| Reaction Details |
|---|
 | Report a problem with these data |
| Target | M18 aspartyl aminopeptidase |
|---|
| Ligand | BDBM55142 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | QFRET-based biochemical high throughput dose response assay for inhibitors of the Plasmodium falciparum M18 Aspartyl Aminopeptidase (PFM18AAP). |
|---|
| IC50 | 609.09±n/a nM |
|---|
| Citation |  PubChem, PC QFRET-based biochemical high throughput dose response assay for inhibitors of the Plasmodium falciparum M18 Aspartyl Aminopeptidase (PFM18AAP). PubChem Bioassay(2009)[AID] PubChem, PC QFRET-based biochemical high throughput dose response assay for inhibitors of the Plasmodium falciparum M18 Aspartyl Aminopeptidase (PFM18AAP). PubChem Bioassay(2009)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| M18 aspartyl aminopeptidase |
|---|
| Name: | M18 aspartyl aminopeptidase |
|---|
| Synonyms: | n/a |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 65642.43 |
|---|
| Organism: | Plasmodium falciparum 3D7 |
|---|
| Description: | gi_23505220 |
|---|
| Residue: | 570 |
|---|
| Sequence: | MDKKAREYAQDALKFIQRSGSNFLACKNLKERLENNGFINLSEGETWNLNKNEGYVLCKE
NRNICGFFVGKNFNIDTGSILISIGHIDSCALKISPNNNVIKKKIHQINVECYGSGLWHT
WFDRSLGLSGQVLYKKGNKLVEKLIQINKSVLFLPSLAIHLQNRTRYDFSVKINYENHIK
PIISTTLFNQLNKCKRNNVHHDTILTTDTKFSHKENSQNKRDDQMCHSFNDKDVSNHNLD
KNTIEHLTNQQNEEKNKHTKDNPNSKDIVEHINTDNSYPLLYLLSKELNCKEEDILDFEL
CLMDTQEPCFTGVYEEFIEGARFDNLLGSFCVFEGFIELVNSIKNHTSNENTNHTNNITN
DINDNIHNNLYISIGYDHEEIGSLSEVGARSYCTKNFIDRIISSVFKKEIHEKNLSVQEI
YGNLVNRSFILNVDMAHCSHPNYPETVQDNHQLFFHEGIAIKYNTNKNYVTSPLHASLIK
RTFELYYNKYKQQIKYQNFMVKNDTPCGSTVGSMVAANLSMPGIDIGIPQLAMHSIREIA
AVHDVFFLIKGVFAFYTYYNQVLSTCVHDK
|
|
|
|---|
| BDBM55142 |
|---|
| n/a |
|---|
| Name | BDBM55142 |
|---|
| Synonyms: | 3-nitro-4-(1H-1,2,4-triazol-5-ylthio)benzoic acid [2-(carbethoxyamino)-2-keto-ethyl] ester | 3-nitro-4-(1H-1,2,4-triazol-5-ylthio)benzoic acid [2-(ethoxycarbonylamino)-2-oxoethyl] ester | MLS001004551 | SMR000348099 | [2-(ethoxycarbonylamino)-2-oxidanylidene-ethyl] 3-nitro-4-(1H-1,2,4-triazol-5-ylsulfanyl)benzoate | [2-(ethoxycarbonylamino)-2-oxoethyl] 3-nitro-4-(1H-1,2,4-triazol-5-ylsulfanyl)benzoate | cid_2083851 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H13N5O7S |
|---|
| Mol. Mass. | 395.347 |
|---|
| SMILES | CCOC(=O)NC(=O)COC(=O)c1ccc(Sc2nnc[nH]2)c(c1)[N+]([O-])=O |
|---|
| Structure | 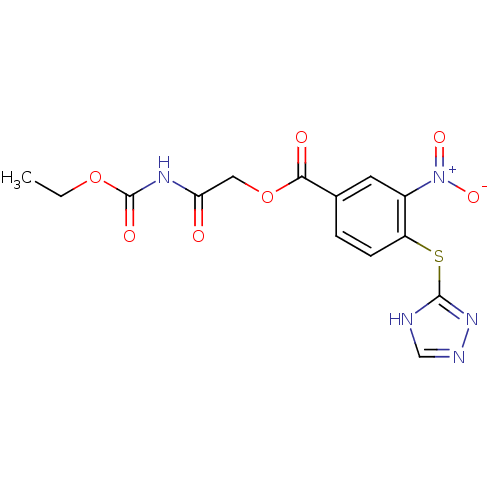
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC QFRET-based biochemical high throughput dose response assay for inhibitors of the Plasmodium falciparum M18 Aspartyl Aminopeptidase (PFM18AAP). PubChem Bioassay(2009)[AID]
PubChem, PC QFRET-based biochemical high throughput dose response assay for inhibitors of the Plasmodium falciparum M18 Aspartyl Aminopeptidase (PFM18AAP). PubChem Bioassay(2009)[AID]