| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Guanine nucleotide-binding protein G(o) subunit alpha |
|---|
| Ligand | BDBM67702 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Dose response, multiplexed high-throughput screen for small molecule regulators of RGS family protein interactions, specifically RGS8-Galphao with additional round of SAR compounds |
|---|
| EC50 | >30000±0 nM |
|---|
| Citation |  PubChem, PC Dose response, multiplexed high-throughput screen for small molecule regulators of RGS family protein interactions, specifically RGS8-Galphao with additional round of SAR compounds PubChem Bioassay(2010)[AID] PubChem, PC Dose response, multiplexed high-throughput screen for small molecule regulators of RGS family protein interactions, specifically RGS8-Galphao with additional round of SAR compounds PubChem Bioassay(2010)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Guanine nucleotide-binding protein G(o) subunit alpha |
|---|
| Name: | Guanine nucleotide-binding protein G(o) subunit alpha |
|---|
| Synonyms: | GNAO1 | GNAO_HUMAN | Guanine nucleotide-binding protein G(o), alpha subunit 1 | guanine nucleotide-binding protein G(o) subunit alpha isoform a |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 40043.59 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_321106 |
|---|
| Residue: | 354 |
|---|
| Sequence: | MGCTLSAEERAALERSKAIEKNLKEDGISAAKDVKLLLLGAGESGKSTIVKQMKIIHEDG
FSGEDVKQYKPVVYSNTIQSLAAIVRAMDTLGIEYGDKERKADAKMVCDVVSRMEDTEPF
SAELLSAMMRLWGDSGIQECFNRSREYQLNDSAKYYLDSLDRIGAADYQPTEQDILRTRV
KTTGIVETHFTFKNLHFRLFDVGGQRSERKKWIHCFEDVTAIIFCVALSGYDQVLHEDET
TNRMHESLMLFDSICNNKFFIDTSIILFLNKKDLFGEKIKKSPLTICFPEYTGPNTYEDA
AAYIQAQFESKNRSPNKEIYCHMTCATDTNNIQVVFDAVTDIIIANNLRGCGLY
|
|
|
|---|
| BDBM67702 |
|---|
| n/a |
|---|
| Name | BDBM67702 |
|---|
| Synonyms: | 3-chloranyl-N-(3-imidazol-1-ylpropyl)-6-nitro-1-benzothiophene-2-carboxamide | 3-chloro-N-(3-imidazol-1-ylpropyl)-6-nitro-1-benzothiophene-2-carboxamide | 3-chloro-N-(3-imidazol-1-ylpropyl)-6-nitro-benzothiophene-2-carboxamide | 3-chloro-N-[3-(1-imidazolyl)propyl]-6-nitro-1-benzothiophene-2-carboxamide | KSC-1-102c | KUC103875N | cid_2933863 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H13ClN4O3S |
|---|
| Mol. Mass. | 364.807 |
|---|
| SMILES | [O-][N+](=O)c1ccc2c(Cl)c(sc2c1)C(=O)NCCCn1ccnc1 |
|---|
| Structure | 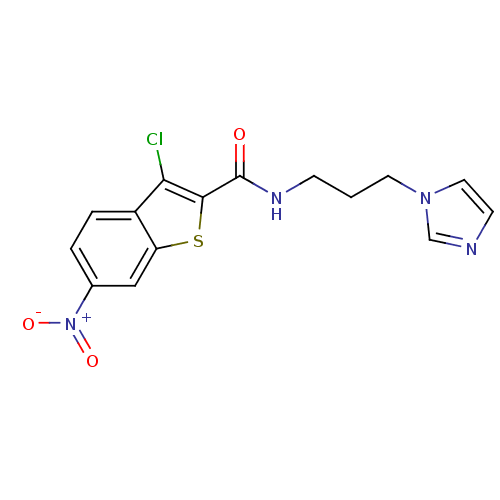
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Dose response, multiplexed high-throughput screen for small molecule regulators of RGS family protein interactions, specifically RGS8-Galphao with additional round of SAR compounds PubChem Bioassay(2010)[AID]
PubChem, PC Dose response, multiplexed high-throughput screen for small molecule regulators of RGS family protein interactions, specifically RGS8-Galphao with additional round of SAR compounds PubChem Bioassay(2010)[AID]