| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2C |
|---|
| Ligand | BDBM50081704 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_1874 |
|---|
| IC50 | 33±n/a nM |
|---|
| Citation |  Macor, JE; Blake, J; Fox, CB; Johnson, C; Koe, BK; Lebel, LA; Morrone, JM; Ryan, K; Schmidt, AW; Schulz, DW Synthesis and serotonergic pharmacology of the enantiomers of 3-[(N-methylpyrrolidin-2-yl)methyl]-5-methoxy-1H-indole: discovery of stereogenic differentiation in the aminoethyl side chain of the neurotransmitter serotonin. J Med Chem35:4503-5 (1992) [PubMed] Macor, JE; Blake, J; Fox, CB; Johnson, C; Koe, BK; Lebel, LA; Morrone, JM; Ryan, K; Schmidt, AW; Schulz, DW Synthesis and serotonergic pharmacology of the enantiomers of 3-[(N-methylpyrrolidin-2-yl)methyl]-5-methoxy-1H-indole: discovery of stereogenic differentiation in the aminoethyl side chain of the neurotransmitter serotonin. J Med Chem35:4503-5 (1992) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2C |
|---|
| Name: | 5-hydroxytryptamine receptor 2C |
|---|
| Synonyms: | 5-HT2C | 5-hydroxytryptamine receptor 2C | 5-hydroxytryptamine receptor 2C (5HT2c) | 5HT2C_RAT | 5ht1c | Htr1c | Htr2c | Serotonin (5-HT) receptor | Serotonin receptor 2a and 2c (5HT2A and 5HT2C) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 51935.10 |
|---|
| Organism: | Rattus norvegicus (Rat) |
|---|
| Description: | P08909 |
|---|
| Residue: | 460 |
|---|
| Sequence: | MVNLGNAVRSLLMHLIGLLVWQFDISISPVAAIVTDTFNSSDGGRLFQFPDGVQNWPALS
IVVIIIMTIGGNILVIMAVSMEKKLHNATNYFLMSLAIADMLVGLLVMPLSLLAILYDYV
WPLPRYLCPVWISLDVLFSTASIMHLCAISLDRYVAIRNPIEHSRFNSRTKAIMKIAIVW
AISIGVSVPIPVIGLRDESKVFVNNTTCVLNDPNFVLIGSFVAFFIPLTIMVITYFLTIY
VLRRQTLMLLRGHTEEELANMSLNFLNCCCKKNGGEEENAPNPNPDQKPRRKKKEKRPRG
TMQAINNEKKASKVLGIVFFVFLIMWCPFFITNILSVLCGKACNQKLMEKLLNVFVWIGY
VCSGINPLVYTLFNKIYRRAFSKYLRCDYKPDKKPPVRQIPRVAATALSGRELNVNIYRH
TNERVARKANDPEPGIEMQVENLELPVNPSNVVSERISSV
|
|
|
|---|
| BDBM50081704 |
|---|
| n/a |
|---|
| Name | BDBM50081704 |
|---|
| Synonyms: | 5-Methoxy-3-((R)-1-methyl-pyrrolidin-2-ylmethyl)-1H-indole | CHEMBL137485 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H20N2O |
|---|
| Mol. Mass. | 244.3321 |
|---|
| SMILES | COc1ccc2[nH]cc(C[C@H]3CCCN3C)c2c1 |
|---|
| Structure | 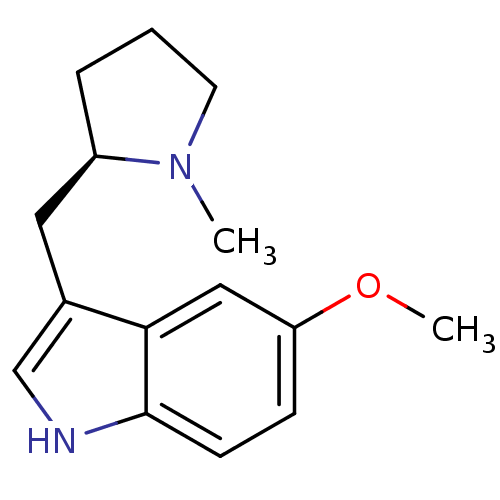
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Macor, JE; Blake, J; Fox, CB; Johnson, C; Koe, BK; Lebel, LA; Morrone, JM; Ryan, K; Schmidt, AW; Schulz, DW Synthesis and serotonergic pharmacology of the enantiomers of 3-[(N-methylpyrrolidin-2-yl)methyl]-5-methoxy-1H-indole: discovery of stereogenic differentiation in the aminoethyl side chain of the neurotransmitter serotonin. J Med Chem35:4503-5 (1992) [PubMed]
Macor, JE; Blake, J; Fox, CB; Johnson, C; Koe, BK; Lebel, LA; Morrone, JM; Ryan, K; Schmidt, AW; Schulz, DW Synthesis and serotonergic pharmacology of the enantiomers of 3-[(N-methylpyrrolidin-2-yl)methyl]-5-methoxy-1H-indole: discovery of stereogenic differentiation in the aminoethyl side chain of the neurotransmitter serotonin. J Med Chem35:4503-5 (1992) [PubMed]