| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histamine H1 receptor |
|---|
| Ligand | BDBM50035398 |
|---|
| Substrate/Competitor | n/a |
|---|
| Ki | >10000±n/a nM |
|---|
| Comments | PDSP_222 |
|---|
| Citation |  Arneric, SP; Sullivan, JP; Briggs, CA; Donnelly-Roberts, D; Anderson, DJ; Raszkiewicz, JL; Hughes, ML; Cadman, ED; Adams, P; Garvey, DS (S)-3-methyl-5-(1-methyl-2-pyrrolidinyl) isoxazole (ABT 418): a novel cholinergic ligand with cognition-enhancing and anxiolytic activities: I. In vitro characterization. J Pharmacol Exp Ther270:310-8 (1994) [PubMed] Arneric, SP; Sullivan, JP; Briggs, CA; Donnelly-Roberts, D; Anderson, DJ; Raszkiewicz, JL; Hughes, ML; Cadman, ED; Adams, P; Garvey, DS (S)-3-methyl-5-(1-methyl-2-pyrrolidinyl) isoxazole (ABT 418): a novel cholinergic ligand with cognition-enhancing and anxiolytic activities: I. In vitro characterization. J Pharmacol Exp Ther270:310-8 (1994) [PubMed] |
|---|
| More Info.: | Get all data from this article |
|---|
| |
| Histamine H1 receptor |
|---|
| Name: | Histamine H1 receptor |
|---|
| Synonyms: | H1R | HH1R | HISTAMINE H1 | HRH1 | HRH1_HUMAN |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 55808.72 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Cell pellets from SK-N-MC cells transfected with human H1 receptor were used in binding assay. |
|---|
| Residue: | 487 |
|---|
| Sequence: | MSLPNSSCLLEDKMCEGNKTTMASPQLMPLVVVLSTICLVTVGLNLLVLYAVRSERKLHT
VGNLYIVSLSVADLIVGAVVMPMNILYLLMSKWSLGRPLCLFWLSMDYVASTASIFSVFI
LCIDRYRSVQQPLRYLKYRTKTRASATILGAWFLSFLWVIPILGWNHFMQQTSVRREDKC
ETDFYDVTWFKVMTAIINFYLPTLLMLWFYAKIYKAVRQHCQHRELINRSLPSFSEIKLR
PENPKGDAKKPGKESPWEVLKRKPKDAGGGSVLKSPSQTPKEMKSPVVFSQEDDREVDKL
YCFPLDIVHMQAAAEGSSRDYVAVNRSHGQLKTDEQGLNTHGASEISEDQMLGDSQSFSR
TDSDTTTETAPGKGKLRSGSNTGLDYIKFTWKRLRSHSRQYVSGLHMNRERKAAKQLGFI
MAAFILCWIPYFIFFMVIAFCKNCCNEHLHMFTIWLGYINSTLNPLIYPLCNENFKKTFK
RILHIRS
|
|
|
|---|
| BDBM50035398 |
|---|
| n/a |
|---|
| Name | BDBM50035398 |
|---|
| Synonyms: | (S)-1-Methyl-2-(3-methyl-isoxazol-5-yl)-pyrrolidinium | (S)-3-methyl-5-(1-methylpyrrolidin-2-yl)isoxazole | 3-Methyl-5-((S)-1-methyl-pyrrolidin-2-yl)-isoxazole | ABT-418 | CHEMBL274525 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C9H14N2O |
|---|
| Mol. Mass. | 166.2203 |
|---|
| SMILES | CN1CCC[C@H]1c1cc(C)no1 |r| |
|---|
| Structure | 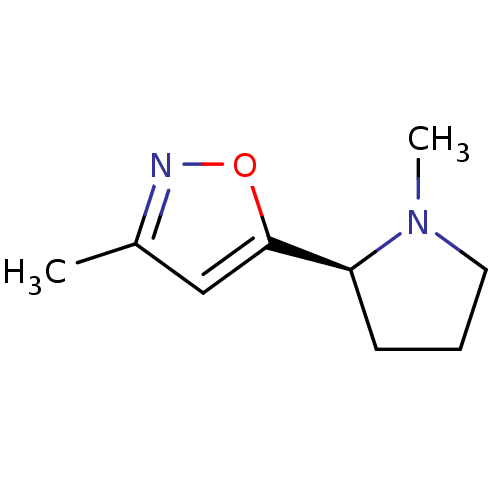
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Arneric, SP; Sullivan, JP; Briggs, CA; Donnelly-Roberts, D; Anderson, DJ; Raszkiewicz, JL; Hughes, ML; Cadman, ED; Adams, P; Garvey, DS (S)-3-methyl-5-(1-methyl-2-pyrrolidinyl) isoxazole (ABT 418): a novel cholinergic ligand with cognition-enhancing and anxiolytic activities: I. In vitro characterization. J Pharmacol Exp Ther270:310-8 (1994) [PubMed]
Arneric, SP; Sullivan, JP; Briggs, CA; Donnelly-Roberts, D; Anderson, DJ; Raszkiewicz, JL; Hughes, ML; Cadman, ED; Adams, P; Garvey, DS (S)-3-methyl-5-(1-methyl-2-pyrrolidinyl) isoxazole (ABT 418): a novel cholinergic ligand with cognition-enhancing and anxiolytic activities: I. In vitro characterization. J Pharmacol Exp Ther270:310-8 (1994) [PubMed]