| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-2A adrenergic receptor |
|---|
| Ligand | BDBM31046 |
|---|
| Substrate/Competitor | n/a |
|---|
| Ki | 4.22±n/a nM |
|---|
| Comments | PDSP_1479 |
|---|
| Citation |  Bylund, DB; Blaxall, HS; Iversen, LJ; Caron, MG; Lefkowitz, RJ; Lomasney, JW Pharmacological characteristics of alpha 2-adrenergic receptors: comparison of pharmacologically defined subtypes with subtypes identified by molecular cloning. Mol Pharmacol42:1-5 (1992) [PubMed] Bylund, DB; Blaxall, HS; Iversen, LJ; Caron, MG; Lefkowitz, RJ; Lomasney, JW Pharmacological characteristics of alpha 2-adrenergic receptors: comparison of pharmacologically defined subtypes with subtypes identified by molecular cloning. Mol Pharmacol42:1-5 (1992) [PubMed] |
|---|
| More Info.: | Get all data from this article |
|---|
| |
| Alpha-2A adrenergic receptor |
|---|
| Name: | Alpha-2A adrenergic receptor |
|---|
| Synonyms: | ADA2A_HUMAN | ADRA2A | ADRA2R | ADRAR | Adrenergic alpha2A | Adrenergic receptor alpha | Alpha-2 adrenergic receptor subtype C10 | Alpha-2A adrenoceptor | Alpha-2A adrenoreceptor | Alpha-2AAR | alpha-2A adrenergic receptor [Homo sapiens] |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48979.91 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P08913 |
|---|
| Residue: | 450 |
|---|
| Sequence: | MGSLQPDAGNASWNGTEAPGGGARATPYSLQVTLTLVCLAGLLMLLTVFGNVLVIIAVFT
SRALKAPQNLFLVSLASADILVATLVIPFSLANEVMGYWYFGKAWCEIYLALDVLFCTSS
IVHLCAISLDRYWSITQAIEYNLKRTPRRIKAIIITVWVISAVISFPPLISIEKKGGGGG
PQPAEPRCEINDQKWYVISSCIGSFFAPCLIMILVYVRIYQIAKRRTRVPPSRRGPDAVA
APPGGTERRPNGLGPERSAGPGGAEAEPLPTQLNGAPGEPAPAGPRDTDALDLEESSSSD
HAERPPGPRRPERGPRGKGKARASQVKPGDSLPRRGPGATGIGTPAAGPGEERVGAAKAS
RWRGRQNREKRFTFVLAVVIGVFVVCWFPFFFTYTLTAVGCSVPRTLFKFFFWFGYCNSS
LNPVIYTIFNHDFRRAFKKILCRGDRKRIV
|
|
|
|---|
| BDBM31046 |
|---|
| n/a |
|---|
| Name | BDBM31046 |
|---|
| Synonyms: | 3-[4,5-dihydro-1H-imidazol-2-ylmethyl-(4-methylphenyl)amino]phenol;methanesulfonic acid | 3-[N-(2-imidazolin-2-ylmethyl)-4-methyl-anilino]phenol;hydrochloride | 3-[N-(2-imidazolin-2-ylmethyl)-4-methyl-anilino]phenol;mesylic acid | 3-[N-(4,5-dihydro-1H-imidazol-2-ylmethyl)-4-methylanilino]phenol;methanesulfonic acid | MLS000069487 | PHENTOLAMINE | PHENTOLAMINE MESYLATE | SMR000059054 | cid_5775 | cid_91430 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H19N3O |
|---|
| Mol. Mass. | 281.3523 |
|---|
| SMILES | Cc1ccc(cc1)N(CC1=NCCN1)c1cccc(O)c1 |t:10| |
|---|
| Structure | 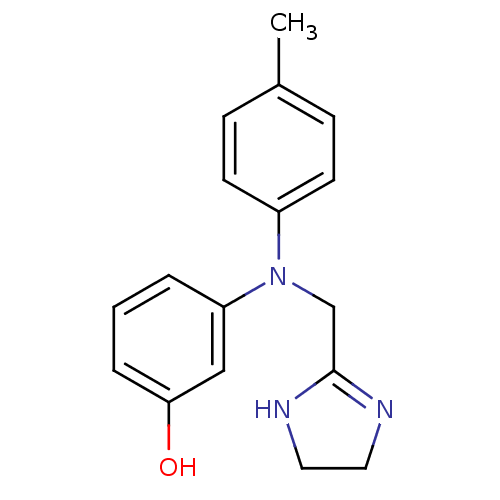
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bylund, DB; Blaxall, HS; Iversen, LJ; Caron, MG; Lefkowitz, RJ; Lomasney, JW Pharmacological characteristics of alpha 2-adrenergic receptors: comparison of pharmacologically defined subtypes with subtypes identified by molecular cloning. Mol Pharmacol42:1-5 (1992) [PubMed]
Bylund, DB; Blaxall, HS; Iversen, LJ; Caron, MG; Lefkowitz, RJ; Lomasney, JW Pharmacological characteristics of alpha 2-adrenergic receptors: comparison of pharmacologically defined subtypes with subtypes identified by molecular cloning. Mol Pharmacol42:1-5 (1992) [PubMed]