| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cannabinoid receptor 1 |
|---|
| Ligand | BDBM21280 |
|---|
| Substrate/Competitor | BDBM21244 |
|---|
| Meas. Tech. | CB Receptor Binding Assay |
|---|
| Ki | >2820±n/a nM |
|---|
| Citation |  Silvestri, R; Cascio, MG; La Regina, G; Piscitelli, F; Lavecchia, A; Brizzi, A; Pasquini, S; Botta, M; Novellino, E; Di Marzo, V; Corelli, F Synthesis, Cannabinoid Receptor Affinity, and Molecular Modeling Studies of Substituted 1-Aryl-5-(1H-pyrrol-1-yl)-1H-pyrazole-3-carboxamides. J Med Chem51:1560-76 (2008) [PubMed] Article Silvestri, R; Cascio, MG; La Regina, G; Piscitelli, F; Lavecchia, A; Brizzi, A; Pasquini, S; Botta, M; Novellino, E; Di Marzo, V; Corelli, F Synthesis, Cannabinoid Receptor Affinity, and Molecular Modeling Studies of Substituted 1-Aryl-5-(1H-pyrrol-1-yl)-1H-pyrazole-3-carboxamides. J Med Chem51:1560-76 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Cannabinoid receptor 1 |
|---|
| Name: | Cannabinoid receptor 1 |
|---|
| Synonyms: | CANN6 | CANNABINOID CB1 | CB-R | CB1 | CNR | CNR1 | CNR1_HUMAN | Cannabinoid CB1 receptor | Cannabinoid receptor | Cannabinoid receptor 1 (CB1) | Cannabinoid receptor 1 (brain) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52868.96 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21554 |
|---|
| Residue: | 472 |
|---|
| Sequence: | MKSILDGLADTTFRTITTDLLYVGSNDIQYEDIKGDMASKLGYFPQKFPLTSFRGSPFQE
KMTAGDNPQLVPADQVNITEFYNKSLSSFKENEENIQCGENFMDIECFMVLNPSQQLAIA
VLSLTLGTFTVLENLLVLCVILHSRSLRCRPSYHFIGSLAVADLLGSVIFVYSFIDFHVF
HRKDSRNVFLFKLGGVTASFTASVGSLFLTAIDRYISIHRPLAYKRIVTRPKAVVAFCLM
WTIAIVIAVLPLLGWNCEKLQSVCSDIFPHIDETYLMFWIGVTSVLLLFIVYAYMYILWK
AHSHAVRMIQRGTQKSIIIHTSEDGKVQVTRPDQARMDIRLAKTLVLILVVLIICWGPLL
AIMVYDVFGKMNKLIKTVFAFCSMLCLLNSTVNPIIYALRSKDLRHAFRSMFPSCEGTAQ
PLDNSMGDSDCLHKHANNAASVHRAAESCIKSTVKIAKVTMSVSTDTSAEAL
|
|
|
|---|
| BDBM21280 |
|---|
| BDBM21244 |
|---|
| Name | BDBM21280 |
|---|
| Synonyms: | 5-(4-chloro-3-methylphenyl)-1-[(4-methylphenyl)methyl]-N-[(1S)-1,3,3-trimethylbicyclo[2.2.1]heptan-2-yl]-1H-pyrazole-3-carboxamide | N-(1,3,3-trimethylbicyclo(2.2.1)heptan-2-yl)-5-(4-chloro-3-methylphenyl)-1-(4-methylbenzyl)pyrazole-3-carboxamide | SR 144528 | SR-144528 | SR144528 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H34ClN3O |
|---|
| Mol. Mass. | 476.053 |
|---|
| SMILES | Cc1ccc(Cn2nc(cc2-c2ccc(Cl)c(C)c2)C(=O)NC2[C@@]3(C)CCC(C3)C2(C)C)cc1 |r,TLB:21:22:28:26.25| |
|---|
| Structure | 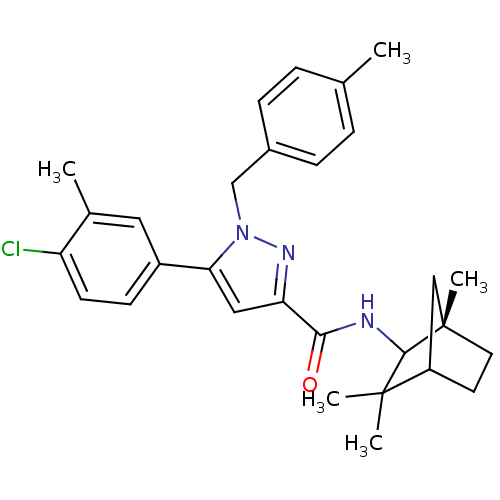
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Silvestri, R; Cascio, MG; La Regina, G; Piscitelli, F; Lavecchia, A; Brizzi, A; Pasquini, S; Botta, M; Novellino, E; Di Marzo, V; Corelli, F Synthesis, Cannabinoid Receptor Affinity, and Molecular Modeling Studies of Substituted 1-Aryl-5-(1H-pyrrol-1-yl)-1H-pyrazole-3-carboxamides. J Med Chem51:1560-76 (2008) [PubMed] Article
Silvestri, R; Cascio, MG; La Regina, G; Piscitelli, F; Lavecchia, A; Brizzi, A; Pasquini, S; Botta, M; Novellino, E; Di Marzo, V; Corelli, F Synthesis, Cannabinoid Receptor Affinity, and Molecular Modeling Studies of Substituted 1-Aryl-5-(1H-pyrrol-1-yl)-1H-pyrazole-3-carboxamides. J Med Chem51:1560-76 (2008) [PubMed] Article