| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 11B1, mitochondrial |
|---|
| Ligand | BDBM8871 |
|---|
| Substrate/Competitor | BDBM8582 |
|---|
| Meas. Tech. | CYP11B Assay |
|---|
| pH | 7.4±n/a |
|---|
| Temperature | 310.15±n/a K |
|---|
| IC50 | 3.3±n/a nM |
|---|
| Citation |  Ulmschneider, S; Muller-Vieira, U; Mitrenga, M; Hartmann, RW; Oberwinkler-Marchais, S; Klein, CD; Bureik, M; Bernhardt, R; Antes, I; Lengauer, T Synthesis and evaluation of imidazolylmethylenetetrahydronaphthalenes and imidazolylmethyleneindanes: potent inhibitors of aldosterone synthase. J Med Chem48:1796-805 (2005) [PubMed] Article Ulmschneider, S; Muller-Vieira, U; Mitrenga, M; Hartmann, RW; Oberwinkler-Marchais, S; Klein, CD; Bureik, M; Bernhardt, R; Antes, I; Lengauer, T Synthesis and evaluation of imidazolylmethylenetetrahydronaphthalenes and imidazolylmethyleneindanes: potent inhibitors of aldosterone synthase. J Med Chem48:1796-805 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Inhibition_Run data, Solution Info, Assay Method |
|---|
| |
| Cytochrome P450 11B1, mitochondrial |
|---|
| Name: | Cytochrome P450 11B1, mitochondrial |
|---|
| Synonyms: | C11B1_HUMAN | CYP11B1 | CYPXIB1 | Cytochrome P450 11B, mitochondrial precursor | Cytochrome P450 11B1 | Cytochrome P450 11B1 (CYP11B1) | Cytochrome P450 11B1, mitochondrial | S11BH |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57591.44 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P15538 |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALRAKAEVCMAVPWLSLQRAQALGTRAARVPRTVLPFEAMPRRPGNRWLRLLQIWREQG
YEDLHLEVHQTFQELGPIFRYDLGGAGMVCVMLPEDVEKLQQVDSLHPHRMSLEPWVAYR
QHRGHKCGVFLLNGPEWRFNRLRLNPEVLSPNAVQRFLPMVDAVARDFSQALKKKVLQNA
RGSLTLDVQPSIFHYTIEASNLALFGERLGLVGHSPSSASLNFLHALEVMFKSTVQLMFM
PRSLSRWTSPKVWKEHFEAWDCIFQYGDNCIQKIYQELAFSRPQQYTSIVAELLLNAELS
PDAIKANSMELTAGSVDTTVFPLLMTLFELARNPNVQQALRQESLAAAASISEHPQKATT
ELPLLRAALKETLRLYPVGLFLERVASSDLVLQNYHIPAGTLVRVFLYSLGRNPALFPRP
ERYNPQRWLDIRGSGRNFYHVPFGFGMRQCLGRRLAEAEMLLLLHHVLKHLQVETLTQED
IKMVYSFILRPSMFPLLTFRAIN
|
|
|
|---|
| BDBM8871 |
|---|
| BDBM8582 |
|---|
| Name | BDBM8871 |
|---|
| Synonyms: | 5-[(1E)-1,2,3,4-tetrahydronaphthalen-1-ylidenemethyl]-1H-imidazole | 5-[(E)-3,4-Dihydronaphthalen-1(2H)-ylidenemethyl]-1H-imidazole | Imidazolylmethylenetetrahydronaphthalene 1a | Imidazolylmethylenetetrahydronaphthalene 1b |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H14N2 |
|---|
| Mol. Mass. | 210.2744 |
|---|
| SMILES | C1CC(=Cc2cnc[nH]2)c2ccccc2C1 |w:3.3| |
|---|
| Structure | 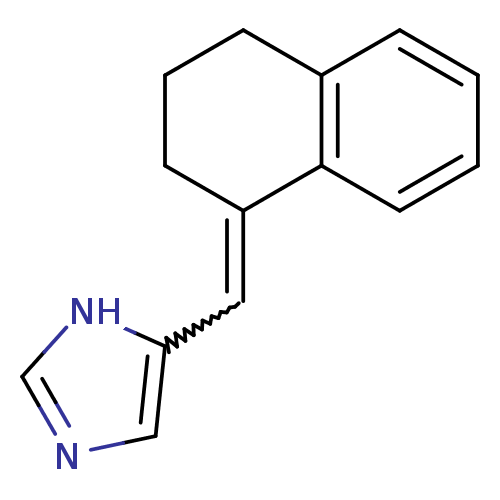
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ulmschneider, S; Muller-Vieira, U; Mitrenga, M; Hartmann, RW; Oberwinkler-Marchais, S; Klein, CD; Bureik, M; Bernhardt, R; Antes, I; Lengauer, T Synthesis and evaluation of imidazolylmethylenetetrahydronaphthalenes and imidazolylmethyleneindanes: potent inhibitors of aldosterone synthase. J Med Chem48:1796-805 (2005) [PubMed] Article
Ulmschneider, S; Muller-Vieira, U; Mitrenga, M; Hartmann, RW; Oberwinkler-Marchais, S; Klein, CD; Bureik, M; Bernhardt, R; Antes, I; Lengauer, T Synthesis and evaluation of imidazolylmethylenetetrahydronaphthalenes and imidazolylmethyleneindanes: potent inhibitors of aldosterone synthase. J Med Chem48:1796-805 (2005) [PubMed] Article