

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070471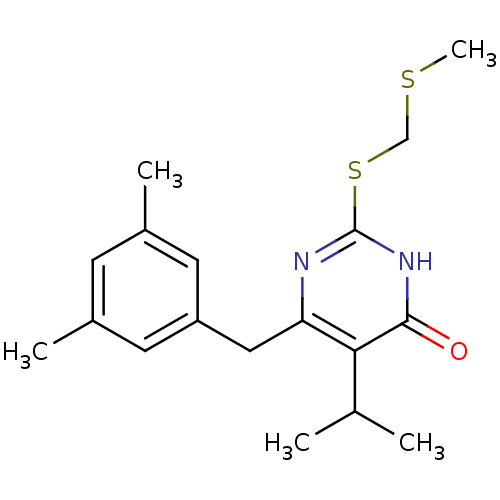 (6-(3,5-Dimethyl-benzyl)-5-isopropyl-2-methylsulfan...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 4.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute Curated by ChEMBL | Assay Description Compound was evaluated for enzymatic inhibitory activity against recombinant HIV reverse transcriptase | Bioorg Med Chem Lett 8: 1461-6 (1999) BindingDB Entry DOI: 10.7270/Q24X56XN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070469 (6-Benzyl-5-isopropyl-2-methylsulfanylmethylsulfany...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 6.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute Curated by ChEMBL | Assay Description Compound was evaluated for enzymatic inhibitory activity against recombinant HIV reverse transcriptase | Bioorg Med Chem Lett 8: 1461-6 (1999) BindingDB Entry DOI: 10.7270/Q24X56XN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070468 (6-Benzyl-5-ethyl-2-methylsulfanylmethylsulfanyl-3H...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 9.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute Curated by ChEMBL | Assay Description Compound was evaluated for enzymatic inhibitory activity against recombinant HIV reverse transcriptase | Bioorg Med Chem Lett 8: 1461-6 (1999) BindingDB Entry DOI: 10.7270/Q24X56XN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50070470 (6-Benzyl-5-methyl-2-methylsulfanylmethylsulfanyl-3...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.88E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute Curated by ChEMBL | Assay Description Compound was evaluated for enzymatic inhibitory activity against recombinant HIV reverse transcriptase | Bioorg Med Chem Lett 8: 1461-6 (1999) BindingDB Entry DOI: 10.7270/Q24X56XN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase/RNaseH (Human immunodeficiency virus 1) | BDBM50002692 ((AZT) 1-(4-Azido-5-hydroxymethyl-tetrahydro-furan-...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | DrugBank PubMed | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Institute Curated by ChEMBL | Assay Description Compound was evaluated for enzymatic inhibitory activity against recombinant HIV reverse transcriptase | Bioorg Med Chem Lett 8: 1461-6 (1999) BindingDB Entry DOI: 10.7270/Q24X56XN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||