| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histone deacetylase 6 |
|---|
| Ligand | BDBM19410 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1670960 (CHEMBL4020989) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Mehndiratta, S; Wang, RS; Huang, HL; Su, CJ; Hsu, CM; Wu, YW; Pan, SL; Liou, JP 4-Indolyl-N-hydroxyphenylacrylamides as potent HDAC class I and IIB inhibitors in�vitro and in�vivo. Eur J Med Chem134:13-23 (2017) [PubMed] Article Mehndiratta, S; Wang, RS; Huang, HL; Su, CJ; Hsu, CM; Wu, YW; Pan, SL; Liou, JP 4-Indolyl-N-hydroxyphenylacrylamides as potent HDAC class I and IIB inhibitors in�vitro and in�vivo. Eur J Med Chem134:13-23 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histone deacetylase 6 |
|---|
| Name: | Histone deacetylase 6 |
|---|
| Synonyms: | Cereblon/Histone deacetylase 6 | HD6 | HDAC6 | HDAC6_HUMAN | Histone deacetylase 6 (HDAC6) | Human HDAC6 | KIAA0901 | ORF Names:JM21 |
|---|
| Type: | Chromatin regulator; hydrolase; repressor |
|---|
| Mol. Mass.: | 131381.51 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q9UBN7 |
|---|
| Residue: | 1215 |
|---|
| Sequence: | MTSTGQDSTTTRQRRSRQNPQSPPQDSSVTSKRNIKKGAVPRSIPNLAEVKKKGKMKKLG
QAMEEDLIVGLQGMDLNLEAEALAGTGLVLDEQLNEFHCLWDDSFPEGPERLHAIKEQLI
QEGLLDRCVSFQARFAEKEELMLVHSLEYIDLMETTQYMNEGELRVLADTYDSVYLHPNS
YSCACLASGSVLRLVDAVLGAEIRNGMAIIRPPGHHAQHSLMDGYCMFNHVAVAARYAQQ
KHRIRRVLIVDWDVHHGQGTQFTFDQDPSVLYFSIHRYEQGRFWPHLKASNWSTTGFGQG
QGYTINVPWNQVGMRDADYIAAFLHVLLPVALEFQPQLVLVAAGFDALQGDPKGEMAATP
AGFAQLTHLLMGLAGGKLILSLEGGYNLRALAEGVSASLHTLLGDPCPMLESPGAPCRSA
QASVSCALEALEPFWEVLVRSTETVERDNMEEDNVEESEEEGPWEPPVLPILTWPVLQSR
TGLVYDQNMMNHCNLWDSHHPEVPQRILRIMCRLEELGLAGRCLTLTPRPATEAELLTCH
SAEYVGHLRATEKMKTRELHRESSNFDSIYICPSTFACAQLATGAACRLVEAVLSGEVLN
GAAVVRPPGHHAEQDAACGFCFFNSVAVAARHAQTISGHALRILIVDWDVHHGNGTQHMF
EDDPSVLYVSLHRYDHGTFFPMGDEGASSQIGRAAGTGFTVNVAWNGPRMGDADYLAAWH
RLVLPIAYEFNPELVLVSAGFDAARGDPLGGCQVSPEGYAHLTHLLMGLASGRIILILEG
GYNLTSISESMAACTRSLLGDPPPLLTLPRPPLSGALASITETIQVHRRYWRSLRVMKVE
DREGPSSSKLVTKKAPQPAKPRLAERMTTREKKVLEAGMGKVTSASFGEESTPGQTNSET
AVVALTQDQPSEAATGGATLAQTISEAAIGGAMLGQTTSEEAVGGATPDQTTSEETVGGA
ILDQTTSEDAVGGATLGQTTSEEAVGGATLAQTTSEAAMEGATLDQTTSEEAPGGTELIQ
TPLASSTDHQTPPTSPVQGTTPQISPSTLIGSLRTLELGSESQGASESQAPGEENLLGEA
AGGQDMADSMLMQGSRGLTDQAIFYAVTPLPWCPHLVAVCPIPAAGLDVTQPCGDCGTIQ
ENWVCLSCYQVYCGRYINGHMLQHHGNSGHPLVLSYIDLSAWCYYCQAYVHHQALLDVKN
IAHQNKFGEDMPHPH
|
|
|
|---|
| BDBM19410 |
|---|
| n/a |
|---|
| Name | BDBM19410 |
|---|
| Synonyms: | CHEMBL27759 | MS-275 | US11377423, MS-275 | US11672788, Compound Entinostat | US9265734, MS-275 | US9796664, Compound MS-275 | benzamide-type inhibitor, 3 | pyridin-3-ylmethyl N-({4-[(2-aminophenyl)carbamoyl]phenyl}methyl)carbamate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H20N4O3 |
|---|
| Mol. Mass. | 376.4085 |
|---|
| SMILES | Nc1ccccc1NC(=O)c1ccc(CNC(=O)OCc2cccnc2)cc1 |
|---|
| Structure | 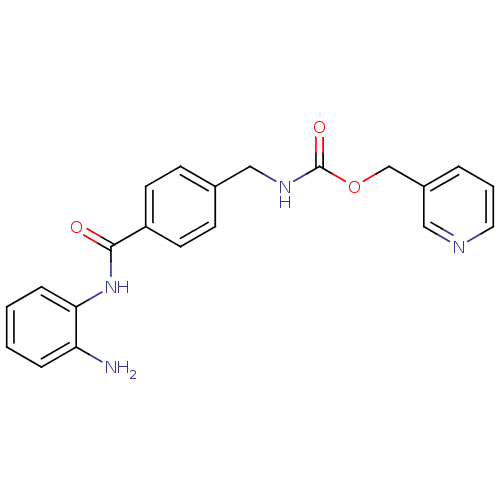
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Mehndiratta, S; Wang, RS; Huang, HL; Su, CJ; Hsu, CM; Wu, YW; Pan, SL; Liou, JP 4-Indolyl-N-hydroxyphenylacrylamides as potent HDAC class I and IIB inhibitors in�vitro and in�vivo. Eur J Med Chem134:13-23 (2017) [PubMed] Article
Mehndiratta, S; Wang, RS; Huang, HL; Su, CJ; Hsu, CM; Wu, YW; Pan, SL; Liou, JP 4-Indolyl-N-hydroxyphenylacrylamides as potent HDAC class I and IIB inhibitors in�vitro and in�vivo. Eur J Med Chem134:13-23 (2017) [PubMed] Article