| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1D |
|---|
| Ligand | BDBM50005835 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1973 (CHEMBL617578) |
|---|
| EC50 | 97±n/a nM |
|---|
| Citation |  Macor, JE; Blank, DH; Fox, CB; Lebel, LA; Newman, ME; Post, RJ; Ryan, K; Schmidt, AW; Schulz, DW; Koe, BK 5-[(3-nitropyrid-2-yl)amino]indoles: novel serotonin agonists with selectivity for the 5-HT1D receptor. Variation of the C3 substituent on the indole template leads to increased 5-HT1D receptor selectivity. J Med Chem37:2509-12 (1994) [PubMed] Macor, JE; Blank, DH; Fox, CB; Lebel, LA; Newman, ME; Post, RJ; Ryan, K; Schmidt, AW; Schulz, DW; Koe, BK 5-[(3-nitropyrid-2-yl)amino]indoles: novel serotonin agonists with selectivity for the 5-HT1D receptor. Variation of the C3 substituent on the indole template leads to increased 5-HT1D receptor selectivity. J Med Chem37:2509-12 (1994) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 1D |
|---|
| Name: | 5-hydroxytryptamine receptor 1D |
|---|
| Synonyms: | 5-HT-1D | 5-HT1D | 5-hydroxytryptamine receptor 1D | 5HT1D_RAT | Htr1d | Serotonin 1 (5-HT1) receptor | Serotonin 1B/1D receptor | Serotonin receptor 1D |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 41551.75 |
|---|
| Organism: | RAT |
|---|
| Description: | 5-HT1D HTR1D RAT::P28565 |
|---|
| Residue: | 374 |
|---|
| Sequence: | MSLPNQSLEGLPQEASNRSLNATGAWDPEVLQALRISLVVVLSIITLATVLSNAFVLTTI
LLTKKLHTPANYLIGSLATTDLLVSILVMPISIAYTTTRTWNFGQILCDIWVSSDITCCT
ASILHLCVIALDRYWAITDALEYSKRRTAGHAAAMIAAVWAISICISIPPLFWRQATAHE
EMSDCLVNTSQISYTIYSTCGAFYIPSILLIILYGRIYVAARSRILNPPSLYGKRFTTAQ
LITGSAGSSLCSLNPSLHESHTHTVGSPLFFNQVKIKLADSILERKRISAARERKATKTL
GIILGAFIICWLPFFVVSLVLPICRDSCWIHPALFDFFTWLGYLNSLINPVIYTVFNEDF
RQAFQRVVHFRKAS
|
|
|
|---|
| BDBM50005835 |
|---|
| n/a |
|---|
| Name | BDBM50005835 |
|---|
| Synonyms: | (3-[2-(dimethylamino)ethyl]-1H-indol-5-yl)-N-methylmethanesulfonamide | 1-[3-(2-dimethylaminoethyl)-1H-indol-5-yl]-N-methyl-methanesulfonamide | 1-{3-[2-(dimethylamino)ethyl]-1H-indol-5-yl}-N-methylmethanesulfonamide | 3-(2-(dimethylamino)ethyl)-N-methyl-1H-indole-5-methanesulfonamide | 3-[2-(dimethylamino)ethyl]-N-methylindole-5-methanesulfonamide | CHEMBL128 | SUMATRIPTAN | Sumatran | Sumax |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H21N3O2S |
|---|
| Mol. Mass. | 295.4 |
|---|
| SMILES | CNS(=O)(=O)Cc1ccc2[nH]cc(CCN(C)C)c2c1 |
|---|
| Structure | 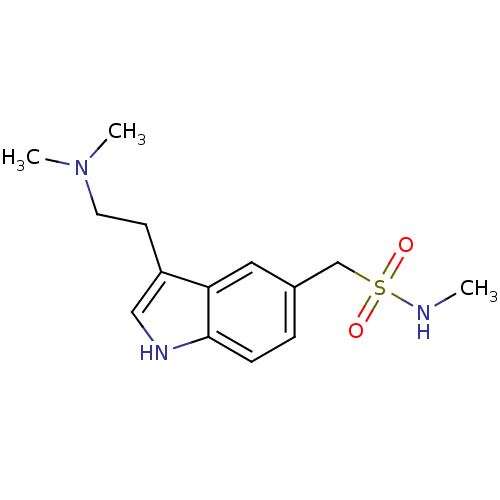
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Macor, JE; Blank, DH; Fox, CB; Lebel, LA; Newman, ME; Post, RJ; Ryan, K; Schmidt, AW; Schulz, DW; Koe, BK 5-[(3-nitropyrid-2-yl)amino]indoles: novel serotonin agonists with selectivity for the 5-HT1D receptor. Variation of the C3 substituent on the indole template leads to increased 5-HT1D receptor selectivity. J Med Chem37:2509-12 (1994) [PubMed]
Macor, JE; Blank, DH; Fox, CB; Lebel, LA; Newman, ME; Post, RJ; Ryan, K; Schmidt, AW; Schulz, DW; Koe, BK 5-[(3-nitropyrid-2-yl)amino]indoles: novel serotonin agonists with selectivity for the 5-HT1D receptor. Variation of the C3 substituent on the indole template leads to increased 5-HT1D receptor selectivity. J Med Chem37:2509-12 (1994) [PubMed]