| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Proprotein convertase subtilisin/kexin type 9 |
|---|
| Ligand | BDBM50014066 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_206137 (CHEMBL814094) |
|---|
| KOFF | 3500 s-1 |
|---|
| Citation |  Tous, G; Bush, A; Tous, A; Jordan, F O'-(epoxyalkyl)tyrosines and (epoxyalkyl)phenylalanine as irreversible inactivators of serine proteases: synthesis and inhibition mechanism. J Med Chem33:1620-34 (1990) [PubMed] Tous, G; Bush, A; Tous, A; Jordan, F O'-(epoxyalkyl)tyrosines and (epoxyalkyl)phenylalanine as irreversible inactivators of serine proteases: synthesis and inhibition mechanism. J Med Chem33:1620-34 (1990) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Proprotein convertase subtilisin/kexin type 9 |
|---|
| Name: | Proprotein convertase subtilisin/kexin type 9 |
|---|
| Synonyms: | NARC-1 | NARC1 | Neural apoptosis-regulated convertase 1 | PC9 | PCSK9 | PCSK9_HUMAN | Proprotein convertase 9 | Proprotein convertase subtilisin/kexin type 9 | Proprotein convertase subtilisin/kexin type 9 (PCSK9) | Subtilisin/kexin type 9 | Subtilisin/kexin-like protease PC9 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 74286.93 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q8NBP7 |
|---|
| Residue: | 692 |
|---|
| Sequence: | MGTVSSRRSWWPLPLLLLLLLLLGPAGARAQEDEDGDYEELVLALRSEEDGLAEAPEHGT
TATFHRCAKDPWRLPGTYVVVLKEETHLSQSERTARRLQAQAARRGYLTKILHVFHGLLP
GFLVKMSGDLLELALKLPHVDYIEEDSSVFAQSIPWNLERITPPRYRADEYQPPDGGSLV
EVYLLDTSIQSDHREIEGRVMVTDFENVPEEDGTRFHRQASKCDSHGTHLAGVVSGRDAG
VAKGASMRSLRVLNCQGKGTVSGTLIGLEFIRKSQLVQPVGPLVVLLPLAGGYSRVLNAA
CQRLARAGVVLVTAAGNFRDDACLYSPASAPEVITVGATNAQDQPVTLGTLGTNFGRCVD
LFAPGEDIIGASSDCSTCFVSQSGTSQAAAHVAGIAAMMLSAEPELTLAELRQRLIHFSA
KDVINEAWFPEDQRVLTPNLVAALPPSTHGAGWQLFCRTVWSAHSGPTRMATAVARCAPD
EELLSCSSFSRSGKRRGERMEAQGGKLVCRAHNAFGGEGVYAIARCCLLPQANCSVHTAP
PAEASMGTRVHCHQQGHVLTGCSSHWEVEDLGTHKPPVLRPRGQPNQCVGHREASIHASC
CHAPGLECKVKEHGIPAPQEQVTVACEEGWTLTGCSALPGTSHVLGAYAVDNTCVVRSRD
VSTTGSTSEGAVTAVAICCRSRHLAQASQELQ
|
|
|
|---|
| BDBM50014066 |
|---|
| n/a |
|---|
| Name | BDBM50014066 |
|---|
| Synonyms: | 2-Acetylamino-3-(4-oxiranylmethoxy-phenyl)-propionic acid ethyl ester | CHEMBL42526 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H21NO5 |
|---|
| Mol. Mass. | 307.3416 |
|---|
| SMILES | CCOC(=O)[C@H](Cc1ccc(OCC2CO2)cc1)NC(C)=O |
|---|
| Structure | 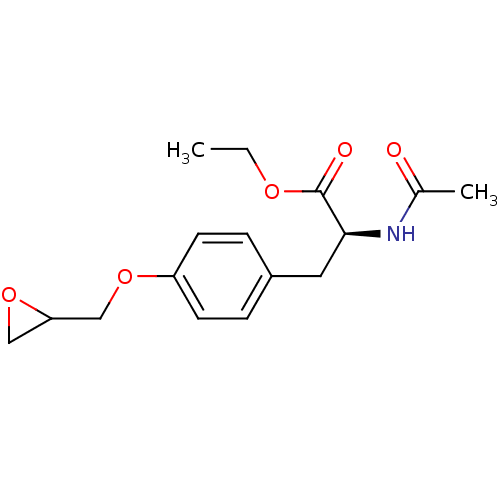
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tous, G; Bush, A; Tous, A; Jordan, F O'-(epoxyalkyl)tyrosines and (epoxyalkyl)phenylalanine as irreversible inactivators of serine proteases: synthesis and inhibition mechanism. J Med Chem33:1620-34 (1990) [PubMed]
Tous, G; Bush, A; Tous, A; Jordan, F O'-(epoxyalkyl)tyrosines and (epoxyalkyl)phenylalanine as irreversible inactivators of serine proteases: synthesis and inhibition mechanism. J Med Chem33:1620-34 (1990) [PubMed]