| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Disintegrin and metalloproteinase domain-containing protein 17 [215-477,S266A,N452Q] |
|---|
| Ligand | BDBM23455 |
|---|
| Substrate/Competitor | BDBM23453 |
|---|
| Meas. Tech. | TACE Inhibition Assay |
|---|
| pH | 7.3±n/a |
|---|
| Temperature | 295.15±n/a K |
|---|
| Ki | 700±n/a nM |
|---|
| Citation |  Zhu, Z; Mazzola, R; Sinning, L; McKittrick, B; Niu, X; Lundell, D; Sun, J; Orth, P; Guo, Z; Madison, V; Ingram, R; Beyer, BM Discovery of novel hydroxamates as highly potent tumor necrosis factor-alpha converting enzyme inhibitors: Part I--discovery of two binding modes. J Med Chem51:725-36 (2008) [PubMed] Article Zhu, Z; Mazzola, R; Sinning, L; McKittrick, B; Niu, X; Lundell, D; Sun, J; Orth, P; Guo, Z; Madison, V; Ingram, R; Beyer, BM Discovery of novel hydroxamates as highly potent tumor necrosis factor-alpha converting enzyme inhibitors: Part I--discovery of two binding modes. J Med Chem51:725-36 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Inhibition_Run data, Solution Info, Assay Method |
|---|
| |
| Disintegrin and metalloproteinase domain-containing protein 17 [215-477,S266A,N452Q] |
|---|
| Name: | Disintegrin and metalloproteinase domain-containing protein 17 [215-477,S266A,N452Q] |
|---|
| Synonyms: | A disintegrin and metalloproteinase domain 17 | ADA17_HUMAN | ADAM 17 | ADAM17 | CD156b antigen | CSVP | Snake venom-like protease | TACE | TNF-alpha-Converting Enzyme |
|---|
| Type: | Single-pass type I membrane protein; metalloprotease |
|---|
| Mol. Mass.: | 29695.98 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | The catalytic domain of recombinant human TNF-converting enzyme (residues 215-477) with two mutations (S266A and N452Q) and a 6xHis was purified from the baculovirus/Hi5 cells expression system. |
|---|
| Residue: | 263 |
|---|
| Sequence: | RADPDPMKNTCKLLVVADHRFYRYMGRGEESTTTNYLIELIDRVDDIYRNTAWDNAGFKG
YGIQIEQIRILKSPQEVKPGEKHYNMAKSYPNEEKDAWDVKMLLEQFSFDIAEEASKVCL
AHLFTYQDFDMGTLGLAYVGSPRANSHGGVCPKAYYSPVGKKNIYLNSGLTSTKNYGKTI
LTKEADLVTTHELGHNFGAEHDPDGLAECAPNEDQGGKYVMYPIAVSGDHENNKMFSQCS
KQSIYKTIESKAQECFQERSNKV
|
|
|
|---|
| BDBM23455 |
|---|
| BDBM23453 |
|---|
| Name | BDBM23455 |
|---|
| Synonyms: | (1R,2R)-1-N-hydroxy-2-{3-[(2-methylquinolin-4-yl)methoxy]phenyl}cyclopropane-1,2-dicarboxamide | hydroxamate deriv., 29 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H21N3O4 |
|---|
| Mol. Mass. | 391.4198 |
|---|
| SMILES | Cc1cc(COc2cccc(c2)[C@]2(C[C@H]2C(=O)NO)C(N)=O)c2ccccc2n1 |r| |
|---|
| Structure | 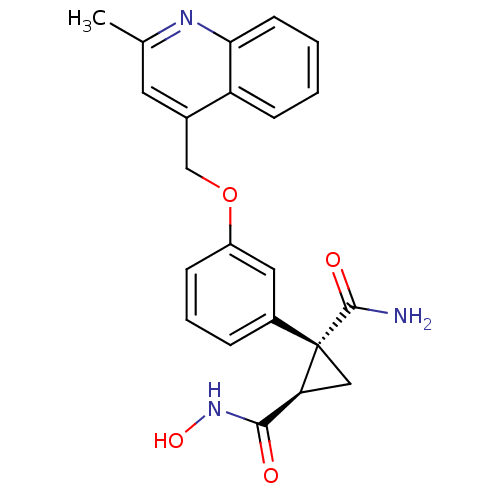
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zhu, Z; Mazzola, R; Sinning, L; McKittrick, B; Niu, X; Lundell, D; Sun, J; Orth, P; Guo, Z; Madison, V; Ingram, R; Beyer, BM Discovery of novel hydroxamates as highly potent tumor necrosis factor-alpha converting enzyme inhibitors: Part I--discovery of two binding modes. J Med Chem51:725-36 (2008) [PubMed] Article
Zhu, Z; Mazzola, R; Sinning, L; McKittrick, B; Niu, X; Lundell, D; Sun, J; Orth, P; Guo, Z; Madison, V; Ingram, R; Beyer, BM Discovery of novel hydroxamates as highly potent tumor necrosis factor-alpha converting enzyme inhibitors: Part I--discovery of two binding modes. J Med Chem51:725-36 (2008) [PubMed] Article