| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Dipeptidyl peptidase 4 |
|---|
| Ligand | BDBM12646 |
|---|
| Substrate/Competitor | BDBM11057 |
|---|
| Meas. Tech. | DPPIV Inhibition Assay |
|---|
| Ki | 3±n/a nM |
|---|
| Citation |  Madar, DJ; Kopecka, H; Pireh, D; Yong, H; Pei, Z; Li, X; Wiedeman, PE; Djuric, SW; Von Geldern, TW; Fickes, MG; Bhagavatula, L; McDermott, T; Wittenberger, S; Richards, SJ; Longenecker, KL; Stewart, KD; Lubben, TH; Ballaron, SJ; Stashko, MA; Long, MA; Wells, H; Zinker, BA; Mika, AK; Beno, DW; Kempf-Grote, AJ; Polakowski, J; Segreti, J; Reinhart, GA; Fryer, RM; Sham, HL; Trevillyan, JM Discovery of 2-[4-{{2-(2S,5R)-2-cyano-5-ethynyl-1-pyrrolidinyl]-2-oxoethyl]amino]-4-methyl-1-piperidinyl]-4-pyridinecarboxylic acid (ABT-279): a very potent, selective, effective, and well-tolerated inhibitor of dipeptidyl peptidase-IV, useful for the treatment of diabetes. J Med Chem49:6416-20 (2006) [PubMed] Article Madar, DJ; Kopecka, H; Pireh, D; Yong, H; Pei, Z; Li, X; Wiedeman, PE; Djuric, SW; Von Geldern, TW; Fickes, MG; Bhagavatula, L; McDermott, T; Wittenberger, S; Richards, SJ; Longenecker, KL; Stewart, KD; Lubben, TH; Ballaron, SJ; Stashko, MA; Long, MA; Wells, H; Zinker, BA; Mika, AK; Beno, DW; Kempf-Grote, AJ; Polakowski, J; Segreti, J; Reinhart, GA; Fryer, RM; Sham, HL; Trevillyan, JM Discovery of 2-[4-{{2-(2S,5R)-2-cyano-5-ethynyl-1-pyrrolidinyl]-2-oxoethyl]amino]-4-methyl-1-piperidinyl]-4-pyridinecarboxylic acid (ABT-279): a very potent, selective, effective, and well-tolerated inhibitor of dipeptidyl peptidase-IV, useful for the treatment of diabetes. J Med Chem49:6416-20 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Dipeptidyl peptidase 4 |
|---|
| Name: | Dipeptidyl peptidase 4 |
|---|
| Synonyms: | ADABP | ADCP2 | Adenosine deaminase complexing protein 2 | CD26 | CD_antigen=CD26 | DPP IV | DPP4 | DPP4_HUMAN | DPPIV | Dipeptidyl peptidase 4 (DDP-IV) | Dipeptidyl peptidase 4 (DPP IV) | Dipeptidyl peptidase 4 (DPP-4) | Dipeptidyl peptidase 4 (DPP4) | Dipeptidyl peptidase 4 (DPPIV) | Dipeptidyl peptidase 4 membrane form | Dipeptidyl peptidase 4 soluble form | Dipeptidyl peptidase IV | Dipeptidyl peptidase IV (DDP-4) | Dipeptidyl peptidase IV (DDP-IV) | Dipeptidyl peptidase IV (DPP IV) | Dipeptidyl peptidase IV membrane form | Dipeptidyl peptidase IV soluble form | Dipeptidyl peptidase-IV (DPP-4) | Dipeptidyl peptidase-IV (DPP-IV) | T-cell activation antigen CD26 | TP103 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 88271.01 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P27487 |
|---|
| Residue: | 766 |
|---|
| Sequence: | MKTPWKVLLGLLGAAALVTIITVPVVLLNKGTDDATADSRKTYTLTDYLKNTYRLKLYSL
RWISDHEYLYKQENNILVFNAEYGNSSVFLENSTFDEFGHSINDYSISPDGQFILLEYNY
VKQWRHSYTASYDIYDLNKRQLITEERIPNNTQWVTWSPVGHKLAYVWNNDIYVKIEPNL
PSYRITWTGKEDIIYNGITDWVYEEEVFSAYSALWWSPNGTFLAYAQFNDTEVPLIEYSF
YSDESLQYPKTVRVPYPKAGAVNPTVKFFVVNTDSLSSVTNATSIQITAPASMLIGDHYL
CDVTWATQERISLQWLRRIQNYSVMDICDYDESSGRWNCLVARQHIEMSTTGWVGRFRPS
EPHFTLDGNSFYKIISNEEGYRHICYFQIDKKDCTFITKGTWEVIGIEALTSDYLYYISN
EYKGMPGGRNLYKIQLSDYTKVTCLSCELNPERCQYYSVSFSKEAKYYQLRCSGPGLPLY
TLHSSVNDKGLRVLEDNSALDKMLQNVQMPSKKLDFIILNETKFWYQMILPPHFDKSKKY
PLLLDVYAGPCSQKADTVFRLNWATYLASTENIIVASFDGRGSGYQGDKIMHAINRRLGT
FEVEDQIEAARQFSKMGFVDNKRIAIWGWSYGGYVTSMVLGSGSGVFKCGIAVAPVSRWE
YYDSVYTERYMGLPTPEDNLDHYRNSTVMSRAENFKQVEYLLIHGTADDNVHFQQSAQIS
KALVDVGVDFQAMWYTDEDHGIASSTAHQHIYTHMSHFIKQCFSLP
|
|
|
|---|
| BDBM12646 |
|---|
| BDBM11057 |
|---|
| Name | BDBM12646 |
|---|
| Synonyms: | (2S,5R)-5-ethynyl-1-(N-(4-methyl-1-(5-cyano-pyridin-2-yl)piperidin-4-yl)glycyl)pyrrolidine-2-carbonitrile | 6-[4-({2-[(2S,5R)-2-cyano-5-ethynylpyrrolidin-1-yl]-2-oxoethyl}amino)-4-methylpiperidin-1-yl]pyridine-3-carbonitrile | ABT-279 analogue | C5-substituted pyrrolidine 40 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H24N6O |
|---|
| Mol. Mass. | 376.4549 |
|---|
| SMILES | CC1(CCN(CC1)c1ccc(cn1)C#N)NCC(=O)N1[C@H](CC[C@H]1C#N)C#C |r| |
|---|
| Structure | 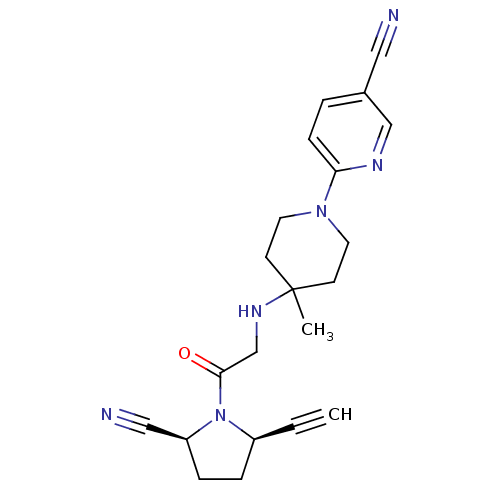
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Madar, DJ; Kopecka, H; Pireh, D; Yong, H; Pei, Z; Li, X; Wiedeman, PE; Djuric, SW; Von Geldern, TW; Fickes, MG; Bhagavatula, L; McDermott, T; Wittenberger, S; Richards, SJ; Longenecker, KL; Stewart, KD; Lubben, TH; Ballaron, SJ; Stashko, MA; Long, MA; Wells, H; Zinker, BA; Mika, AK; Beno, DW; Kempf-Grote, AJ; Polakowski, J; Segreti, J; Reinhart, GA; Fryer, RM; Sham, HL; Trevillyan, JM Discovery of 2-[4-{{2-(2S,5R)-2-cyano-5-ethynyl-1-pyrrolidinyl]-2-oxoethyl]amino]-4-methyl-1-piperidinyl]-4-pyridinecarboxylic acid (ABT-279): a very potent, selective, effective, and well-tolerated inhibitor of dipeptidyl peptidase-IV, useful for the treatment of diabetes. J Med Chem49:6416-20 (2006) [PubMed] Article
Madar, DJ; Kopecka, H; Pireh, D; Yong, H; Pei, Z; Li, X; Wiedeman, PE; Djuric, SW; Von Geldern, TW; Fickes, MG; Bhagavatula, L; McDermott, T; Wittenberger, S; Richards, SJ; Longenecker, KL; Stewart, KD; Lubben, TH; Ballaron, SJ; Stashko, MA; Long, MA; Wells, H; Zinker, BA; Mika, AK; Beno, DW; Kempf-Grote, AJ; Polakowski, J; Segreti, J; Reinhart, GA; Fryer, RM; Sham, HL; Trevillyan, JM Discovery of 2-[4-{{2-(2S,5R)-2-cyano-5-ethynyl-1-pyrrolidinyl]-2-oxoethyl]amino]-4-methyl-1-piperidinyl]-4-pyridinecarboxylic acid (ABT-279): a very potent, selective, effective, and well-tolerated inhibitor of dipeptidyl peptidase-IV, useful for the treatment of diabetes. J Med Chem49:6416-20 (2006) [PubMed] Article