

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50279966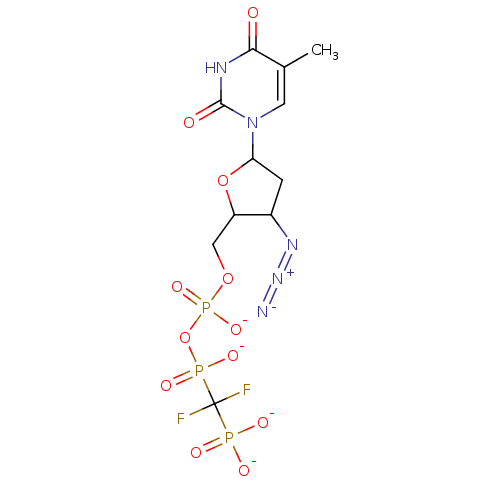 (Azidothymidine difluoromethylenephosphonate deriva...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article | 2.23E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory constant was determined in an HIV-1 reverse transcriptase assay in which the [3H]dTTP concentration was varied (i.e. 40, 20, 10, 6, and 4 ... | Bioorg Med Chem Lett 1: 357-360 (1991) Article DOI: 10.1016/S0960-894X(01)80472-8 BindingDB Entry DOI: 10.7270/Q2PV6KVZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50370476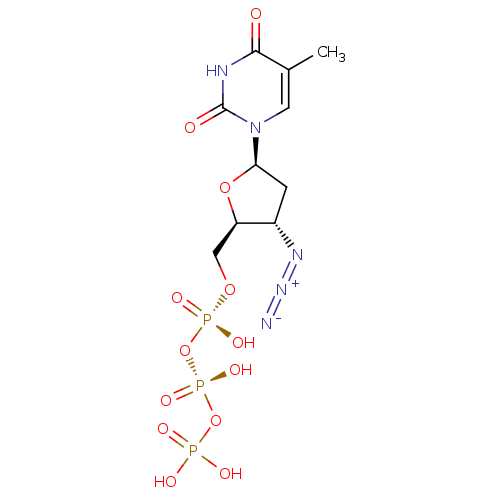 (Combivir | ZIDOVUDINE TRIPHOSPHATE) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid PDB UniChem Similars | PDB Article | n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound concentration that causes 50 % inhibition of HIV-1 peptide-derived reverse transcriptase (RT) obtained from HIV-1-infected H9 cell cultures | Bioorg Med Chem Lett 1: 357-360 (1991) Article DOI: 10.1016/S0960-894X(01)80472-8 BindingDB Entry DOI: 10.7270/Q2PV6KVZ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50279966 (Azidothymidine difluoromethylenephosphonate deriva...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article | n/a | n/a | 620 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound concentration that causes 50 % inhibition of HIV-1 peptide-derived reverse transcriptase (RT) | Bioorg Med Chem Lett 1: 357-360 (1991) Article DOI: 10.1016/S0960-894X(01)80472-8 BindingDB Entry DOI: 10.7270/Q2PV6KVZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50279965 (Azidothymidine difluoromethylenephosphonate deriva...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article | n/a | n/a | 6.95E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound concentration that causes 50 % inhibition of HIV-1 peptide-derived reverse transcriptase (RT) | Bioorg Med Chem Lett 1: 357-360 (1991) Article DOI: 10.1016/S0960-894X(01)80472-8 BindingDB Entry DOI: 10.7270/Q2PV6KVZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||