| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 7 |
|---|
| Ligand | BDBM31046 |
|---|
| Substrate/Competitor | n/a |
|---|
| Ki | 4.2±n/a nM |
|---|
| Comments | PDSP_1479 |
|---|
| Citation |  Greenberg, DA; Prichard, DC; Snyder, SH Alpha-noradrenergic receptor binding in mammalian brain: differential labeling of agonist and antagonist states. Life Sci19:69-76 (1976) [PubMed] Article Greenberg, DA; Prichard, DC; Snyder, SH Alpha-noradrenergic receptor binding in mammalian brain: differential labeling of agonist and antagonist states. Life Sci19:69-76 (1976) [PubMed] Article |
|---|
| More Info.: | Get all data from this article |
|---|
| |
| 5-hydroxytryptamine receptor 7 |
|---|
| Name: | 5-hydroxytryptamine receptor 7 |
|---|
| Synonyms: | 5-HT-7 | 5-HT-X | 5-HT7 | 5-HT7L | 5-HT7S | 5-Hydroxytryptamine receptor 7 (5-HT7) | 5-hydroxytryptamine receptor 7 | 5-hydroxytryptamine receptor 7 (5-HT-7) | 5HT7R_RAT | Adrenergic Alpha | GPRFO | Htr7 | Serotonin (5-HT) receptor | Serotonin Receptor 7 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 49852.62 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Rat cloned 5-HT7R. |
|---|
| Residue: | 448 |
|---|
| Sequence: | MMDVNSSGRPDLYGHLRSLILPEVGRGLQDLSPDGGAHPVVSSWMPHLLSGFLEVTASPA
PTWDAPPDNVSGCGEQINYGRVEKVVIGSILTLITLLTIAGNCLVVISVCFVKKLRQPSN
YLIVSLALADLSVAVAVMPFVSVTDLIGGKWIFGHFFCNVFIAMDVMCCTASIMTLCVIS
IDRYLGITRPLTYPVRQNGKCMAKMILSVWLLSASITLPPLFGWAQNVNDDKVCLISQDF
GYTIYSTAVAFYIPMSVMLFMYYQIYKAARKSAAKHKFPGFPRVQPESVISLNGVVKLQK
EVEECANLSRLLKHERKNISIFKREQKAATTLGIIVGAFTVCWLPFFLLSTARPFICGTS
CSCIPLWVERTCLWLGYANSLINPFIYAFFNRDLRTTYRSLLQCQYRNINRKLSAAGMHE
ALKLAERPERSEFVLQNSDHCGKKGHDT
|
|
|
|---|
| BDBM31046 |
|---|
| n/a |
|---|
| Name | BDBM31046 |
|---|
| Synonyms: | 3-[4,5-dihydro-1H-imidazol-2-ylmethyl-(4-methylphenyl)amino]phenol;methanesulfonic acid | 3-[N-(2-imidazolin-2-ylmethyl)-4-methyl-anilino]phenol;hydrochloride | 3-[N-(2-imidazolin-2-ylmethyl)-4-methyl-anilino]phenol;mesylic acid | 3-[N-(4,5-dihydro-1H-imidazol-2-ylmethyl)-4-methylanilino]phenol;methanesulfonic acid | MLS000069487 | PHENTOLAMINE | PHENTOLAMINE MESYLATE | SMR000059054 | cid_5775 | cid_91430 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H19N3O |
|---|
| Mol. Mass. | 281.3523 |
|---|
| SMILES | Cc1ccc(cc1)N(CC1=NCCN1)c1cccc(O)c1 |t:10| |
|---|
| Structure | 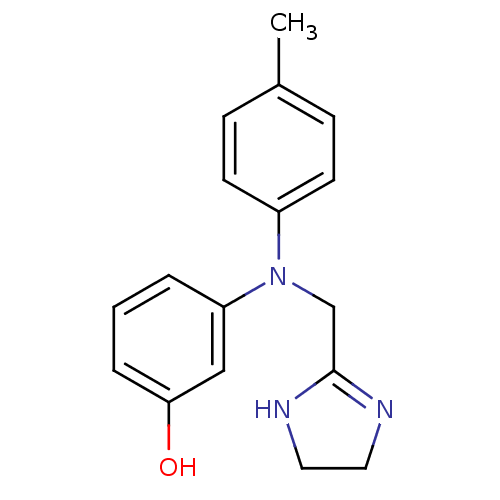
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Greenberg, DA; Prichard, DC; Snyder, SH Alpha-noradrenergic receptor binding in mammalian brain: differential labeling of agonist and antagonist states. Life Sci19:69-76 (1976) [PubMed] Article
Greenberg, DA; Prichard, DC; Snyder, SH Alpha-noradrenergic receptor binding in mammalian brain: differential labeling of agonist and antagonist states. Life Sci19:69-76 (1976) [PubMed] Article