| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cholecystokinin |
|---|
| Ligand | BDBM50007921 |
|---|
| Substrate/Competitor | n/a |
|---|
| Ki | 28.6±n/a nM |
|---|
| Comments | PDSP_2673 |
|---|
| Citation |  Van Dijk, A; Richards, JG; Trzeciak, A; Gillessen, D; Möhler, H Cholecystokinin receptors: biochemical demonstration and autoradiographical localization in rat brain and pancreas using [3H] cholecystokinin8 as radioligand. J Neurosci4:1021-33 (1984) [PubMed] Van Dijk, A; Richards, JG; Trzeciak, A; Gillessen, D; Möhler, H Cholecystokinin receptors: biochemical demonstration and autoradiographical localization in rat brain and pancreas using [3H] cholecystokinin8 as radioligand. J Neurosci4:1021-33 (1984) [PubMed] |
|---|
| More Info.: | Get all data from this article |
|---|
| |
| Cholecystokinin |
|---|
| Name: | Cholecystokinin |
|---|
| Synonyms: | CCKN_RAT | Cck |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 12844.29 |
|---|
| Organism: | RAT |
|---|
| Description: | Cholecystokinin 0 RAT::P01355 |
|---|
| Residue: | 115 |
|---|
| Sequence: | MKCGVCLCVVMAVLAAGALAQPVVPVEAVDPMEQRAEEAPRRQLRAVLRPDSEPRARLGA
LLARYIQQVRKAPSGRMSVLKNLQGLDPSHRISDRDYMGWMDFGRRSAEDYEYPS
|
|
|
|---|
| BDBM50007921 |
|---|
| n/a |
|---|
| Name | BDBM50007921 |
|---|
| Synonyms: | 3-{2-[2-(2-{2-[2-(2-Amino-3-carboxy-propionylamino)-3-(4-hydroxy-phenyl)-propionylamino]-4-methylsulfanyl-butyrylamino}-acetylamino)-3-(1H-indol-3-yl)-propionylamino]-4-methylsulfanyl-butyrylamino}-N-(1-carbamoyl-2-phenyl-ethyl)-succinamic acid | Asp-Tyr-Met-Gly-Trp-Met-Asp-Phe-NH2 | CHEMBL216166 | Desulfated CCK8 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C49H62N10O13S2 |
|---|
| Mol. Mass. | 1063.206 |
|---|
| SMILES | CSCC[C@H](NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(N)=O |
|---|
| Structure | 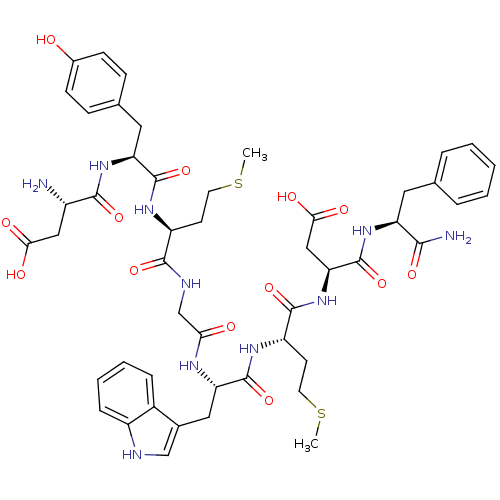
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Van Dijk, A; Richards, JG; Trzeciak, A; Gillessen, D; Möhler, H Cholecystokinin receptors: biochemical demonstration and autoradiographical localization in rat brain and pancreas using [3H] cholecystokinin8 as radioligand. J Neurosci4:1021-33 (1984) [PubMed]
Van Dijk, A; Richards, JG; Trzeciak, A; Gillessen, D; Möhler, H Cholecystokinin receptors: biochemical demonstration and autoradiographical localization in rat brain and pancreas using [3H] cholecystokinin8 as radioligand. J Neurosci4:1021-33 (1984) [PubMed]