| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Reverse transcriptase/RNaseH |
|---|
| Ligand | BDBM50407560 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_195844 (CHEMBL799143) |
|---|
| IC50 | 12000±n/a nM |
|---|
| Citation |  Krawczyk, SH; Nassiri, MR; Kucera, LS; Kern, ER; Ptak, RG; Wotring, LL; Drach, JC; Townsend, LB Synthesis and antiproliferative and antiviral activity of 2'-deoxy-2'-fluoroarabinofuranosyl analogs of the nucleoside antibiotics toyocamycin and sangivamycin. J Med Chem38:4106-14 (1995) [PubMed] Krawczyk, SH; Nassiri, MR; Kucera, LS; Kern, ER; Ptak, RG; Wotring, LL; Drach, JC; Townsend, LB Synthesis and antiproliferative and antiviral activity of 2'-deoxy-2'-fluoroarabinofuranosyl analogs of the nucleoside antibiotics toyocamycin and sangivamycin. J Med Chem38:4106-14 (1995) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Reverse transcriptase/RNaseH |
|---|
| Name: | Reverse transcriptase/RNaseH |
|---|
| Synonyms: | HIV-1 Reverse Transcriptase RNase H | Human immunodeficiency virus type 1 reverse transcriptase | Reverse transcriptase/RNaseH |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 65229.15 |
|---|
| Organism: | Human immunodeficiency virus 1 |
|---|
| Description: | ChEMBL_1473730 |
|---|
| Residue: | 566 |
|---|
| Sequence: | PISPIETVPVKLKPGMDGPKVKQWPLTEEKIKALVEICTEMEKEGKISKIGPENPYNTPV
FAIKKKDSTKWRKLVDFRELNKRTQDFWEVQLGIPHPAGLKKRKSVTVLDVGDAYFSVPL
DEDFRKYTAFTIPSINNETPGIRYQYNVLPQGWKGSPAIFQSSMTKILEPFRKQNPDIVI
YQYMDDLYVGSDLEIGQHRTKIEELRQHLLRWGLTTPDKKHQKEPPFLWMGYELHPDKWT
VQPIVLPEKDSWTVNDIQKLVGKLNWASQIYPGIRVRQLCKLLRGTKALTEVIPLTEEAE
LELAENREILKEPVHGVYYDPSKDLIAEIQKQGQGQWTYQIYQEPFKNLRTGKYARMRGA
HTNDVKQLTEAVQKITTESIVIWGKTPKFKLPIQKETWETWWTEYWQATWIPEWEFVNTP
PLVKLWYQLEKEPIVGAETFYVDGAANRETKLGKAGYVTNRGRQKVVTLTDTTNQKTELQ
AIYLALQDSGLEVNIVTDSQYALGIIQAQPDQSESELVNQIIEQLIKKEKVYLAWVPAHK
GIGGNEQVDKLVSAGIRKVLFLDGID
|
|
|
|---|
| BDBM50407560 |
|---|
| n/a |
|---|
| Name | BDBM50407560 |
|---|
| Synonyms: | CHEMBL473686 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C12H12FN5O3 |
|---|
| Mol. Mass. | 293.2538 |
|---|
| SMILES | Nc1ncnc2n(cc(C#N)c12)[C@@H]1O[C@H](CO)[C@@H](O)[C@@H]1F |r| |
|---|
| Structure | 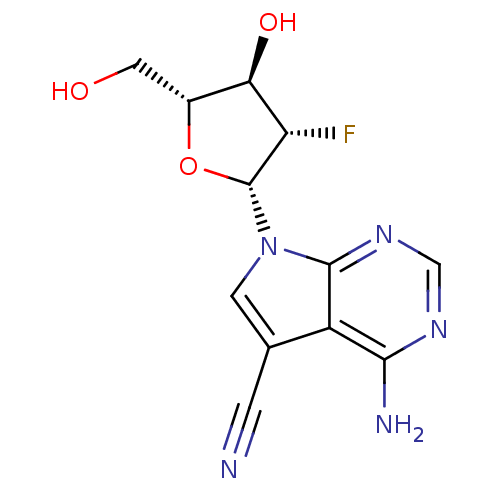
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Krawczyk, SH; Nassiri, MR; Kucera, LS; Kern, ER; Ptak, RG; Wotring, LL; Drach, JC; Townsend, LB Synthesis and antiproliferative and antiviral activity of 2'-deoxy-2'-fluoroarabinofuranosyl analogs of the nucleoside antibiotics toyocamycin and sangivamycin. J Med Chem38:4106-14 (1995) [PubMed]
Krawczyk, SH; Nassiri, MR; Kucera, LS; Kern, ER; Ptak, RG; Wotring, LL; Drach, JC; Townsend, LB Synthesis and antiproliferative and antiviral activity of 2'-deoxy-2'-fluoroarabinofuranosyl analogs of the nucleoside antibiotics toyocamycin and sangivamycin. J Med Chem38:4106-14 (1995) [PubMed]