| Reaction Details |
|---|
 | Report a problem with these data |
| Target | cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A [432-779] |
|---|
| Ligand | BDBM14777 |
|---|
| Substrate/Competitor | BDBM10851 |
|---|
| Meas. Tech. | Phosphodiesterase (PDE) Inhibition Assay |
|---|
| IC50 | 19000±n/a nM |
|---|
| Citation |  Card, GL; England, BP; Suzuki, Y; Fong, D; Powell, B; Lee, B; Luu, C; Tabrizizad, M; Gillette, S; Ibrahim, PN; Artis, DR; Bollag, G; Milburn, MV; Kim, SH; Schlessinger, J; Zhang, KY Structural basis for the activity of drugs that inhibit phosphodiesterases. Structure12:2233-47 (2004) [PubMed] Article Card, GL; England, BP; Suzuki, Y; Fong, D; Powell, B; Lee, B; Luu, C; Tabrizizad, M; Gillette, S; Ibrahim, PN; Artis, DR; Bollag, G; Milburn, MV; Kim, SH; Schlessinger, J; Zhang, KY Structural basis for the activity of drugs that inhibit phosphodiesterases. Structure12:2233-47 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A [432-779] |
|---|
| Name: | cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A [432-779] |
|---|
| Synonyms: | PDE10A | PDE10_HUMAN | Phosphodiesterase Type 10 (PDE10A) | cAMP and cAMP-inhibited cGMP 3,5-cyclic phosphodiesterase 10A |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 40036.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Recombinant catalytic domain (M432-D779) of human PDE10A. |
|---|
| Residue: | 348 |
|---|
| Sequence: | MEKLSYHSICTSEEWQGLMQFTLPVRLCKEIELFHFDIGPFENMWPGIFVYMVHRSCGTS
CFELEKLCRFIMSVKKNYRRVPYHNWKHAVTVAHCMYAILQNNHTLFTDLERKGLLIACL
CHDLDHRGFSNSYLQKFDHPLAALYSTSTMEQHHFSQTVSILQLEGHNIFSTLSSSEYEQ
VLEIIRKAIIATDLALYFGNRKQLEEMYQTGSLNLNNQSHRDRVIGLMMTACDLCSVTKL
WPVTKLTANDIYAEFWAEGDEMKKLGIQPIPMMDRDKKDEVPQGQLGFYNAVAIPCYTTL
TQILPPTEPLLKACRDNLSQWEKVIRGEETATWISSPSVAQKAAASED
|
|
|
|---|
| BDBM14777 |
|---|
| BDBM10851 |
|---|
| Name | BDBM14777 |
|---|
| Synonyms: | (2R,8R)-2-(2H-1,3-benzodioxol-5-yl)-6-methyl-3,6,17-triazatetracyclo[8.7.0.0^{3,8}.0^{11,16}]heptadeca-1(10),11(16),12,14-tetraene-4,7-dione | (6R,12aR)-6-(1,3-benzodioxol-5-yl)-2-methyl-2,3,6,7,12,12a-hexahydropyrazino[1,2,1,6]pyrido[3,4-b]indole-1,4-dione | CHEMBL779 | Cialis | GF 196960 | Tadalafil |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H19N3O4 |
|---|
| Mol. Mass. | 389.404 |
|---|
| SMILES | [H][C@]12Cc3c([nH]c4ccccc34)[C@H](N1C(=O)CN(C)C2=O)c1ccc2OCOc2c1 |r| |
|---|
| Structure | 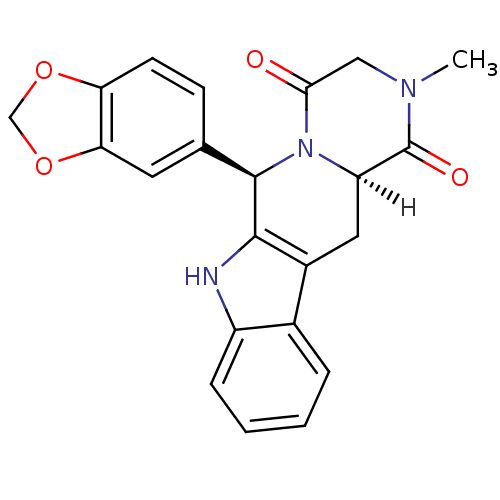
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Card, GL; England, BP; Suzuki, Y; Fong, D; Powell, B; Lee, B; Luu, C; Tabrizizad, M; Gillette, S; Ibrahim, PN; Artis, DR; Bollag, G; Milburn, MV; Kim, SH; Schlessinger, J; Zhang, KY Structural basis for the activity of drugs that inhibit phosphodiesterases. Structure12:2233-47 (2004) [PubMed] Article
Card, GL; England, BP; Suzuki, Y; Fong, D; Powell, B; Lee, B; Luu, C; Tabrizizad, M; Gillette, S; Ibrahim, PN; Artis, DR; Bollag, G; Milburn, MV; Kim, SH; Schlessinger, J; Zhang, KY Structural basis for the activity of drugs that inhibit phosphodiesterases. Structure12:2233-47 (2004) [PubMed] Article