| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A2a |
|---|
| Ligand | BDBM21216 |
|---|
| Substrate/Competitor | BDBM21241 |
|---|
| Meas. Tech. | Human A1 Adenosine Receptor Binding Assay |
|---|
| Ki | 3440±n/a nM |
|---|
| Citation |  Cosimelli, B; Greco, G; Ehlardo, M; Novellino, E; Da Settimo, F; Taliani, S; La Motta, C; Bellandi, M; Tuccinardi, T; Martinelli, A; Ciampi, O; Trincavelli, ML; Martini, C Derivatives of 4-Amino-6-hydroxy-2-mercaptopyrimidine as Novel, Potent, and Selective A3 Adenosine Receptor Antagonists. J Med Chem51:1764-70 (2008) [PubMed] Article Cosimelli, B; Greco, G; Ehlardo, M; Novellino, E; Da Settimo, F; Taliani, S; La Motta, C; Bellandi, M; Tuccinardi, T; Martinelli, A; Ciampi, O; Trincavelli, ML; Martini, C Derivatives of 4-Amino-6-hydroxy-2-mercaptopyrimidine as Novel, Potent, and Selective A3 Adenosine Receptor Antagonists. J Med Chem51:1764-70 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Adenosine receptor A2a |
|---|
| Name: | Adenosine receptor A2a |
|---|
| Synonyms: | A2A adenosine receptor (hA2A) | AA2AR_HUMAN | ADENOSINE A2 | ADENOSINE A2a | ADORA2 | ADORA2A | Adenosine A2A receptor (A2AAR) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44716.46 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P29274 |
|---|
| Residue: | 412 |
|---|
| Sequence: | MPIMGSSVYITVELAIAVLAILGNVLVCWAVWLNSNLQNVTNYFVVSLAAADIAVGVLAI
PFAITISTGFCAACHGCLFIACFVLVLTQSSIFSLLAIAIDRYIAIRIPLRYNGLVTGTR
AKGIIAICWVLSFAIGLTPMLGWNNCGQPKEGKNHSQGCGEGQVACLFEDVVPMNYMVYF
NFFACVLVPLLLMLGVYLRIFLAARRQLKQMESQPLPGERARSTLQKEVHAAKSLAIIVG
LFALCWLPLHIINCFTFFCPDCSHAPLWLMYLAIVLSHTNSVVNPFIYAYRIREFRQTFR
KIIRSHVLRQQEPFKAAGTSARVLAAHGSDGEQVSLRLNGHPPGVWANGSAPHPERRPNG
YALGLVSGGSAQESQGNTGLPDVELLSHELKGVCPEPPGLDDPLAQDGAGVS
|
|
|
|---|
| BDBM21216 |
|---|
| BDBM21241 |
|---|
| Name | BDBM21216 |
|---|
| Synonyms: | 2-[4-(ethylsulfanyl)-1H-1,3-benzodiazol-2-yl]quinoxaline | 2-[4-(ethylsulfanyl)-1H-benzimidazol-2-yl]quinoxaline | CHEMBL198008 | benzimidazole-quinoxaline, C2 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H14N4S |
|---|
| Mol. Mass. | 306.385 |
|---|
| SMILES | CCSc1cccc2nc([nH]c12)-c1cnc2ccccc2n1 |
|---|
| Structure | 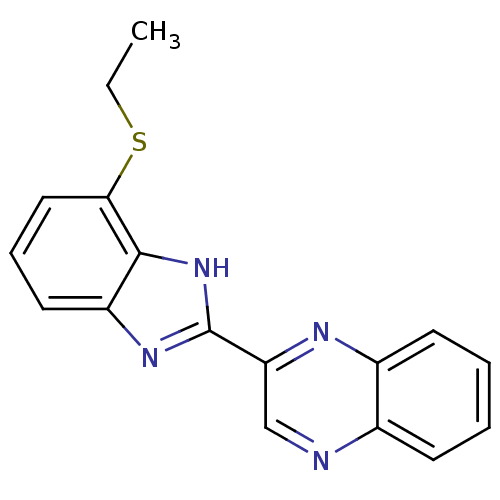
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Cosimelli, B; Greco, G; Ehlardo, M; Novellino, E; Da Settimo, F; Taliani, S; La Motta, C; Bellandi, M; Tuccinardi, T; Martinelli, A; Ciampi, O; Trincavelli, ML; Martini, C Derivatives of 4-Amino-6-hydroxy-2-mercaptopyrimidine as Novel, Potent, and Selective A3 Adenosine Receptor Antagonists. J Med Chem51:1764-70 (2008) [PubMed] Article
Cosimelli, B; Greco, G; Ehlardo, M; Novellino, E; Da Settimo, F; Taliani, S; La Motta, C; Bellandi, M; Tuccinardi, T; Martinelli, A; Ciampi, O; Trincavelli, ML; Martini, C Derivatives of 4-Amino-6-hydroxy-2-mercaptopyrimidine as Novel, Potent, and Selective A3 Adenosine Receptor Antagonists. J Med Chem51:1764-70 (2008) [PubMed] Article