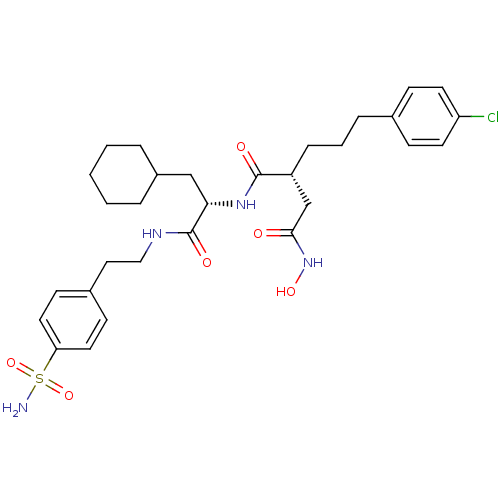
((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-{(S)-2-cyc...)Show SMILES NS(=O)(=O)c1ccc(CCNC(=O)[C@H](CC2CCCCC2)NC(=O)[C@H](CCCc2ccc(Cl)cc2)CC(=O)NO)cc1 Show InChI InChI=1S/C30H41ClN4O6S/c31-25-13-9-21(10-14-25)7-4-8-24(20-28(36)35-39)29(37)34-27(19-23-5-2-1-3-6-23)30(38)33-18-17-22-11-15-26(16-12-22)42(32,40)41/h9-16,23-24,27,39H,1-8,17-20H2,(H,33,38)(H,34,37)(H,35,36)(H2,32,40,41)/t24-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50101495
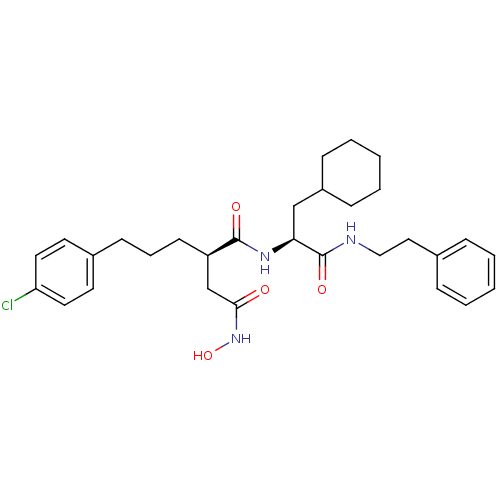
((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-((S)-2-cyc...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(Cl)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C30H40ClN3O4/c31-26-16-14-23(15-17-26)12-7-13-25(21-28(35)34-38)29(36)33-27(20-24-10-5-2-6-11-24)30(37)32-19-18-22-8-3-1-4-9-22/h1,3-4,8-9,14-17,24-25,27,38H,2,5-7,10-13,18-21H2,(H,32,37)(H,33,36)(H,34,35)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283707
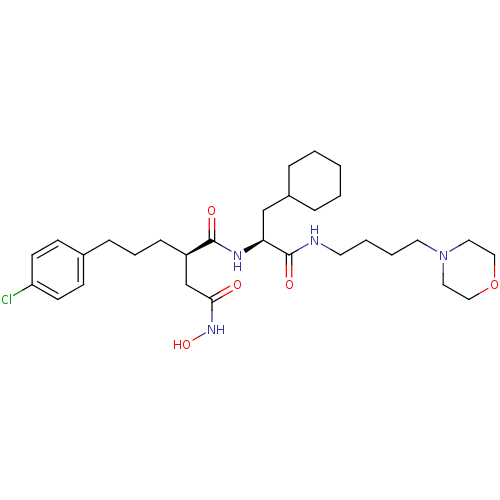
((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-[(S)-2-cyc...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(Cl)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCCCN1CCOCC1 Show InChI InChI=1S/C30H47ClN4O5/c31-26-13-11-23(12-14-26)9-6-10-25(22-28(36)34-39)29(37)33-27(21-24-7-2-1-3-8-24)30(38)32-15-4-5-16-35-17-19-40-20-18-35/h11-14,24-25,27,39H,1-10,15-22H2,(H,32,38)(H,33,37)(H,34,36)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283714
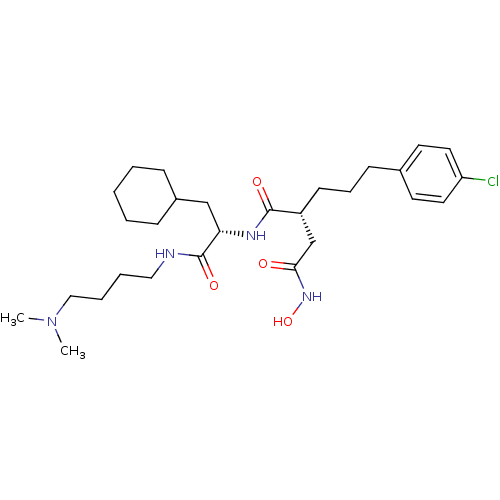
((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-[(S)-2-cyc...)Show SMILES CN(C)CCCCNC(=O)[C@H](CC1CCCCC1)NC(=O)[C@H](CCCc1ccc(Cl)cc1)CC(=O)NO Show InChI InChI=1S/C28H45ClN4O4/c1-33(2)18-7-6-17-30-28(36)25(19-22-9-4-3-5-10-22)31-27(35)23(20-26(34)32-37)12-8-11-21-13-15-24(29)16-14-21/h13-16,22-23,25,37H,3-12,17-20H2,1-2H3,(H,30,36)(H,31,35)(H,32,34)/t23-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50101530
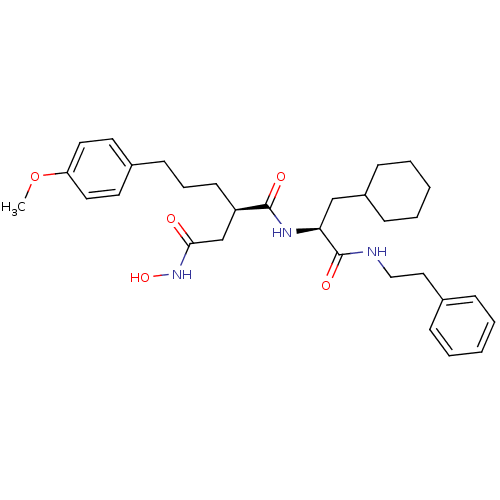
((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES COc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCc2ccccc2)cc1 Show InChI InChI=1S/C31H43N3O5/c1-39-27-17-15-24(16-18-27)13-8-14-26(22-29(35)34-38)30(36)33-28(21-25-11-6-3-7-12-25)31(37)32-20-19-23-9-4-2-5-10-23/h2,4-5,9-10,15-18,25-26,28,38H,3,6-8,11-14,19-22H2,1H3,(H,32,37)(H,33,36)(H,34,35)/t26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50101526
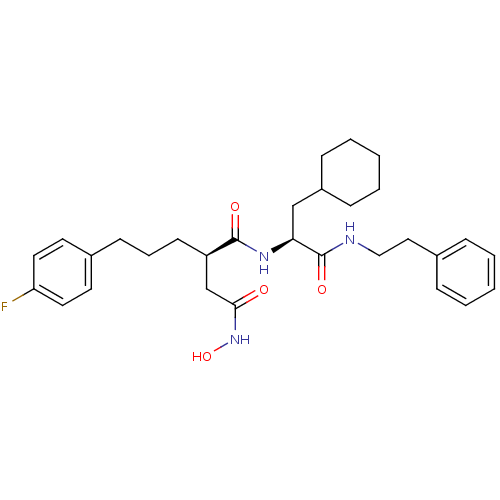
((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(F)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C30H40FN3O4/c31-26-16-14-23(15-17-26)12-7-13-25(21-28(35)34-38)29(36)33-27(20-24-10-5-2-6-11-24)30(37)32-19-18-22-8-3-1-4-9-22/h1,3-4,8-9,14-17,24-25,27,38H,2,5-7,10-13,18-21H2,(H,32,37)(H,33,36)(H,34,35)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50101492
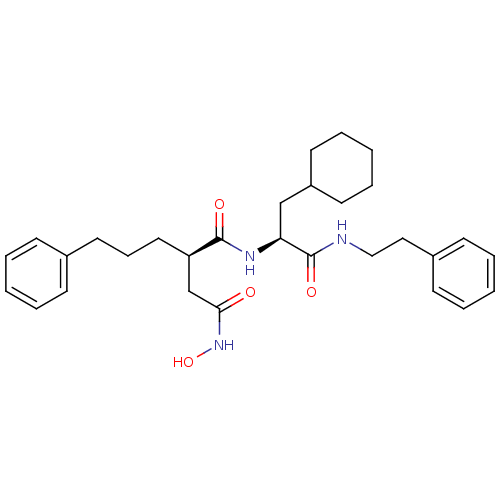
((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCCc1ccccc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C30H41N3O4/c34-28(33-37)22-26(18-10-17-23-11-4-1-5-12-23)29(35)32-27(21-25-15-8-3-9-16-25)30(36)31-20-19-24-13-6-2-7-14-24/h1-2,4-7,11-14,25-27,37H,3,8-10,15-22H2,(H,31,36)(H,32,35)(H,33,34)/t26-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50101513

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES Cc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCc2ccccc2)cc1 Show InChI InChI=1S/C31H43N3O4/c1-23-15-17-25(18-16-23)13-8-14-27(22-29(35)34-38)30(36)33-28(21-26-11-6-3-7-12-26)31(37)32-20-19-24-9-4-2-5-10-24/h2,4-5,9-10,15-18,26-28,38H,3,6-8,11-14,19-22H2,1H3,(H,32,37)(H,33,36)(H,34,35)/t27-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50101494
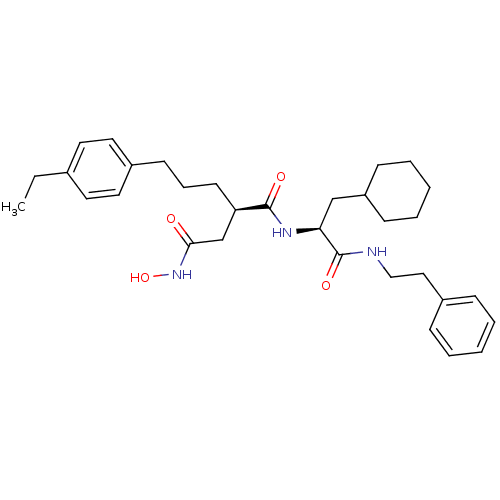
((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES CCc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCc2ccccc2)cc1 Show InChI InChI=1S/C32H45N3O4/c1-2-24-16-18-26(19-17-24)14-9-15-28(23-30(36)35-39)31(37)34-29(22-27-12-7-4-8-13-27)32(38)33-21-20-25-10-5-3-6-11-25/h3,5-6,10-11,16-19,27-29,39H,2,4,7-9,12-15,20-23H2,1H3,(H,33,38)(H,34,37)(H,35,36)/t28-,29+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50101528
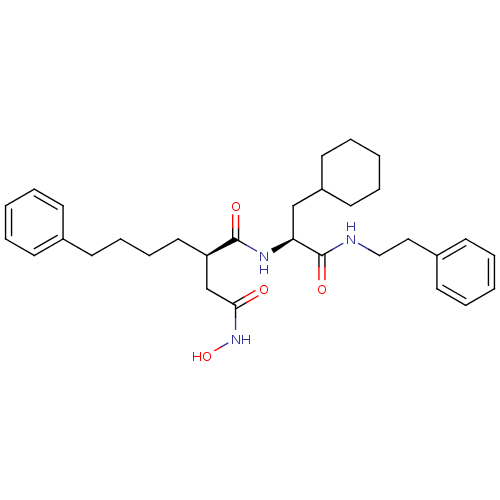
((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCCCc1ccccc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C31H43N3O4/c35-29(34-38)23-27(19-11-10-16-24-12-4-1-5-13-24)30(36)33-28(22-26-17-8-3-9-18-26)31(37)32-21-20-25-14-6-2-7-15-25/h1-2,4-7,12-15,26-28,38H,3,8-11,16-23H2,(H,32,37)(H,33,36)(H,34,35)/t27-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.254 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50101510

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(cc1)C(F)(F)F)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C31H40F3N3O4/c32-31(33,34)26-16-14-23(15-17-26)12-7-13-25(21-28(38)37-41)29(39)36-27(20-24-10-5-2-6-11-24)30(40)35-19-18-22-8-3-1-4-9-22/h1,3-4,8-9,14-17,24-25,27,41H,2,5-7,10-13,18-21H2,(H,35,40)(H,36,39)(H,37,38)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50101529

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES CC(C)C[C@H](CC(=O)NO)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C25H39N3O4/c1-18(2)15-21(17-23(29)28-32)24(30)27-22(16-20-11-7-4-8-12-20)25(31)26-14-13-19-9-5-3-6-10-19/h3,5-6,9-10,18,20-22,32H,4,7-8,11-17H2,1-2H3,(H,26,31)(H,27,30)(H,28,29)/t21-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283712

(4-{2-[(S)-3-Cyclohexyl-2-((R)-2-hydroxycarbamoylme...)Show SMILES ONC(=O)C[C@@H](CCCc1ccccc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccc(cc1)S(O)(=O)=O Show InChI InChI=1S/C30H41N3O7S/c34-28(33-37)21-25(13-7-12-22-8-3-1-4-9-22)29(35)32-27(20-24-10-5-2-6-11-24)30(36)31-19-18-23-14-16-26(17-15-23)41(38,39)40/h1,3-4,8-9,14-17,24-25,27,37H,2,5-7,10-13,18-21H2,(H,31,36)(H,32,35)(H,33,34)(H,38,39,40)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50283701

(3-{(S)-2-[(R)-5-(4-Chloro-phenyl)-2-hydroxycarbamo...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(Cl)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCC(O)=O Show InChI InChI=1S/C25H36ClN3O6/c26-20-11-9-17(10-12-20)7-4-8-19(16-22(30)29-35)24(33)28-21(15-18-5-2-1-3-6-18)25(34)27-14-13-23(31)32/h9-12,18-19,21,35H,1-8,13-16H2,(H,27,34)(H,28,33)(H,29,30)(H,31,32)/t19-,21+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50283708

(3-{(S)-2-[(R)-5-(4-Chloro-phenyl)-2-hydroxycarbamo...)Show SMILES COC(=O)CCNC(=O)[C@H](CC1CCCCC1)NC(=O)[C@H](CCCc1ccc(Cl)cc1)CC(=O)NO Show InChI InChI=1S/C26H38ClN3O6/c1-36-24(32)14-15-28-26(34)22(16-19-6-3-2-4-7-19)29-25(33)20(17-23(31)30-35)9-5-8-18-10-12-21(27)13-11-18/h10-13,19-20,22,35H,2-9,14-17H2,1H3,(H,28,34)(H,29,33)(H,30,31)/t20-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.960 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50283711

((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-[(S)-2-cyc...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(Cl)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCC(=O)N1CCOCC1 Show InChI InChI=1S/C29H43ClN4O6/c30-24-11-9-21(10-12-24)7-4-8-23(20-26(35)33-39)28(37)32-25(19-22-5-2-1-3-6-22)29(38)31-14-13-27(36)34-15-17-40-18-16-34/h9-12,22-23,25,39H,1-8,13-20H2,(H,31,38)(H,32,37)(H,33,35)/t23-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50283705

((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-{(S)-2-cyc...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(Cl)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCNS(=O)(=O)N1CCOCC1 Show InChI InChI=1S/C28H44ClN5O7S/c29-24-11-9-21(10-12-24)7-4-8-23(20-26(35)33-38)27(36)32-25(19-22-5-2-1-3-6-22)28(37)30-13-14-31-42(39,40)34-15-17-41-18-16-34/h9-12,22-23,25,31,38H,1-8,13-20H2,(H,30,37)(H,32,36)(H,33,35)/t23-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50101514

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCc1ccccc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C29H39N3O4/c33-27(32-36)21-25(17-16-22-10-4-1-5-11-22)28(34)31-26(20-24-14-8-3-9-15-24)29(35)30-19-18-23-12-6-2-7-13-23/h1-2,4-7,10-13,24-26,36H,3,8-9,14-21H2,(H,30,35)(H,31,34)(H,32,33)/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50283707

((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-[(S)-2-cyc...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(Cl)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCCCN1CCOCC1 Show InChI InChI=1S/C30H47ClN4O5/c31-26-13-11-23(12-14-26)9-6-10-25(22-28(36)34-39)29(37)33-27(21-24-7-2-1-3-8-24)30(38)32-15-4-5-16-35-17-19-40-20-18-35/h11-14,24-25,27,39H,1-10,15-22H2,(H,32,38)(H,33,37)(H,34,36)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50283710

((R)-N*1*-[(S)-2-Cyclohexyl-1-(4-morpholin-4-yl-but...)Show SMILES Cc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCCCN2CCOCC2)cc1 Show InChI InChI=1S/C31H50N4O5/c1-24-12-14-25(15-13-24)10-7-11-27(23-29(36)34-39)30(37)33-28(22-26-8-3-2-4-9-26)31(38)32-16-5-6-17-35-18-20-40-21-19-35/h12-15,26-28,39H,2-11,16-23H2,1H3,(H,32,38)(H,33,37)(H,34,36)/t27-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50283715

((R)-N*1*-{(S)-2-Cyclohexyl-1-[2-(morpholine-4-sulf...)Show SMILES Cc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCNS(=O)(=O)N2CCOCC2)cc1 Show InChI InChI=1S/C29H47N5O7S/c1-22-10-12-23(13-11-22)8-5-9-25(21-27(35)33-38)28(36)32-26(20-24-6-3-2-4-7-24)29(37)30-14-15-31-42(39,40)34-16-18-41-19-17-34/h10-13,24-26,31,38H,2-9,14-21H2,1H3,(H,30,37)(H,32,36)(H,33,35)/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 2.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50283703

((R)-N*1*-{(S)-2-Cyclohexyl-1-[2-(4-sulfamoyl-pheny...)Show SMILES Cc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCc2ccc(cc2)S(N)(=O)=O)cc1 Show InChI InChI=1S/C31H44N4O6S/c1-22-10-12-23(13-11-22)8-5-9-26(21-29(36)35-39)30(37)34-28(20-25-6-3-2-4-7-25)31(38)33-19-18-24-14-16-27(17-15-24)42(32,40)41/h10-17,25-26,28,39H,2-9,18-21H2,1H3,(H,33,38)(H,34,37)(H,35,36)(H2,32,40,41)/t26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50101530

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES COc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCc2ccccc2)cc1 Show InChI InChI=1S/C31H43N3O5/c1-39-27-17-15-24(16-18-27)13-8-14-26(22-29(35)34-38)30(36)33-28(21-25-11-6-3-7-12-25)31(37)32-20-19-23-9-4-2-5-10-23/h2,4-5,9-10,15-18,25-26,28,38H,3,6-8,11-14,19-22H2,1H3,(H,32,37)(H,33,36)(H,34,35)/t26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 4.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50101494

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES CCc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCc2ccccc2)cc1 Show InChI InChI=1S/C32H45N3O4/c1-2-24-16-18-26(19-17-24)14-9-15-28(23-30(36)35-39)31(37)34-29(22-27-12-7-4-8-13-27)32(38)33-21-20-25-10-5-3-6-11-25/h3,5-6,10-11,16-19,27-29,39H,2,4,7-9,12-15,20-23H2,1H3,(H,33,38)(H,34,37)(H,35,36)/t28-,29+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 5.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50283713

((R)-N*1*-[(S)-2-Cyclohexyl-1-(4-dimethylamino-buty...)Show SMILES CN(C)CCCCNC(=O)[C@H](CC1CCCCC1)NC(=O)[C@H](CCCc1ccc(C)cc1)CC(=O)NO Show InChI InChI=1S/C29H48N4O4/c1-22-14-16-23(17-15-22)12-9-13-25(21-27(34)32-37)28(35)31-26(20-24-10-5-4-6-11-24)29(36)30-18-7-8-19-33(2)3/h14-17,24-26,37H,4-13,18-21H2,1-3H3,(H,30,36)(H,31,35)(H,32,34)/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 5.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50283704

((R)-N*1*-[(S)-2-Cyclohexyl-1-(2-morpholin-4-yl-eth...)Show SMILES Cc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCN2CCOCC2)cc1 Show InChI InChI=1S/C29H46N4O5/c1-22-10-12-23(13-11-22)8-5-9-25(21-27(34)32-37)28(35)31-26(20-24-6-3-2-4-7-24)29(36)30-14-15-33-16-18-38-19-17-33/h10-13,24-26,37H,2-9,14-21H2,1H3,(H,30,36)(H,31,35)(H,32,34)/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 6.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50283702

((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-{(S)-2-cyc...)Show SMILES NS(=O)(=O)c1ccc(CCNC(=O)[C@H](CC2CCCCC2)NC(=O)[C@H](CCCc2ccc(Cl)cc2)CC(=O)NO)cc1 Show InChI InChI=1S/C30H41ClN4O6S/c31-25-13-9-21(10-14-25)7-4-8-24(20-28(36)35-39)29(37)34-27(19-23-5-2-1-3-6-23)30(38)33-18-17-22-11-15-26(16-12-22)42(32,40)41/h9-16,23-24,27,39H,1-8,17-20H2,(H,33,38)(H,34,37)(H,35,36)(H2,32,40,41)/t24-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 6.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283706

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCCC1CCCCC1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C30H47N3O4/c34-28(33-37)22-26(18-10-17-23-11-4-1-5-12-23)29(35)32-27(21-25-15-8-3-9-16-25)30(36)31-20-19-24-13-6-2-7-14-24/h2,6-7,13-14,23,25-27,37H,1,3-5,8-12,15-22H2,(H,31,36)(H,32,35)(H,33,34)/t26-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 6.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50101495

((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-((S)-2-cyc...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(Cl)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C30H40ClN3O4/c31-26-16-14-23(15-17-26)12-7-13-25(21-28(35)34-38)29(36)33-27(20-24-10-5-2-6-11-24)30(37)32-19-18-22-8-3-1-4-9-22/h1,3-4,8-9,14-17,24-25,27,38H,2,5-7,10-13,18-21H2,(H,32,37)(H,33,36)(H,34,35)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 7.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50101529

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES CC(C)C[C@H](CC(=O)NO)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C25H39N3O4/c1-18(2)15-21(17-23(29)28-32)24(30)27-22(16-20-11-7-4-8-12-20)25(31)26-14-13-19-9-5-3-6-10-19/h3,5-6,9-10,18,20-22,32H,4,7-8,11-17H2,1-2H3,(H,26,31)(H,27,30)(H,28,29)/t21-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 7.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50283712

(4-{2-[(S)-3-Cyclohexyl-2-((R)-2-hydroxycarbamoylme...)Show SMILES ONC(=O)C[C@@H](CCCc1ccccc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccc(cc1)S(O)(=O)=O Show InChI InChI=1S/C30H41N3O7S/c34-28(33-37)21-25(13-7-12-22-8-3-1-4-9-22)29(35)32-27(20-24-10-5-2-6-11-24)30(36)31-19-18-23-14-16-26(17-15-23)41(38,39)40/h1,3-4,8-9,14-17,24-25,27,37H,2,5-7,10-13,18-21H2,(H,31,36)(H,32,35)(H,33,34)(H,38,39,40)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 8.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50101513

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES Cc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCc2ccccc2)cc1 Show InChI InChI=1S/C31H43N3O4/c1-23-15-17-25(18-16-23)13-8-14-27(22-29(35)34-38)30(36)33-28(21-26-11-6-3-7-12-26)31(37)32-20-19-24-9-4-2-5-10-24/h2,4-5,9-10,15-18,26-28,38H,3,6-8,11-14,19-22H2,1H3,(H,32,37)(H,33,36)(H,34,35)/t27-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 8.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50101492

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCCc1ccccc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C30H41N3O4/c34-28(33-37)22-26(18-10-17-23-11-4-1-5-12-23)29(35)32-27(21-25-15-8-3-9-16-25)30(36)31-20-19-24-13-6-2-7-14-24/h1-2,4-7,11-14,25-27,37H,3,8-10,15-22H2,(H,31,36)(H,32,35)(H,33,34)/t26-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 8.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50101526

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(F)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C30H40FN3O4/c31-26-16-14-23(15-17-26)12-7-13-25(21-28(35)34-38)29(36)33-27(20-24-10-5-2-6-11-24)30(37)32-19-18-22-8-3-1-4-9-22/h1,3-4,8-9,14-17,24-25,27,38H,2,5-7,10-13,18-21H2,(H,32,37)(H,33,36)(H,34,35)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50283714

((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-[(S)-2-cyc...)Show SMILES CN(C)CCCCNC(=O)[C@H](CC1CCCCC1)NC(=O)[C@H](CCCc1ccc(Cl)cc1)CC(=O)NO Show InChI InChI=1S/C28H45ClN4O4/c1-33(2)18-7-6-17-30-28(36)25(19-22-9-4-3-5-10-22)31-27(35)23(20-26(34)32-37)12-8-11-21-13-15-24(29)16-14-21/h13-16,22-23,25,37H,3-12,17-20H2,1-2H3,(H,30,36)(H,31,35)(H,32,34)/t23-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50101529

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES CC(C)C[C@H](CC(=O)NO)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C25H39N3O4/c1-18(2)15-21(17-23(29)28-32)24(30)27-22(16-20-11-7-4-8-12-20)25(31)26-14-13-19-9-5-3-6-10-19/h3,5-6,9-10,18,20-22,32H,4,7-8,11-17H2,1-2H3,(H,26,31)(H,27,30)(H,28,29)/t21-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283700

((R)-N*1*-[(S)-1-((S)-1-Carbamoyl-2-phenyl-ethylcar...)Show SMILES CC(C)C[C@H](CC(=O)NO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C23H36N4O5/c1-14(2)10-17(13-20(28)27-32)22(30)26-19(11-15(3)4)23(31)25-18(21(24)29)12-16-8-6-5-7-9-16/h5-9,14-15,17-19,32H,10-13H2,1-4H3,(H2,24,29)(H,25,31)(H,26,30)(H,27,28)/t17-,18+,19+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
| 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50101514

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCc1ccccc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C29H39N3O4/c33-27(32-36)21-25(17-16-22-10-4-1-5-11-22)28(34)31-26(20-24-14-8-3-9-15-24)29(35)30-19-18-23-12-6-2-7-13-23/h1-2,4-7,10-13,24-26,36H,3,8-9,14-21H2,(H,30,35)(H,31,34)(H,32,33)/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 77 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50101510

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(cc1)C(F)(F)F)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C31H40F3N3O4/c32-31(33,34)26-16-14-23(15-17-26)12-7-13-25(21-28(38)37-41)29(39)36-27(20-24-10-5-2-6-11-24)30(40)35-19-18-22-8-3-1-4-9-22/h1,3-4,8-9,14-17,24-25,27,41H,2,5-7,10-13,18-21H2,(H,35,40)(H,36,39)(H,37,38)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50283700

((R)-N*1*-[(S)-1-((S)-1-Carbamoyl-2-phenyl-ethylcar...)Show SMILES CC(C)C[C@H](CC(=O)NO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C23H36N4O5/c1-14(2)10-17(13-20(28)27-32)22(30)26-19(11-15(3)4)23(31)25-18(21(24)29)12-16-8-6-5-7-9-16/h5-9,14-15,17-19,32H,10-13H2,1-4H3,(H2,24,29)(H,25,31)(H,26,30)(H,27,28)/t17-,18+,19+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
| 127 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50283708

(3-{(S)-2-[(R)-5-(4-Chloro-phenyl)-2-hydroxycarbamo...)Show SMILES COC(=O)CCNC(=O)[C@H](CC1CCCCC1)NC(=O)[C@H](CCCc1ccc(Cl)cc1)CC(=O)NO Show InChI InChI=1S/C26H38ClN3O6/c1-36-24(32)14-15-28-26(34)22(16-19-6-3-2-4-7-19)29-25(33)20(17-23(31)30-35)9-5-8-18-10-12-21(27)13-11-18/h10-13,19-20,22,35H,2-9,14-17H2,1H3,(H,28,34)(H,29,33)(H,30,31)/t20-,22+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 135 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50101528

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCCCc1ccccc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C31H43N3O4/c35-29(34-38)23-27(19-11-10-16-24-12-4-1-5-13-24)30(36)33-28(22-26-17-8-3-9-18-26)31(37)32-21-20-25-14-6-2-7-15-25/h1-2,4-7,12-15,26-28,38H,3,8-11,16-23H2,(H,32,37)(H,33,36)(H,34,35)/t27-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 177 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50283701

(3-{(S)-2-[(R)-5-(4-Chloro-phenyl)-2-hydroxycarbamo...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(Cl)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCC(O)=O Show InChI InChI=1S/C25H36ClN3O6/c26-20-11-9-17(10-12-20)7-4-8-19(16-22(30)29-35)24(33)28-21(15-18-5-2-1-3-6-18)25(34)27-14-13-23(31)32/h9-12,18-19,21,35H,1-8,13-16H2,(H,27,34)(H,28,33)(H,29,30)(H,31,32)/t19-,21+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 193 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50101492

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCCc1ccccc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C30H41N3O4/c34-28(33-37)22-26(18-10-17-23-11-4-1-5-12-23)29(35)32-27(21-25-15-8-3-9-16-25)30(36)31-20-19-24-13-6-2-7-14-24/h1-2,4-7,11-14,25-27,37H,3,8-10,15-22H2,(H,31,36)(H,32,35)(H,33,34)/t26-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 203 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50101526

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(F)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C30H40FN3O4/c31-26-16-14-23(15-17-26)12-7-13-25(21-28(35)34-38)29(36)33-27(20-24-10-5-2-6-11-24)30(37)32-19-18-22-8-3-1-4-9-22/h1,3-4,8-9,14-17,24-25,27,38H,2,5-7,10-13,18-21H2,(H,32,37)(H,33,36)(H,34,35)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 296 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50283705

((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-{(S)-2-cyc...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(Cl)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCNS(=O)(=O)N1CCOCC1 Show InChI InChI=1S/C28H44ClN5O7S/c29-24-11-9-21(10-12-24)7-4-8-23(20-26(35)33-38)27(36)32-25(19-22-5-2-1-3-6-22)28(37)30-13-14-31-42(39,40)34-15-17-41-18-16-34/h9-12,22-23,25,31,38H,1-8,13-20H2,(H,30,37)(H,32,36)(H,33,35)/t23-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 302 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50283700

((R)-N*1*-[(S)-1-((S)-1-Carbamoyl-2-phenyl-ethylcar...)Show SMILES CC(C)C[C@H](CC(=O)NO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C23H36N4O5/c1-14(2)10-17(13-20(28)27-32)22(30)26-19(11-15(3)4)23(31)25-18(21(24)29)12-16-8-6-5-7-9-16/h5-9,14-15,17-19,32H,10-13H2,1-4H3,(H2,24,29)(H,25,31)(H,26,30)(H,27,28)/t17-,18+,19+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
| 303 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50283707

((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-[(S)-2-cyc...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(Cl)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCCCN1CCOCC1 Show InChI InChI=1S/C30H47ClN4O5/c31-26-13-11-23(12-14-26)9-6-10-25(22-28(36)34-39)29(37)33-27(21-24-7-2-1-3-8-24)30(38)32-15-4-5-16-35-17-19-40-20-18-35/h11-14,24-25,27,39H,1-10,15-22H2,(H,32,38)(H,33,37)(H,34,36)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 313 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50283713

((R)-N*1*-[(S)-2-Cyclohexyl-1-(4-dimethylamino-buty...)Show SMILES CN(C)CCCCNC(=O)[C@H](CC1CCCCC1)NC(=O)[C@H](CCCc1ccc(C)cc1)CC(=O)NO Show InChI InChI=1S/C29H48N4O4/c1-22-14-16-23(17-15-22)12-9-13-25(21-27(34)32-37)28(35)31-26(20-24-10-5-4-6-11-24)29(36)30-18-7-8-19-33(2)3/h14-17,24-26,37H,4-13,18-21H2,1-3H3,(H,30,36)(H,31,35)(H,32,34)/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 313 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50283711

((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-[(S)-2-cyc...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(Cl)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCC(=O)N1CCOCC1 Show InChI InChI=1S/C29H43ClN4O6/c30-24-11-9-21(10-12-24)7-4-8-23(20-26(35)33-39)28(37)32-25(19-22-5-2-1-3-6-22)29(38)31-14-13-27(36)34-15-17-40-18-16-34/h9-12,22-23,25,39H,1-8,13-20H2,(H,31,38)(H,32,37)(H,33,35)/t23-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 319 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50283703

((R)-N*1*-{(S)-2-Cyclohexyl-1-[2-(4-sulfamoyl-pheny...)Show SMILES Cc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCc2ccc(cc2)S(N)(=O)=O)cc1 Show InChI InChI=1S/C31H44N4O6S/c1-22-10-12-23(13-11-22)8-5-9-26(21-29(36)35-39)30(37)34-28(20-25-6-3-2-4-7-25)31(38)33-19-18-24-14-16-27(17-15-24)42(32,40)41/h10-17,25-26,28,39H,2-9,18-21H2,1H3,(H,33,38)(H,34,37)(H,35,36)(H2,32,40,41)/t26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 329 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50283702

((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-{(S)-2-cyc...)Show SMILES NS(=O)(=O)c1ccc(CCNC(=O)[C@H](CC2CCCCC2)NC(=O)[C@H](CCCc2ccc(Cl)cc2)CC(=O)NO)cc1 Show InChI InChI=1S/C30H41ClN4O6S/c31-25-13-9-21(10-14-25)7-4-8-24(20-28(36)35-39)29(37)34-27(19-23-5-2-1-3-6-23)30(38)33-18-17-22-11-15-26(16-12-22)42(32,40)41/h9-16,23-24,27,39H,1-8,17-20H2,(H,33,38)(H,34,37)(H,35,36)(H2,32,40,41)/t24-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 382 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50283715

((R)-N*1*-{(S)-2-Cyclohexyl-1-[2-(morpholine-4-sulf...)Show SMILES Cc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCNS(=O)(=O)N2CCOCC2)cc1 Show InChI InChI=1S/C29H47N5O7S/c1-22-10-12-23(13-11-22)8-5-9-25(21-27(35)33-38)28(36)32-26(20-24-6-3-2-4-7-24)29(37)30-14-15-31-42(39,40)34-16-18-41-19-17-34/h10-13,24-26,31,38H,2-9,14-21H2,1H3,(H,30,37)(H,32,36)(H,33,35)/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 385 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50283704

((R)-N*1*-[(S)-2-Cyclohexyl-1-(2-morpholin-4-yl-eth...)Show SMILES Cc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCN2CCOCC2)cc1 Show InChI InChI=1S/C29H46N4O5/c1-22-10-12-23(13-11-22)8-5-9-25(21-27(34)32-37)28(35)31-26(20-24-6-3-2-4-7-24)29(36)30-14-15-33-16-18-38-19-17-33/h10-13,24-26,37H,2-9,14-21H2,1H3,(H,30,36)(H,31,35)(H,32,34)/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 455 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50101514

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCc1ccccc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C29H39N3O4/c33-27(32-36)21-25(17-16-22-10-4-1-5-11-22)28(34)31-26(20-24-14-8-3-9-15-24)29(35)30-19-18-23-12-6-2-7-13-23/h1-2,4-7,10-13,24-26,36H,3,8-9,14-21H2,(H,30,35)(H,31,34)(H,32,33)/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 618 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50283710

((R)-N*1*-[(S)-2-Cyclohexyl-1-(4-morpholin-4-yl-but...)Show SMILES Cc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCCCN2CCOCC2)cc1 Show InChI InChI=1S/C31H50N4O5/c1-24-12-14-25(15-13-24)10-7-11-27(23-29(36)34-39)30(37)33-28(22-26-8-3-2-4-9-26)31(38)32-16-5-6-17-35-18-20-40-21-19-35/h12-15,26-28,39H,2-11,16-23H2,1H3,(H,32,38)(H,33,37)(H,34,36)/t27-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 642 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50101513

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES Cc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCc2ccccc2)cc1 Show InChI InChI=1S/C31H43N3O4/c1-23-15-17-25(18-16-23)13-8-14-27(22-29(35)34-38)30(36)33-28(21-26-11-6-3-7-12-26)31(37)32-20-19-24-9-4-2-5-10-24/h2,4-5,9-10,15-18,26-28,38H,3,6-8,11-14,19-22H2,1H3,(H,32,37)(H,33,36)(H,34,35)/t27-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 921 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50283712

(4-{2-[(S)-3-Cyclohexyl-2-((R)-2-hydroxycarbamoylme...)Show SMILES ONC(=O)C[C@@H](CCCc1ccccc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccc(cc1)S(O)(=O)=O Show InChI InChI=1S/C30H41N3O7S/c34-28(33-37)21-25(13-7-12-22-8-3-1-4-9-22)29(35)32-27(20-24-10-5-2-6-11-24)30(36)31-19-18-23-14-16-26(17-15-23)41(38,39)40/h1,3-4,8-9,14-17,24-25,27,37H,2,5-7,10-13,18-21H2,(H,31,36)(H,32,35)(H,33,34)(H,38,39,40)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 942 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50283714

((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-[(S)-2-cyc...)Show SMILES CN(C)CCCCNC(=O)[C@H](CC1CCCCC1)NC(=O)[C@H](CCCc1ccc(Cl)cc1)CC(=O)NO Show InChI InChI=1S/C28H45ClN4O4/c1-33(2)18-7-6-17-30-28(36)25(19-22-9-4-3-5-10-22)31-27(35)23(20-26(34)32-37)12-8-11-21-13-15-24(29)16-14-21/h13-16,22-23,25,37H,3-12,17-20H2,1-2H3,(H,30,36)(H,31,35)(H,32,34)/t23-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 1.05E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50101530

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES COc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCc2ccccc2)cc1 Show InChI InChI=1S/C31H43N3O5/c1-39-27-17-15-24(16-18-27)13-8-14-26(22-29(35)34-38)30(36)33-28(21-25-11-6-3-7-12-25)31(37)32-20-19-23-9-4-2-5-10-23/h2,4-5,9-10,15-18,25-26,28,38H,3,6-8,11-14,19-22H2,1H3,(H,32,37)(H,33,36)(H,34,35)/t26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 1.53E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50283709
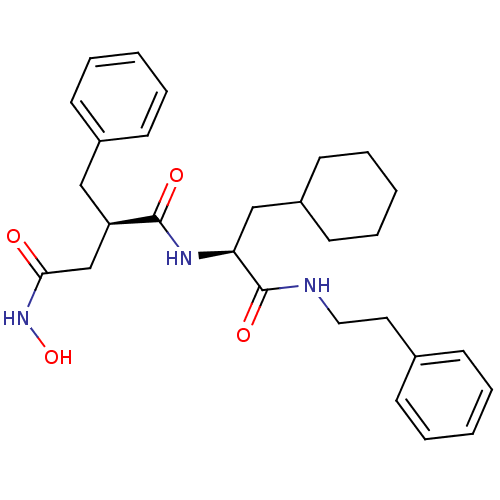
((R)-2-Benzyl-N*1*-((S)-2-cyclohexyl-1-phenethylcar...)Show SMILES ONC(=O)C[C@@H](Cc1ccccc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C28H37N3O4/c32-26(31-35)20-24(18-22-12-6-2-7-13-22)27(33)30-25(19-23-14-8-3-9-15-23)28(34)29-17-16-21-10-4-1-5-11-21/h1-2,4-7,10-13,23-25,35H,3,8-9,14-20H2,(H,29,34)(H,30,33)(H,31,32)/t24-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the gelatinase-A enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50101495

((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-((S)-2-cyc...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(Cl)cc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C30H40ClN3O4/c31-26-16-14-23(15-17-26)12-7-13-25(21-28(35)34-38)29(36)33-27(20-24-10-5-2-6-11-24)30(37)32-19-18-22-8-3-1-4-9-22/h1,3-4,8-9,14-17,24-25,27,38H,2,5-7,10-13,18-21H2,(H,32,37)(H,33,36)(H,34,35)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 2.44E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50101528

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCCCc1ccccc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C31H43N3O4/c35-29(34-38)23-27(19-11-10-16-24-12-4-1-5-13-24)30(36)33-28(22-26-17-8-3-9-18-26)31(37)32-21-20-25-14-6-2-7-15-25/h1-2,4-7,12-15,26-28,38H,3,8-11,16-23H2,(H,32,37)(H,33,36)(H,34,35)/t27-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50101510

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES ONC(=O)C[C@@H](CCCc1ccc(cc1)C(F)(F)F)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C31H40F3N3O4/c32-31(33,34)26-16-14-23(15-17-26)12-7-13-25(21-28(38)37-41)29(39)36-27(20-24-10-5-2-6-11-24)30(40)35-19-18-22-8-3-1-4-9-22/h1,3-4,8-9,14-17,24-25,27,41H,2,5-7,10-13,18-21H2,(H,35,40)(H,36,39)(H,37,38)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50101494

((R)-N*1*-((S)-2-Cyclohexyl-1-phenethylcarbamoyl-et...)Show SMILES CCc1ccc(CCC[C@H](CC(=O)NO)C(=O)N[C@@H](CC2CCCCC2)C(=O)NCCc2ccccc2)cc1 Show InChI InChI=1S/C32H45N3O4/c1-2-24-16-18-26(19-17-24)14-9-15-28(23-30(36)35-39)31(37)34-29(22-27-12-7-4-8-13-27)32(38)33-21-20-25-10-5-3-6-11-25/h3,5-6,10-11,16-19,27-29,39H,2,4,7-9,12-15,20-23H2,1H3,(H,33,38)(H,34,37)(H,35,36)/t28-,29+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 5.12E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50283709

((R)-2-Benzyl-N*1*-((S)-2-cyclohexyl-1-phenethylcar...)Show SMILES ONC(=O)C[C@@H](Cc1ccccc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C28H37N3O4/c32-26(31-35)20-24(18-22-12-6-2-7-13-22)27(33)30-25(19-23-14-8-3-9-15-23)28(34)29-17-16-21-10-4-1-5-11-21/h1-2,4-7,10-13,23-25,35H,3,8-9,14-20H2,(H,29,34)(H,30,33)(H,31,32)/t24-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the collagenase enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50283709

((R)-2-Benzyl-N*1*-((S)-2-cyclohexyl-1-phenethylcar...)Show SMILES ONC(=O)C[C@@H](Cc1ccccc1)C(=O)N[C@@H](CC1CCCCC1)C(=O)NCCc1ccccc1 Show InChI InChI=1S/C28H37N3O4/c32-26(31-35)20-24(18-22-12-6-2-7-13-22)27(33)30-25(19-23-14-8-3-9-15-23)28(34)29-17-16-21-10-4-1-5-11-21/h1-2,4-7,10-13,23-25,35H,3,8-9,14-20H2,(H,29,34)(H,30,33)(H,31,32)/t24-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of the stromelysin enzyme. |
Bioorg Med Chem Lett 4: 2741-2746 (1994)
Article DOI: 10.1016/S0960-894X(01)80587-4
BindingDB Entry DOI: 10.7270/Q2P84BT0 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data