| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tissue-type plasminogen activator |
|---|
| Ligand | BDBM14350 |
|---|
| Substrate/Competitor | BDBM12774 |
|---|
| Meas. Tech. | Enzyme Assay and Determination of the Inhibition Constants |
|---|
| Ki | 29000±n/a nM |
|---|
| Citation |  Katz, BA; Luong, C; Ho, JD; Somoza, JR; Gjerstad, E; Tang, J; Williams, SR; Verner, E; Mackman, RL; Young, WB; Sprengeler, PA; Chan, H; Mortara, K; Janc, JW; McGrath, ME Dissecting and designing inhibitor selectivity determinants at the S1 site using an artificial Ala190 protease (Ala190 uPA). J Mol Biol344:527-47 (2004) [PubMed] Article Katz, BA; Luong, C; Ho, JD; Somoza, JR; Gjerstad, E; Tang, J; Williams, SR; Verner, E; Mackman, RL; Young, WB; Sprengeler, PA; Chan, H; Mortara, K; Janc, JW; McGrath, ME Dissecting and designing inhibitor selectivity determinants at the S1 site using an artificial Ala190 protease (Ala190 uPA). J Mol Biol344:527-47 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Tissue-type plasminogen activator |
|---|
| Name: | Tissue-type plasminogen activator |
|---|
| Synonyms: | Alteplase | PLAT | Reteplase | TPA_HUMAN | Thrombin receptor protein | Tissue-type plasminogen activator | Tissue-type plasminogen activator (tPA) | Tissue-type plasminogen activator precursor | t-PA | t-Plasminogen Activator (tPA) | t-plasminogen activator |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 62931.08 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 562 |
|---|
| Sequence: | MDAMKRGLCCVLLLCGAVFVSPSQEIHARFRRGARSYQVICRDEKTQMIYQQHQSWLRPV
LRSNRVEYCWCNSGRAQCHSVPVKSCSEPRCFNGGTCQQALYFSDFVCQCPEGFAGKCCE
IDTRATCYEDQGISYRGTWSTAESGAECTNWNSSALAQKPYSGRRPDAIRLGLGNHNYCR
NPDRDSKPWCYVFKAGKYSSEFCSTPACSEGNSDCYFGNGSAYRGTHSLTESGASCLPWN
SMILIGKVYTAQNPSAQALGLGKHNYCRNPDGDAKPWCHVLKNRRLTWEYCDVPSCSTCG
LRQYSQPQFRIKGGLFADIASHPWQAAIFAKHRRSPGERFLCGGILISSCWILSAAHCFQ
ERFPPHHLTVILGRTYRVVPGEEEQKFEVEKYIVHKEFDDDTYDNDIALLQLKSDSSRCA
QESSVVRTVCLPPADLQLPDWTECELSGYGKHEALSPFYSERLKEAHVRLYPSSRCTSQH
LLNRTVTDNMLCAGDTRSGGPQANLHDACQGDSGGPLVCLNDGRMTLVGIISWGLGCGQK
DVPGVYTKVTNYLDWIRDNMRP
|
|
|
|---|
| BDBM14350 |
|---|
| BDBM12774 |
|---|
| Name | BDBM14350 |
|---|
| Synonyms: | 2-{5-[amino(iminiumyl)methyl]-6-fluoro-1H-1,3-benzodiazol-2-yl}-6-{[(1S,2S)-2-methylcyclohexyl]oxy}benzen-1-olate | CA-11 | CRA-11092 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H23FN4O2 |
|---|
| Mol. Mass. | 382.4313 |
|---|
| SMILES | C[C@H]1CCCC[C@@H]1Oc1cccc(-c2nc3cc(C(N)=[NH2+])c(F)cc3[nH]2)c1[O-] |r| |
|---|
| Structure | 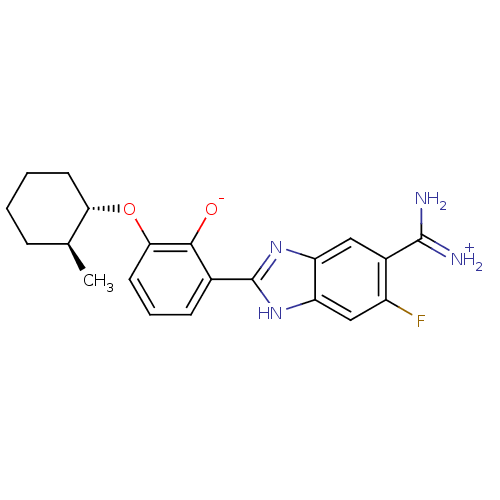
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Katz, BA; Luong, C; Ho, JD; Somoza, JR; Gjerstad, E; Tang, J; Williams, SR; Verner, E; Mackman, RL; Young, WB; Sprengeler, PA; Chan, H; Mortara, K; Janc, JW; McGrath, ME Dissecting and designing inhibitor selectivity determinants at the S1 site using an artificial Ala190 protease (Ala190 uPA). J Mol Biol344:527-47 (2004) [PubMed] Article
Katz, BA; Luong, C; Ho, JD; Somoza, JR; Gjerstad, E; Tang, J; Williams, SR; Verner, E; Mackman, RL; Young, WB; Sprengeler, PA; Chan, H; Mortara, K; Janc, JW; McGrath, ME Dissecting and designing inhibitor selectivity determinants at the S1 site using an artificial Ala190 protease (Ala190 uPA). J Mol Biol344:527-47 (2004) [PubMed] Article