| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Zinc finger protein mex-5 |
|---|
| Ligand | BDBM32274 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Fluorescent Polarization Homogeneous Dose Response HTS to Indentify Inhibitors of Mex-5 Binding to TCR-2 |
|---|
| EC50 | 4179±n/a nM |
|---|
| Citation |  PubChem, PC Fluorescent Polarization Homogeneous Dose Response HTS to Indentify Inhibitors of Mex-5 Binding to TCR-2 PubChem Bioassay(2009)[AID] PubChem, PC Fluorescent Polarization Homogeneous Dose Response HTS to Indentify Inhibitors of Mex-5 Binding to TCR-2 PubChem Bioassay(2009)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Zinc finger protein mex-5 |
|---|
| Name: | Zinc finger protein mex-5 |
|---|
| Synonyms: | MEX5_CAEEL | RecName: Full=Zinc finger protein mex-5 | mex-5 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 52832.78 |
|---|
| Organism: | Caenorhabditis elegans |
|---|
| Description: | gi_55976631 |
|---|
| Residue: | 468 |
|---|
| Sequence: | MKAASNSVSSAGGSVSPTTTQPPLPPGQSSHPQIYDQQMQYYFAAAMPNQPMATYAAQNG
SSQQYAPAAPYYQDANGQYVQVPANGSMAPQQHMMVSGQPYLYMAQPQQGAQQVMQSGQP
QLIYYQQSMAPQAAPMYFHPMQAAPMLPEQMGVMPHTQPAIPPQQQPRQVGVEISSTRTA
PLTSSTPLPTSLEYETVQRDNRNRNIQFRYHRVMEHDELPIDEISKITLDNHNDDTMSAE
KENHFHEHRGEKFGRRGFPIPETDSQQPPNYKTRLCMMHASGIKPCDMGARCKFAHGLKE
LRATDAPARYPNNKYKTKLCKNFARGGTGFCPYGLRCEFVHPTDKEFQNIPPYQRMSHDD
QDYDQDVIPEDYVVARHQPRFMRTGGRATTPTKVMLKHRNVAGSMMCLSNAGRDLQAGGD
YNQPESNEDDLPPHLRRNRRENPPMNKRRTSLSTKWTSEENLGLRGHY
|
|
|
|---|
| BDBM32274 |
|---|
| n/a |
|---|
| Name | BDBM32274 |
|---|
| Synonyms: | 2,5-bis(chloranyl)-3-(4-methylpiperazin-1-yl)-6-(2-piperidin-1-yl-1,3-thiazol-5-yl)cyclohexa-2,5-diene-1,4-dione | 2,5-dichloro-3-(4-methyl-1-piperazinyl)-6-[2-(1-piperidinyl)-1,3-thiazol-5-yl]benzo-1,4-quinone | 2,5-dichloro-3-(4-methyl-1-piperazinyl)-6-[2-(1-piperidinyl)-5-thiazolyl]cyclohexa-2,5-diene-1,4-dione | 2,5-dichloro-3-(4-methylpiperazin-1-yl)-6-(2-piperidin-1-yl-1,3-thiazol-5-yl)cyclohexa-2,5-diene-1,4-dione | 2,5-dichloro-3-(4-methylpiperazino)-6-(2-piperidinothiazol-5-yl)-p-benzoquinone | MLS000548207 | SMR000115103 | cid_1002249 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H22Cl2N4O2S |
|---|
| Mol. Mass. | 441.375 |
|---|
| SMILES | CN1CCN(CC1)C1=C(Cl)C(=O)C(c2cnc(s2)N2CCCCC2)=C(Cl)C1=O |c:8,t:26| |
|---|
| Structure | 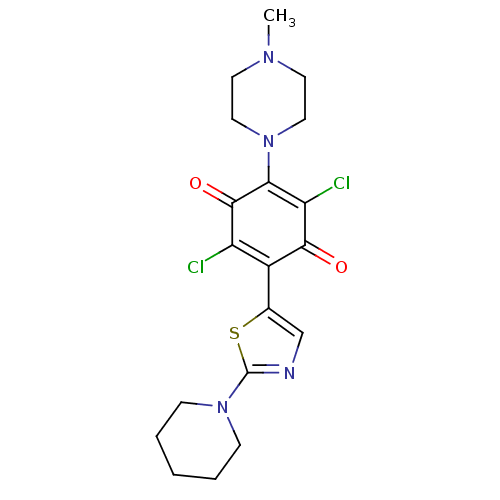
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Fluorescent Polarization Homogeneous Dose Response HTS to Indentify Inhibitors of Mex-5 Binding to TCR-2 PubChem Bioassay(2009)[AID]
PubChem, PC Fluorescent Polarization Homogeneous Dose Response HTS to Indentify Inhibitors of Mex-5 Binding to TCR-2 PubChem Bioassay(2009)[AID]