| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2B |
|---|
| Ligand | BDBM50341448 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_1696905 |
|---|
| Ki | 20±n/a nM |
|---|
| Citation |  Procopiou, PA; Ford, AJ; Gore, PM; Hancock, AP; Hodgson, ST; Holmes, DS; Looker, BE; Vile, S; Clark, KL; Saunders, KA; Slack, RJ; Watts, CJ Identification of selective 8-(piperidin-4-yloxy)quinoline sulfone and sulfonamide histamine H Bioorg Med Chem Lett27:4914-4919 (2017) [PubMed] Article Procopiou, PA; Ford, AJ; Gore, PM; Hancock, AP; Hodgson, ST; Holmes, DS; Looker, BE; Vile, S; Clark, KL; Saunders, KA; Slack, RJ; Watts, CJ Identification of selective 8-(piperidin-4-yloxy)quinoline sulfone and sulfonamide histamine H Bioorg Med Chem Lett27:4914-4919 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2B |
|---|
| Name: | 5-hydroxytryptamine receptor 2B |
|---|
| Synonyms: | 5-HT-2B | 5-HT2B | 5-hydroxytryptamine (serotonin) receptor 2B [Homo sapiens] | 5-hydroxytryptamine receptor 2B (5-HT2B) | 5-hydroxytryptamine receptor 2C (5HT2C) | 5HT2B_HUMAN | HTR2B | Serotonin (5-HT3) receptor | Serotonin 2b (5-HT2b) receptor | Serotonin Receptor 2B |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 54312.47 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Receptor binding assays were performed using human clone stably expressed in CHO cells. |
|---|
| Residue: | 481 |
|---|
| Sequence: | MALSYRVSELQSTIPEHILQSTFVHVISSNWSGLQTESIPEEMKQIVEEQGNKLHWAALL
ILMVIIPTIGGNTLVILAVSLEKKLQYATNYFLMSLAVADLLVGLFVMPIALLTIMFEAM
WPLPLVLCPAWLFLDVLFSTASIMHLCAISVDRYIAIKKPIQANQYNSRATAFIKITVVW
LISIGIAIPVPIKGIETDVDNPNNITCVLTKERFGDFMLFGSLAAFFTPLAIMIVTYFLT
IHALQKKAYLVKNKPPQRLTWLTVSTVFQRDETPCSSPEKVAMLDGSRKDKALPNSGDET
LMRRTSTIGKKSVQTISNEQRASKVLGIVFFLFLLMWCPFFITNITLVLCDSCNQTTLQM
LLEIFVWIGYVSSGVNPLVYTLFNKTFRDAFGRYITCNYRATKSVKTLRKRSSKIYFRNP
MAENSKFFKKHGIRNGINPAMYQSPMRLRSSTIQSSSIILLDTLLLTENEGDKTEEQVSY
V
|
|
|
|---|
| BDBM50341448 |
|---|
| n/a |
|---|
| Name | BDBM50341448 |
|---|
| Synonyms: | 4-(4-Chloro-benzyl)-2-(1-methyl-azepan-4-yl)-2H-phthalazin-1-one | AZELASTINE | Astelin | Astepro | CHEMBL639 | Optilast | Optivar | Rhinolast | US20230414632, Compound II-a | rac-Azelastine |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H24ClN3O |
|---|
| Mol. Mass. | 381.898 |
|---|
| SMILES | CN1CCCC(CC1)n1nc(Cc2ccc(Cl)cc2)c2ccccc2c1=O |
|---|
| Structure | 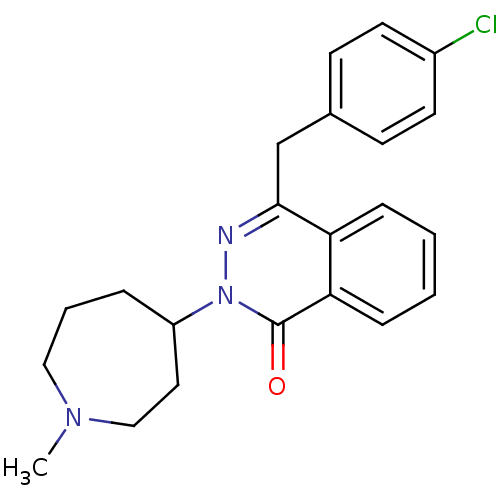
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Procopiou, PA; Ford, AJ; Gore, PM; Hancock, AP; Hodgson, ST; Holmes, DS; Looker, BE; Vile, S; Clark, KL; Saunders, KA; Slack, RJ; Watts, CJ Identification of selective 8-(piperidin-4-yloxy)quinoline sulfone and sulfonamide histamine H Bioorg Med Chem Lett27:4914-4919 (2017) [PubMed] Article
Procopiou, PA; Ford, AJ; Gore, PM; Hancock, AP; Hodgson, ST; Holmes, DS; Looker, BE; Vile, S; Clark, KL; Saunders, KA; Slack, RJ; Watts, CJ Identification of selective 8-(piperidin-4-yloxy)quinoline sulfone and sulfonamide histamine H Bioorg Med Chem Lett27:4914-4919 (2017) [PubMed] Article