

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Acetyl-CoA carboxylase 2 | ||
| Ligand | BDBM307424 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | Inhibitory Activity on Human ACC1 and the ACC2 | ||
| IC50 | 2.00±n/a nM | ||
| Citation |  Kobayashi, N; Asahi, K; Tomida, Y; Ohdan, M; Fumoto, M; Sasaki, Y; Kurahashi, K; Inoue, T; Urabe, T; Nishiura, Y; Iwatsu, M; Miyazaki, K; Ohyabu, N; Wada, T; Katou, M Alkylene derivatives US Patent US10150728 Publication Date 12/11/2018 Kobayashi, N; Asahi, K; Tomida, Y; Ohdan, M; Fumoto, M; Sasaki, Y; Kurahashi, K; Inoue, T; Urabe, T; Nishiura, Y; Iwatsu, M; Miyazaki, K; Ohyabu, N; Wada, T; Katou, M Alkylene derivatives US Patent US10150728 Publication Date 12/11/2018 | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Acetyl-CoA carboxylase 2 | |||
| Name: | Acetyl-CoA carboxylase 2 | ||
| Synonyms: | ACACB | ACACB_HUMAN | ACC-beta | ACC2 | ACCB | Acetyl-CoA carboxylase | Acetyl-CoA carboxylase 2 | Acetyl-CoA carboxylase 2 (ACC) | Acetyl-CoA carboxylase 2 (ACC2) | ||
| Type: | Protein | ||
| Mol. Mass.: | 276535.21 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | O00763 | ||
| Residue: | 2458 | ||
| Sequence: |
| ||
| BDBM307424 | |||
| n/a | |||
| Name | BDBM307424 | ||
| Synonyms: | US10150728, Example I-655 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C22H29ClFN3O4 | ||
| Mol. Mass. | 453.935 | ||
| SMILES | C[C@@H](CO[C@H]1CC[C@@H](CC1)Oc1nc2c(F)cc(OC3CC3)c(Cl)c2n1C)NC(C)=O |r,wU:7.10,1.0,wD:4.3,(6.86,-2.54,;6.86,-1,;5.53,-.23,;4.2,-1,;2.86,-.23,;2.86,1.31,;1.53,2.08,;.19,1.31,;.19,-.23,;1.53,-1,;-1.14,2.08,;-2.47,1.31,;-2.63,-.23,;-4.14,-.55,;-4.91,-1.88,;-4.14,-3.21,;-6.45,-1.88,;-7.22,-.55,;-8.76,-.55,;-9.53,-1.88,;-10.86,-2.65,;-9.53,-3.42,;-6.45,.79,;-7.22,2.12,;-4.91,.79,;-3.88,1.93,;-4.28,3.42,;8.2,-.23,;9.53,-1,;10.86,-.23,;9.53,-2.54,)| | ||
| Structure | 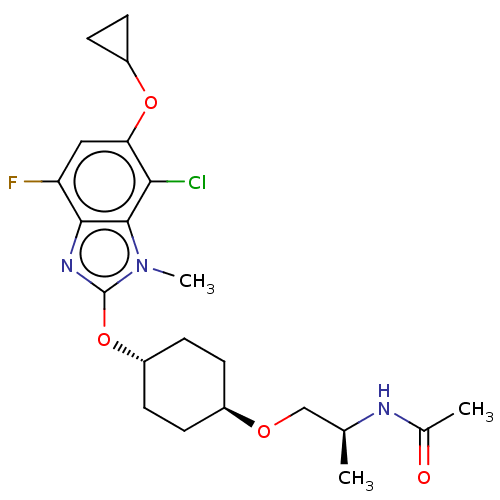
| ||