 Found 76 hits of Enzyme Inhibition Constant Data
Found 76 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50052957
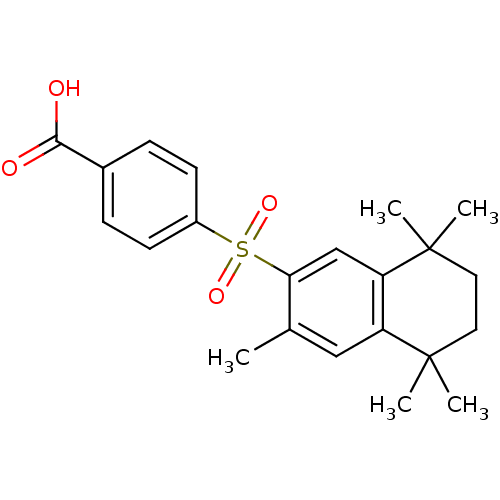
(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O4S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)27(25,26)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-beta
(Mus musculus) | BDBM50052962
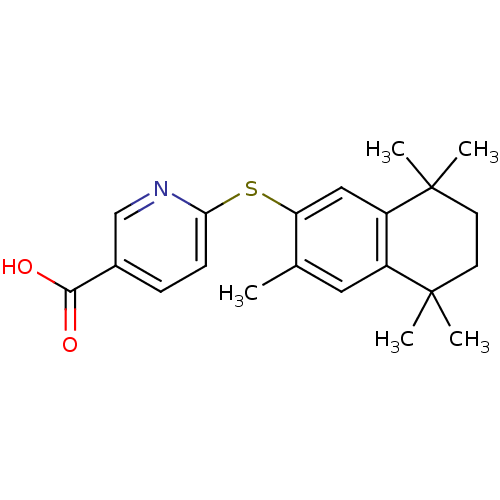
(6-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ccc(cn1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C21H25NO2S/c1-13-10-15-16(21(4,5)9-8-20(15,2)3)11-17(13)25-18-7-6-14(12-22-18)19(23)24/h6-7,10-12H,8-9H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | 87 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Mus musculus) | BDBM50052958
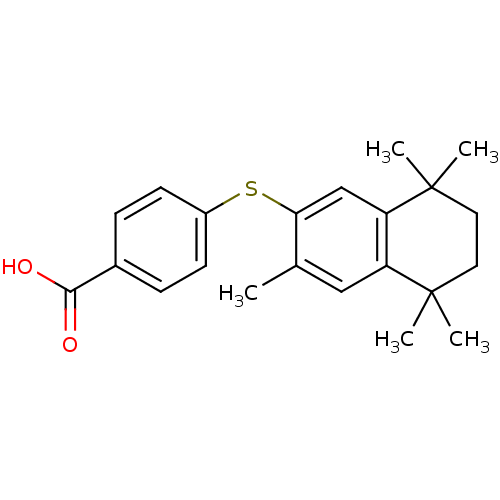
(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O2S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)25-16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-gamma
(Mus musculus) | BDBM31892

(9-cis retinoic acid | 9C-RA | CHEMBL705 | Panretin...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C\C=C\C(\C)=C\C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8-,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Retinoic acid receptor RXR-beta
(Mus musculus) | BDBM50052959
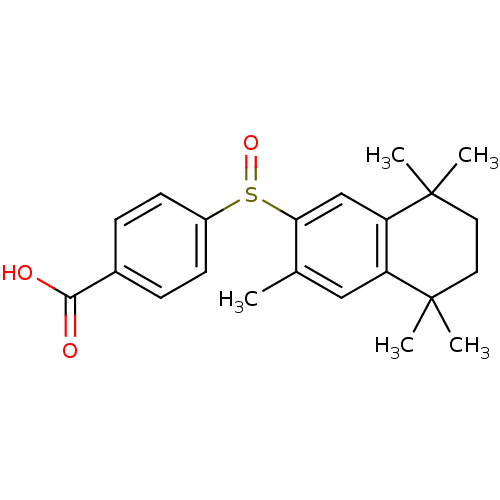
(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O3S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)26(25)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Mus musculus) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| MMDB
Article
PubMed
| n/a | n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-beta
(Homo sapiens (Human)) | BDBM50052961
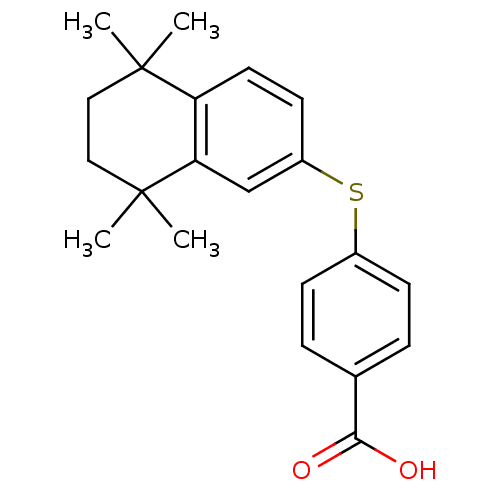
(4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-naphthal...)Show SMILES CC1(C)CCC(C)(C)c2cc(Sc3ccc(cc3)C(O)=O)ccc12 Show InChI InChI=1S/C21H24O2S/c1-20(2)11-12-21(3,4)18-13-16(9-10-17(18)20)24-15-7-5-14(6-8-15)19(22)23/h5-10,13H,11-12H2,1-4H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Mus musculus) | BDBM50052959

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O3S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)26(25)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-beta
(Mus musculus) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM31892

(9-cis retinoic acid | 9C-RA | CHEMBL705 | Panretin...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C\C=C\C(\C)=C\C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8-,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 50 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoic acid receptor RAR beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Retinoic acid receptor RXR-beta
(Mus musculus) | BDBM31892

(9-cis retinoic acid | 9C-RA | CHEMBL705 | Panretin...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C\C=C\C(\C)=C\C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8-,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Retinoic acid receptor RXR-gamma
(Mus musculus) | BDBM50052961

(4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-naphthal...)Show SMILES CC1(C)CCC(C)(C)c2cc(Sc3ccc(cc3)C(O)=O)ccc12 Show InChI InChI=1S/C21H24O2S/c1-20(2)11-12-21(3,4)18-13-16(9-10-17(18)20)24-15-7-5-14(6-8-15)19(22)23/h5-10,13H,11-12H2,1-4H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 2.85E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-gamma
(Homo sapiens (Human)) | BDBM50052958

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O2S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)25-16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 280 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| MMDB
Article
PubMed
| n/a | n/a | n/a | n/a | 80 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoic acid receptor RAR beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Mus musculus) | BDBM50052960
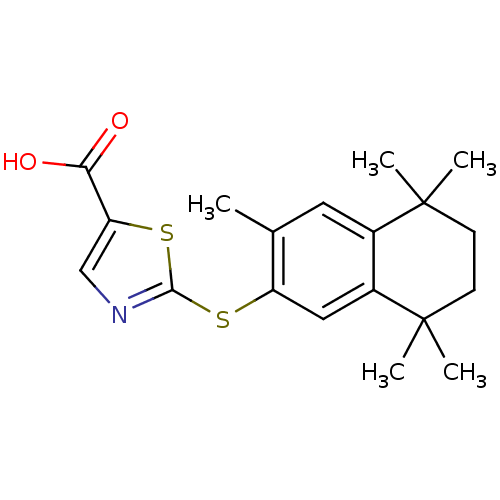
(2-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ncc(s1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C19H23NO2S2/c1-11-8-12-13(19(4,5)7-6-18(12,2)3)9-14(11)23-17-20-10-15(24-17)16(21)22/h8-10H,6-7H2,1-5H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Mus musculus) | BDBM31892

(9-cis retinoic acid | 9C-RA | CHEMBL705 | Panretin...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C\C=C\C(\C)=C\C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8-,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 350 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoic acid receptor RAR alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-gamma
(Mus musculus) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 350 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Mus musculus) | BDBM50052958

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O2S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)25-16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 296 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Mus musculus) | BDBM50052958

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O2S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)25-16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Mus musculus) | BDBM50052957

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O4S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)27(25,26)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Mus musculus) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| MMDB
Article
PubMed
| n/a | n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| MMDB
Article
PubMed
| n/a | n/a | n/a | n/a | 10 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoic acid receptor RAR gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Mus musculus) | BDBM50052958

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O2S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)25-16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50052961

(4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-naphthal...)Show SMILES CC1(C)CCC(C)(C)c2cc(Sc3ccc(cc3)C(O)=O)ccc12 Show InChI InChI=1S/C21H24O2S/c1-20(2)11-12-21(3,4)18-13-16(9-10-17(18)20)24-15-7-5-14(6-8-15)19(22)23/h5-10,13H,11-12H2,1-4H3,(H,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 770 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM31892

(9-cis retinoic acid | 9C-RA | CHEMBL705 | Panretin...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C\C=C\C(\C)=C\C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8-,16-14+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Retinoic acid receptor RXR-gamma
(Mus musculus) | BDBM50052959

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O3S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)26(25)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Mus musculus) | BDBM31892

(9-cis retinoic acid | 9C-RA | CHEMBL705 | Panretin...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C\C=C\C(\C)=C\C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8-,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Retinoic acid receptor RXR-gamma
(Homo sapiens (Human)) | BDBM50052960

(2-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ncc(s1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C19H23NO2S2/c1-11-8-12-13(19(4,5)7-6-18(12,2)3)9-14(11)23-17-20-10-15(24-17)16(21)22/h8-10H,6-7H2,1-5H3,(H,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.90E+3 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-beta
(Homo sapiens (Human)) | BDBM50052960

(2-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ncc(s1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C19H23NO2S2/c1-11-8-12-13(19(4,5)7-6-18(12,2)3)9-14(11)23-17-20-10-15(24-17)16(21)22/h8-10H,6-7H2,1-5H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-beta
(Mus musculus) | BDBM50052960

(2-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ncc(s1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C19H23NO2S2/c1-11-8-12-13(19(4,5)7-6-18(12,2)3)9-14(11)23-17-20-10-15(24-17)16(21)22/h8-10H,6-7H2,1-5H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-beta
(Mus musculus) | BDBM50052958

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O2S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)25-16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 302 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Mus musculus) | BDBM50052960

(2-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ncc(s1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C19H23NO2S2/c1-11-8-12-13(19(4,5)7-6-18(12,2)3)9-14(11)23-17-20-10-15(24-17)16(21)22/h8-10H,6-7H2,1-5H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-gamma
(Homo sapiens (Human)) | BDBM50052961

(4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-naphthal...)Show SMILES CC1(C)CCC(C)(C)c2cc(Sc3ccc(cc3)C(O)=O)ccc12 Show InChI InChI=1S/C21H24O2S/c1-20(2)11-12-21(3,4)18-13-16(9-10-17(18)20)24-15-7-5-14(6-8-15)19(22)23/h5-10,13H,11-12H2,1-4H3,(H,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Mus musculus) | BDBM50052961

(4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-naphthal...)Show SMILES CC1(C)CCC(C)(C)c2cc(Sc3ccc(cc3)C(O)=O)ccc12 Show InChI InChI=1S/C21H24O2S/c1-20(2)11-12-21(3,4)18-13-16(9-10-17(18)20)24-15-7-5-14(6-8-15)19(22)23/h5-10,13H,11-12H2,1-4H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 4.08E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM31892

(9-cis retinoic acid | 9C-RA | CHEMBL705 | Panretin...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C\C=C\C(\C)=C\C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8-,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 45 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoic acid receptor RAR gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Retinoic acid receptor RXR-beta
(Homo sapiens (Human)) | BDBM50052957

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O4S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)27(25,26)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-gamma
(Homo sapiens (Human)) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-beta
(Homo sapiens (Human)) | BDBM50052962

(6-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ccc(cn1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C21H25NO2S/c1-13-10-15-16(21(4,5)9-8-20(15,2)3)11-17(13)25-18-7-6-14(12-22-18)19(23)24/h6-7,10-12H,8-9H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | n/a | 57 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-gamma
(Mus musculus) | BDBM50052958

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O2S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)25-16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 304 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50052962

(6-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ccc(cn1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C21H25NO2S/c1-13-10-15-16(21(4,5)9-8-20(15,2)3)11-17(13)25-18-7-6-14(12-22-18)19(23)24/h6-7,10-12H,8-9H2,1-5H3,(H,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | n/a | 54 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-gamma
(Mus musculus) | BDBM50052962

(6-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ccc(cn1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C21H25NO2S/c1-13-10-15-16(21(4,5)9-8-20(15,2)3)11-17(13)25-18-7-6-14(12-22-18)19(23)24/h6-7,10-12H,8-9H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | 73 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-beta
(Mus musculus) | BDBM50052961

(4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-naphthal...)Show SMILES CC1(C)CCC(C)(C)c2cc(Sc3ccc(cc3)C(O)=O)ccc12 Show InChI InChI=1S/C21H24O2S/c1-20(2)11-12-21(3,4)18-13-16(9-10-17(18)20)24-15-7-5-14(6-8-15)19(22)23/h5-10,13H,11-12H2,1-4H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 3.09E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-gamma
(Homo sapiens (Human)) | BDBM31892

(9-cis retinoic acid | 9C-RA | CHEMBL705 | Panretin...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C\C=C\C(\C)=C\C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8-,16-14+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 140 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Retinoic acid receptor RXR-gamma
(Mus musculus) | BDBM50052960

(2-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ncc(s1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C19H23NO2S2/c1-11-8-12-13(19(4,5)7-6-18(12,2)3)9-14(11)23-17-20-10-15(24-17)16(21)22/h8-10H,6-7H2,1-5H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Mus musculus) | BDBM50052957

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O4S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)27(25,26)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Mus musculus) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-beta
(Homo sapiens (Human)) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Mus musculus) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-gamma
(Mus musculus) | BDBM50052957

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O4S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)27(25,26)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 3.99E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Homo sapiens (Human)) | BDBM50052961

(4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-naphthal...)Show SMILES CC1(C)CCC(C)(C)c2cc(Sc3ccc(cc3)C(O)=O)ccc12 Show InChI InChI=1S/C21H24O2S/c1-20(2)11-12-21(3,4)18-13-16(9-10-17(18)20)24-15-7-5-14(6-8-15)19(22)23/h5-10,13H,11-12H2,1-4H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 570 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoic acid receptor RAR beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-gamma
(Homo sapiens (Human)) | BDBM50052957

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O4S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)27(25,26)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Mus musculus) | BDBM50052959

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O3S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)26(25)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Mus musculus) | BDBM31892

(9-cis retinoic acid | 9C-RA | CHEMBL705 | Panretin...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C\C=C\C(\C)=C\C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8-,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Retinoic acid receptor RXR-alpha
(Mus musculus) | BDBM50052961

(4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-naphthal...)Show SMILES CC1(C)CCC(C)(C)c2cc(Sc3ccc(cc3)C(O)=O)ccc12 Show InChI InChI=1S/C21H24O2S/c1-20(2)11-12-21(3,4)18-13-16(9-10-17(18)20)24-15-7-5-14(6-8-15)19(22)23/h5-10,13H,11-12H2,1-4H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 3.87E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Mus musculus) | BDBM50052961

(4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-naphthal...)Show SMILES CC1(C)CCC(C)(C)c2cc(Sc3ccc(cc3)C(O)=O)ccc12 Show InChI InChI=1S/C21H24O2S/c1-20(2)11-12-21(3,4)18-13-16(9-10-17(18)20)24-15-7-5-14(6-8-15)19(22)23/h5-10,13H,11-12H2,1-4H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 4.24E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50052960

(2-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ncc(s1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C19H23NO2S2/c1-11-8-12-13(19(4,5)7-6-18(12,2)3)9-14(11)23-17-20-10-15(24-17)16(21)22/h8-10H,6-7H2,1-5H3,(H,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.30E+3 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Mus musculus) | BDBM50052959

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O3S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)26(25)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor alpha
(Homo sapiens (Human)) | BDBM31892

(9-cis retinoic acid | 9C-RA | CHEMBL705 | Panretin...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C\C=C\C(\C)=C\C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8-,16-14+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 191 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoic acid receptor RAR alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Retinoic acid receptor RXR-alpha
(Mus musculus) | BDBM50052957

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O4S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)27(25,26)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 2.52E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Mus musculus) | BDBM50052962

(6-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ccc(cn1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C21H25NO2S/c1-13-10-15-16(21(4,5)9-8-20(15,2)3)11-17(13)25-18-7-6-14(12-22-18)19(23)24/h6-7,10-12H,8-9H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-beta
(Homo sapiens (Human)) | BDBM50052959

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O3S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)26(25)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Mus musculus) | BDBM50052960

(2-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ncc(s1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C19H23NO2S2/c1-11-8-12-13(19(4,5)7-6-18(12,2)3)9-14(11)23-17-20-10-15(24-17)16(21)22/h8-10H,6-7H2,1-5H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50052961

(4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-naphthal...)Show SMILES CC1(C)CCC(C)(C)c2cc(Sc3ccc(cc3)C(O)=O)ccc12 Show InChI InChI=1S/C21H24O2S/c1-20(2)11-12-21(3,4)18-13-16(9-10-17(18)20)24-15-7-5-14(6-8-15)19(22)23/h5-10,13H,11-12H2,1-4H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 340 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoic acid receptor RAR gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Mus musculus) | BDBM50052961

(4-(5,5,8,8-Tetramethyl-5,6,7,8-tetrahydro-naphthal...)Show SMILES CC1(C)CCC(C)(C)c2cc(Sc3ccc(cc3)C(O)=O)ccc12 Show InChI InChI=1S/C21H24O2S/c1-20(2)11-12-21(3,4)18-13-16(9-10-17(18)20)24-15-7-5-14(6-8-15)19(22)23/h5-10,13H,11-12H2,1-4H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Mus musculus) | BDBM50052960

(2-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ncc(s1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C19H23NO2S2/c1-11-8-12-13(19(4,5)7-6-18(12,2)3)9-14(11)23-17-20-10-15(24-17)16(21)22/h8-10H,6-7H2,1-5H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-beta
(Homo sapiens (Human)) | BDBM50052958

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O2S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)25-16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 300 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Mus musculus) | BDBM31892

(9-cis retinoic acid | 9C-RA | CHEMBL705 | Panretin...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C\C=C\C(\C)=C\C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8-,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 900 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR alpha |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Mus musculus) | BDBM50052959

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O3S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)26(25)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50052958

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O2S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)25-16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 280 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR alpha;NA=not active |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-gamma
(Homo sapiens (Human)) | BDBM50052959

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O3S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)26(25)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-beta
(Mus musculus) | BDBM50052957

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O4S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)27(25,26)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 4.12E+3 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoid X receptor beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor beta
(Mus musculus) | BDBM50052957

(4-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1S(=O)(=O)c1ccc(cc1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C22H26O4S/c1-14-12-17-18(22(4,5)11-10-21(17,2)3)13-19(14)27(25,26)16-8-6-15(7-9-16)20(23)24/h6-9,12-13H,10-11H2,1-5H3,(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Dissociation constant for binding to Retinoic acid receptor beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-gamma
(Homo sapiens (Human)) | BDBM50052962

(6-(3,5,5,8,8-Pentamethyl-5,6,7,8-tetrahydro-naphth...)Show SMILES Cc1cc2c(cc1Sc1ccc(cn1)C(O)=O)C(C)(C)CCC2(C)C Show InChI InChI=1S/C21H25NO2S/c1-13-10-15-16(21(4,5)9-8-20(15,2)3)11-17(13)25-18-7-6-14(12-22-18)19(23)24/h6-7,10-12H,8-9H2,1-5H3,(H,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | n/a | n/a | 42 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR gamma |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-beta
(Homo sapiens (Human)) | BDBM31892

(9-cis retinoic acid | 9C-RA | CHEMBL705 | Panretin...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C\C=C\C(\C)=C\C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8-,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 200 | n/a | n/a | n/a | n/a |
Allergan, Inc
Curated by ChEMBL
| Assay Description
Transcriptional activation of Retinoid X receptor RXR beta |
J Med Chem 39: 3556-63 (1996)
Article DOI: 10.1021/jm960386h
BindingDB Entry DOI: 10.7270/Q2959GNV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data