| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase CSK |
|---|
| Ligand | BDBM267935 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1706721 (CHEMBL4057954) |
|---|
| IC50 | 626±n/a nM |
|---|
| Citation |  Boga, SB; Alhassan, AB; Liu, J; Guiadeen, D; Krikorian, A; Gao, X; Wang, J; Yu, Y; Anand, R; Liu, S; Yang, C; Wu, H; Cai, J; Zhu, H; Desai, J; Maloney, K; Gao, YD; Fischmann, TO; Presland, J; Mansueto, M; Xu, Z; Leccese, E; Knemeyer, I; Garlisi, CG; Bays, N; Stivers, P; Brandish, PE; Hicks, A; Cooper, A; Kim, RM; Kozlowski, JA Discovery of 3-morpholino-imidazole[1,5-a]pyrazine BTK inhibitors for rheumatoid arthritis. Bioorg Med Chem Lett27:3939-3943 (2017) [PubMed] Article Boga, SB; Alhassan, AB; Liu, J; Guiadeen, D; Krikorian, A; Gao, X; Wang, J; Yu, Y; Anand, R; Liu, S; Yang, C; Wu, H; Cai, J; Zhu, H; Desai, J; Maloney, K; Gao, YD; Fischmann, TO; Presland, J; Mansueto, M; Xu, Z; Leccese, E; Knemeyer, I; Garlisi, CG; Bays, N; Stivers, P; Brandish, PE; Hicks, A; Cooper, A; Kim, RM; Kozlowski, JA Discovery of 3-morpholino-imidazole[1,5-a]pyrazine BTK inhibitors for rheumatoid arthritis. Bioorg Med Chem Lett27:3939-3943 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase CSK |
|---|
| Name: | Tyrosine-protein kinase CSK |
|---|
| Synonyms: | C-SRC kinase | C-src tyrosine kinase | CSK | CSK_HUMAN | Protein-tyrosine kinase CYL | Tyrosine Kinase CSK |
|---|
| Type: | Tyrosine-protein kinase |
|---|
| Mol. Mass.: | 50707.30 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P41240 |
|---|
| Residue: | 450 |
|---|
| Sequence: | MSAIQAAWPSGTECIAKYNFHGTAEQDLPFCKGDVLTIVAVTKDPNWYKAKNKVGREGII
PANYVQKREGVKAGTKLSLMPWFHGKITREQAERLLYPPETGLFLVRESTNYPGDYTLCV
SCDGKVEHYRIMYHASKLSIDEEVYFENLMQLVEHYTSDADGLCTRLIKPKVMEGTVAAQ
DEFYRSGWALNMKELKLLQTIGKGEFGDVMLGDYRGNKVAVKCIKNDATAQAFLAEASVM
TQLRHSNLVQLLGVIVEEKGGLYIVTEYMAKGSLVDYLRSRGRSVLGGDCLLKFSLDVCE
AMEYLEGNNFVHRDLAARNVLVSEDNVAKVSDFGLTKEASSTQDTGKLPVKWTAPEALRE
KKFSTKSDVWSFGILLWEIYSFGRVPYPRIPLKDVVPRVEKGYKMDAPDGCPPAVYEVMK
NCWHLDAAMRPSFLQLREQLEHIKTHELHL
|
|
|
|---|
| BDBM267935 |
|---|
| n/a |
|---|
| Name | BDBM267935 |
|---|
| Synonyms: | 4-{8-amino-3-[(2R)-4-oxetan-3-ylmorpholin-2-yl]imidazo[1,5-a]pyrazin-1-yl}-N-[4-(trifluoromethyl)pyridin-2-yl]benzamide | US9718828, Example, 551 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H23F4N7O4 |
|---|
| Mol. Mass. | 573.4989 |
|---|
| SMILES | COCC(=O)N1CCO[C@H](C1)c1nc(-c2ccc(cc2F)C(=O)Nc2cc(ccn2)C(F)(F)F)c2c(N)nccn12 |r| |
|---|
| Structure | 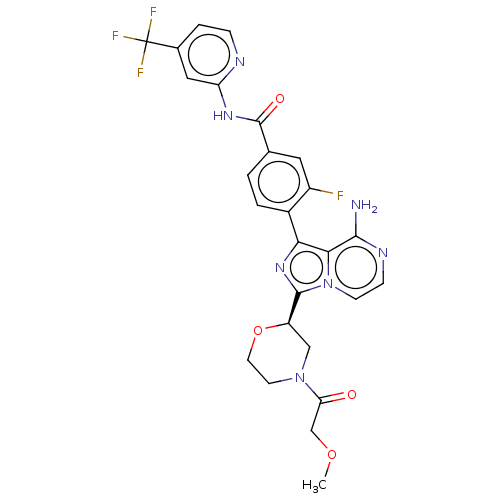
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Boga, SB; Alhassan, AB; Liu, J; Guiadeen, D; Krikorian, A; Gao, X; Wang, J; Yu, Y; Anand, R; Liu, S; Yang, C; Wu, H; Cai, J; Zhu, H; Desai, J; Maloney, K; Gao, YD; Fischmann, TO; Presland, J; Mansueto, M; Xu, Z; Leccese, E; Knemeyer, I; Garlisi, CG; Bays, N; Stivers, P; Brandish, PE; Hicks, A; Cooper, A; Kim, RM; Kozlowski, JA Discovery of 3-morpholino-imidazole[1,5-a]pyrazine BTK inhibitors for rheumatoid arthritis. Bioorg Med Chem Lett27:3939-3943 (2017) [PubMed] Article
Boga, SB; Alhassan, AB; Liu, J; Guiadeen, D; Krikorian, A; Gao, X; Wang, J; Yu, Y; Anand, R; Liu, S; Yang, C; Wu, H; Cai, J; Zhu, H; Desai, J; Maloney, K; Gao, YD; Fischmann, TO; Presland, J; Mansueto, M; Xu, Z; Leccese, E; Knemeyer, I; Garlisi, CG; Bays, N; Stivers, P; Brandish, PE; Hicks, A; Cooper, A; Kim, RM; Kozlowski, JA Discovery of 3-morpholino-imidazole[1,5-a]pyrazine BTK inhibitors for rheumatoid arthritis. Bioorg Med Chem Lett27:3939-3943 (2017) [PubMed] Article