| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prothrombin |
|---|
| Ligand | BDBM13850 |
|---|
| Substrate/Competitor | BDBM13573 |
|---|
| Meas. Tech. | Thrombin Inhibition Assay |
|---|
| Ki | 270±n/a nM |
|---|
| Citation |  Matter, H; Defossa, E; Heinelt, U; Blohm, PM; Schneider, D; Muller, A; Herok, S; Schreuder, H; Liesum, A; Brachvogel, V; Lonze, P; Walser, A; Al-Obeidi, F; Wildgoose, P Design and quantitative structure-activity relationship of 3-amidinobenzyl-1H-indole-2-carboxamides as potent, nonchiral, and selective inhibitors of blood coagulation factor Xa. J Med Chem45:2749-69 (2002) [PubMed] Article Matter, H; Defossa, E; Heinelt, U; Blohm, PM; Schneider, D; Muller, A; Herok, S; Schreuder, H; Liesum, A; Brachvogel, V; Lonze, P; Walser, A; Al-Obeidi, F; Wildgoose, P Design and quantitative structure-activity relationship of 3-amidinobenzyl-1H-indole-2-carboxamides as potent, nonchiral, and selective inhibitors of blood coagulation factor Xa. J Med Chem45:2749-69 (2002) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Prothrombin |
|---|
| Name: | Prothrombin |
|---|
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 70029.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00734 |
|---|
| Residue: | 622 |
|---|
| Sequence: | MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLEREC
VEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHV
NITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQE
CSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASA
QAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETG
DGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYI
DGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTEN
DLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHP
VCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDST
RIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKY
GFYTHVFRLKKWIQKVIDQFGE
|
|
|
|---|
| BDBM13850 |
|---|
| BDBM13573 |
|---|
| Name | BDBM13850 |
|---|
| Synonyms: | 1-[(3-carbamimidoylphenyl)methyl]-4-hydroxy-N-[(2,3,5-trichlorophenyl)methyl]-1H-indole-2-carboxamide | 3-amidinobenzylindole carboxamide 34 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H19Cl3N4O2 |
|---|
| Mol. Mass. | 501.792 |
|---|
| SMILES | NC(=N)c1cccc(Cn2c(cc3c(O)cccc23)C(=O)NCc2cc(Cl)cc(Cl)c2Cl)c1 |
|---|
| Structure | 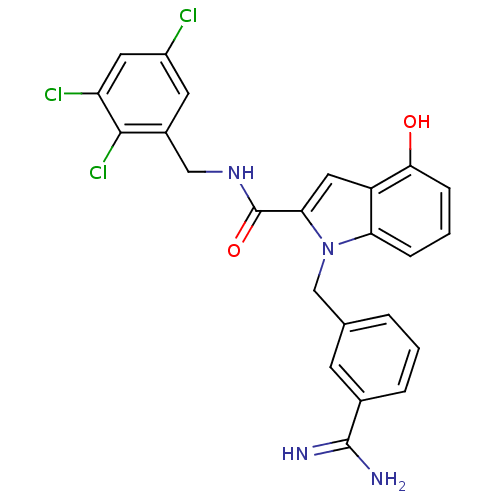
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Matter, H; Defossa, E; Heinelt, U; Blohm, PM; Schneider, D; Muller, A; Herok, S; Schreuder, H; Liesum, A; Brachvogel, V; Lonze, P; Walser, A; Al-Obeidi, F; Wildgoose, P Design and quantitative structure-activity relationship of 3-amidinobenzyl-1H-indole-2-carboxamides as potent, nonchiral, and selective inhibitors of blood coagulation factor Xa. J Med Chem45:2749-69 (2002) [PubMed] Article
Matter, H; Defossa, E; Heinelt, U; Blohm, PM; Schneider, D; Muller, A; Herok, S; Schreuder, H; Liesum, A; Brachvogel, V; Lonze, P; Walser, A; Al-Obeidi, F; Wildgoose, P Design and quantitative structure-activity relationship of 3-amidinobenzyl-1H-indole-2-carboxamides as potent, nonchiral, and selective inhibitors of blood coagulation factor Xa. J Med Chem45:2749-69 (2002) [PubMed] Article