| Reaction Details |
|---|
 | Report a problem with these data |
| Target | High affinity cAMP-specific 3',5'-cyclic phosphodiesterase 7A |
|---|
| Ligand | BDBM50246908 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1677116 (CHEMBL4027259) |
|---|
| IC50 | 3460±n/a nM |
|---|
| Citation |  Carzaniga, L; Amari, G; Rizzi, A; Capaldi, C; De Fanti, R; Ghidini, E; Villetti, G; Carnini, C; Moretto, N; Facchinetti, F; Caruso, P; Marchini, G; Battipaglia, L; Patacchini, R; Cenacchi, V; Volta, R; Amadei, F; Pappani, A; Capacchi, S; Bagnacani, V; Delcanale, M; Puccini, P; Catinella, S; Civelli, M; Armani, E Discovery and Optimization of Thiazolidinyl and Pyrrolidinyl Derivatives as Inhaled PDE4 Inhibitors for Respiratory Diseases. J Med Chem60:10026-10046 (2017) [PubMed] Article Carzaniga, L; Amari, G; Rizzi, A; Capaldi, C; De Fanti, R; Ghidini, E; Villetti, G; Carnini, C; Moretto, N; Facchinetti, F; Caruso, P; Marchini, G; Battipaglia, L; Patacchini, R; Cenacchi, V; Volta, R; Amadei, F; Pappani, A; Capacchi, S; Bagnacani, V; Delcanale, M; Puccini, P; Catinella, S; Civelli, M; Armani, E Discovery and Optimization of Thiazolidinyl and Pyrrolidinyl Derivatives as Inhaled PDE4 Inhibitors for Respiratory Diseases. J Med Chem60:10026-10046 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| High affinity cAMP-specific 3',5'-cyclic phosphodiesterase 7A |
|---|
| Name: | High affinity cAMP-specific 3',5'-cyclic phosphodiesterase 7A |
|---|
| Synonyms: | 3',5'-cyclic phosphodiesterase | PDE7A | PDE7A_HUMAN | Phosphodiesterase 7 | Phosphodiesterase 7 (PDE7) | Phosphodiesterase 7A | Phosphodiesterase 7A (PDE7A1) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55514.96 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q13946 |
|---|
| Residue: | 482 |
|---|
| Sequence: | MEVCYQLPVLPLDRPVPQHVLSRRGAISFSSSSALFGCPNPRQLSQRRGAISYDSSDQTA
LYIRMLGDVRVRSRAGFESERRGSHPYIDFRIFHSQSEIEVSVSARNIRRLLSFQRYLRS
SRFFRGTAVSNSLNILDDDYNGQAKCMLEKVGNWNFDIFLFDRLTNGNSLVSLTFHLFSL
HGLIEYFHLDMMKLRRFLVMIQEDYHSQNPYHNAVHAADVTQAMHCYLKEPKLANSVTPW
DILLSLIAAATHDLDHPGVNQPFLIKTNHYLATLYKNTSVLENHHWRSAVGLLRESGLFS
HLPLESRQQMETQIGALILATDISRQNEYLSLFRSHLDRGDLCLEDTRHRHLVLQMALKC
ADICNPCRTWELSKQWSEKVTEEFFHQGDIEKKYHLGVSPLCDRHTESIANIQIGFMTYL
VEPLFTEWARFSNTRLSQTMLGHVGLNKASWKGLQREQSSSEDTDAAFELNSQLLPQENR
LS
|
|
|
|---|
| BDBM50246908 |
|---|
| n/a |
|---|
| Name | BDBM50246908 |
|---|
| Synonyms: | CHEMBL4093768 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H33Cl2F2N3O8S |
|---|
| Mol. Mass. | 728.588 |
|---|
| SMILES | CN(C)C(=O)c1cccc(c1)S(=O)(=O)N1CCC[C@H]1C(=O)O[C@@H](Cc1c(Cl)c[n+]([O-])cc1Cl)c1ccc(OC(F)F)c(OCC2CC2)c1 |r| |
|---|
| Structure | 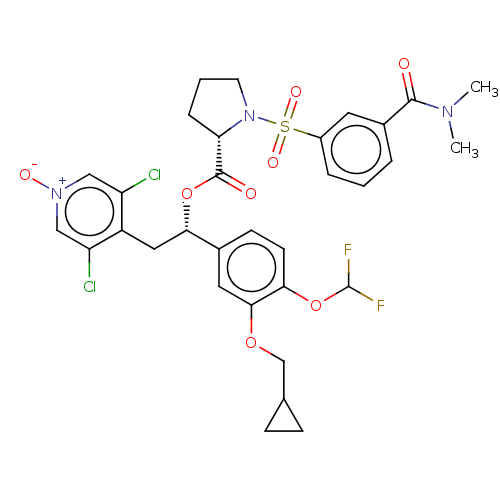
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Carzaniga, L; Amari, G; Rizzi, A; Capaldi, C; De Fanti, R; Ghidini, E; Villetti, G; Carnini, C; Moretto, N; Facchinetti, F; Caruso, P; Marchini, G; Battipaglia, L; Patacchini, R; Cenacchi, V; Volta, R; Amadei, F; Pappani, A; Capacchi, S; Bagnacani, V; Delcanale, M; Puccini, P; Catinella, S; Civelli, M; Armani, E Discovery and Optimization of Thiazolidinyl and Pyrrolidinyl Derivatives as Inhaled PDE4 Inhibitors for Respiratory Diseases. J Med Chem60:10026-10046 (2017) [PubMed] Article
Carzaniga, L; Amari, G; Rizzi, A; Capaldi, C; De Fanti, R; Ghidini, E; Villetti, G; Carnini, C; Moretto, N; Facchinetti, F; Caruso, P; Marchini, G; Battipaglia, L; Patacchini, R; Cenacchi, V; Volta, R; Amadei, F; Pappani, A; Capacchi, S; Bagnacani, V; Delcanale, M; Puccini, P; Catinella, S; Civelli, M; Armani, E Discovery and Optimization of Thiazolidinyl and Pyrrolidinyl Derivatives as Inhaled PDE4 Inhibitors for Respiratory Diseases. J Med Chem60:10026-10046 (2017) [PubMed] Article