

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM10245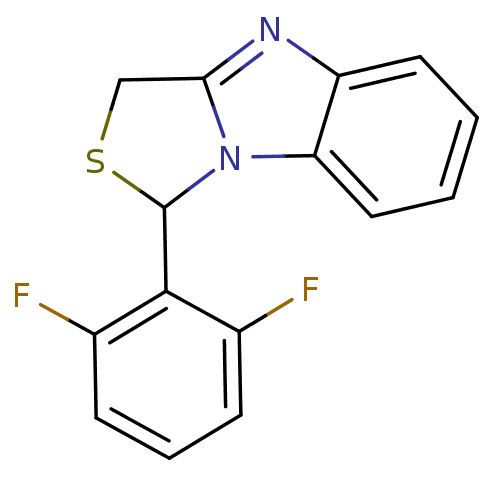 (3-(2,6-difluorophenyl)-4-thia-2,7-diazatricyclo[6....) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description The quantity of compound required to reduce WT Reverse transcriptase activity by 50% | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50061574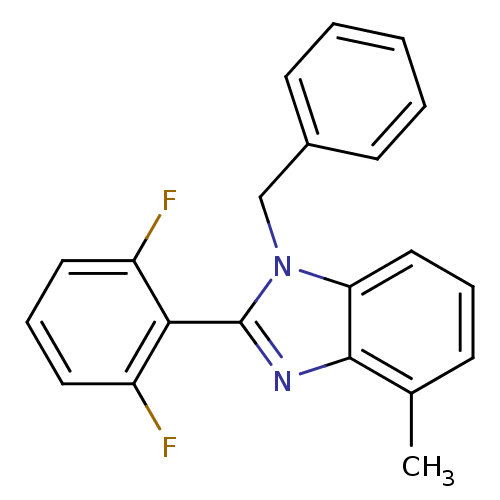 (1-Benzyl-2-(2,6-difluoro-phenyl)-4-methyl-1H-benzo...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description The quantity of compound required to reduce WT Reverse transcriptase activity by 50% | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50061572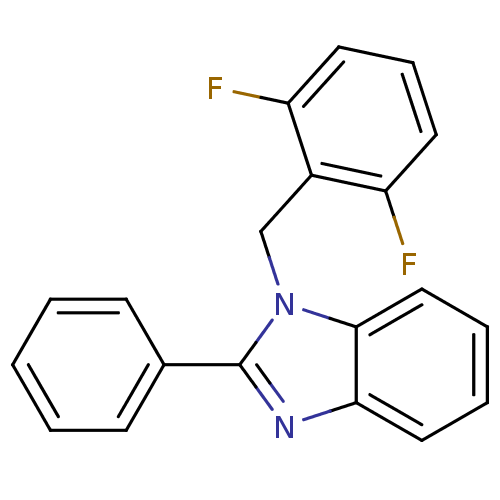 (1-(2,6-Difluoro-benzyl)-2-phenyl-1H-benzoimidazole...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description The quantity of compound required to reduce WT Reverse transcriptase activity by 50% | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50061575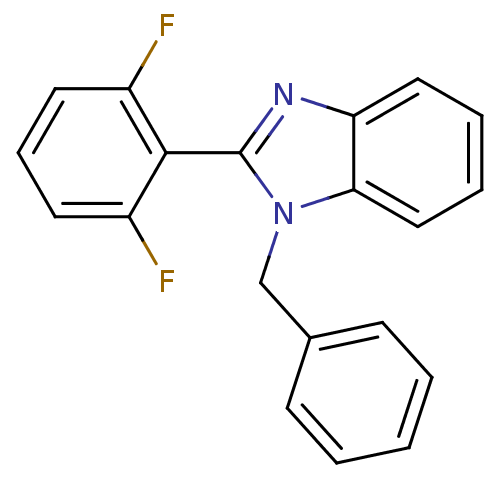 (1-Benzyl-2-(2,6-difluoro-phenyl)-1H-benzoimidazole...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.86E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description The quantity of compound required to reduce WT Reverse transcriptase activity by 50% | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17666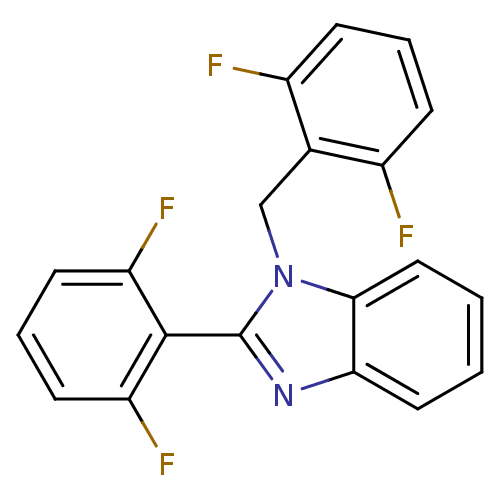 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.10E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description The quantity of compound required to reduce WT Reverse transcriptase activity by 50% | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17665 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | 2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description The quantity of compound required to reduce WT Reverse transcriptase activity by 50% | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17666 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 840 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with 4xAZT/L100I (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cyt... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17666 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 270 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with 4xAZT (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50036504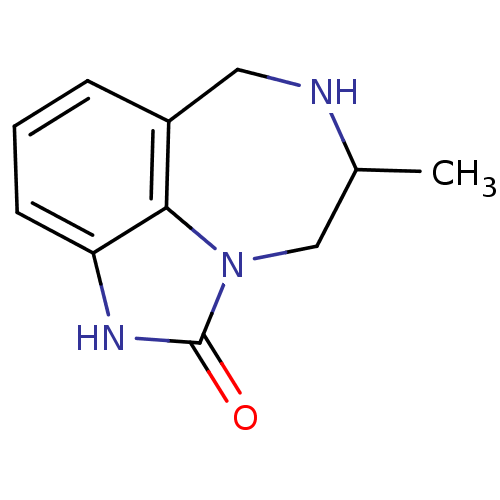 ((R-(-))-8-Methyl-6,7,8,9-tetrahydro-2H-2,7,9a-tria...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 6.20E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with V179D (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17666 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with 4xAZT/Y181C (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cyt... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50036504 ((R-(-))-8-Methyl-6,7,8,9-tetrahydro-2H-2,7,9a-tria...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 200 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with L74V (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathic... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17665 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | n/a | n/a | 2.30E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with Y188C (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17666 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with V106A (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM10245 (3-(2,6-difluorophenyl)-4-thia-2,7-diazatricyclo[6....) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | n/a | n/a | 700 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with L74V (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathic... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50036504 ((R-(-))-8-Methyl-6,7,8,9-tetrahydro-2H-2,7,9a-tria...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 300 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with NL4-3(WT) NNRTI (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50036504 ((R-(-))-8-Methyl-6,7,8,9-tetrahydro-2H-2,7,9a-tria...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 300 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with 4xAZT (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50036504 ((R-(-))-8-Methyl-6,7,8,9-tetrahydro-2H-2,7,9a-tria...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with A98G (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathic... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50036504 ((R-(-))-8-Methyl-6,7,8,9-tetrahydro-2H-2,7,9a-tria...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.74E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with K101E (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17666 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 4.75E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with A98G (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathic... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50036504 ((R-(-))-8-Methyl-6,7,8,9-tetrahydro-2H-2,7,9a-tria...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with V108I (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM10245 (3-(2,6-difluorophenyl)-4-thia-2,7-diazatricyclo[6....) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with 4xAZT/L100I (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cyt... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50036504 ((R-(-))-8-Methyl-6,7,8,9-tetrahydro-2H-2,7,9a-tria...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.25E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with V106A (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM10245 (3-(2,6-difluorophenyl)-4-thia-2,7-diazatricyclo[6....) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | n/a | n/a | 9.70E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with V108I (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17665 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | n/a | n/a | 3.50E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with 4xAZT/Y181C (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cyt... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17666 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 460 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with L74V (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathic... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17665 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with L74V (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathic... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17665 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | n/a | n/a | 1.67E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance(antiviral activity) of the compound with K101E (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathic... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM10245 (3-(2,6-difluorophenyl)-4-thia-2,7-diazatricyclo[6....) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | n/a | n/a | 1.77E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance(antiviral activity) of the compound with A98G (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathic ... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM10245 (3-(2,6-difluorophenyl)-4-thia-2,7-diazatricyclo[6....) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | n/a | n/a | 1.63E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with Y181C (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17666 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.85E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with NL4-3(WT) NNRTI (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50036504 ((R-(-))-8-Methyl-6,7,8,9-tetrahydro-2H-2,7,9a-tria...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | >1.74E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with 4xAZT/L100I (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cyt... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17666 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.04E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with V108I (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM10245 (3-(2,6-difluorophenyl)-4-thia-2,7-diazatricyclo[6....) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with 4xAZT (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17666 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with K101E (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50036504 ((R-(-))-8-Methyl-6,7,8,9-tetrahydro-2H-2,7,9a-tria...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | >1.74E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with Y188C (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17665 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | n/a | n/a | 200 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with 4xAZT/L100I (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cyt... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM10245 (3-(2,6-difluorophenyl)-4-thia-2,7-diazatricyclo[6....) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | n/a | n/a | 1.22E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with L100I (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17665 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with V106A (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17665 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with V108I (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate v | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17666 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 2.30E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with V179D (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM10245 (3-(2,6-difluorophenyl)-4-thia-2,7-diazatricyclo[6....) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with K103N (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM10245 (3-(2,6-difluorophenyl)-4-thia-2,7-diazatricyclo[6....) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | n/a | n/a | 1.45E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with 4xAZT/Y181C (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cyt... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17665 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | n/a | n/a | 6.00E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with Y181C (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17666 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.29E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with K103N (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17665 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with A98G (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathic... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM10245 (3-(2,6-difluorophenyl)-4-thia-2,7-diazatricyclo[6....) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with K101E (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17665 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | n/a | n/a | 8.10E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with K103N (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50036504 ((R-(-))-8-Methyl-6,7,8,9-tetrahydro-2H-2,7,9a-tria...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.74E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with L100I (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17666 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.17E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with Y188C (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17665 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | n/a | n/a | 500 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with V179D (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50036504 ((R-(-))-8-Methyl-6,7,8,9-tetrahydro-2H-2,7,9a-tria...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.74E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with K103N (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM10245 (3-(2,6-difluorophenyl)-4-thia-2,7-diazatricyclo[6....) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | n/a | n/a | 3.10E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with V179D (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17665 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | n/a | n/a | 500 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with NL4-3(WT) NNRTI (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17666 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.52E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with Y181C (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50036504 ((R-(-))-8-Methyl-6,7,8,9-tetrahydro-2H-2,7,9a-tria...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 4.20E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with Y181C (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM10245 (3-(2,6-difluorophenyl)-4-thia-2,7-diazatricyclo[6....) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with V106A (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17665 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with 4xAZT (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM10245 (3-(2,6-difluorophenyl)-4-thia-2,7-diazatricyclo[6....) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with NL4-3(WT) NNRTI (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50036504 ((R-(-))-8-Methyl-6,7,8,9-tetrahydro-2H-2,7,9a-tria...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity)of the compound with 4xAZT/Y181C (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cyto... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM17665 (2-(2,6-difluorophenyl)-1-[(2,6-difluorophenyl)meth...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | n/a | n/a | 300 | n/a | n/a | n/a | n/a |
National Cancer Institute-Frederick Cancer Research and Development Center Curated by ChEMBL | Assay Description Cross Resistance (antiviral activity) of the compound with L100I (Nonnucleoside reverse transcriptase inhibitor) resistant HIV isolate from cytopathi... | J Med Chem 40: 4199-207 (1998) Article DOI: 10.1021/jm970096g BindingDB Entry DOI: 10.7270/Q25B01KN | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||