| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-1A adrenergic receptor |
|---|
| Ligand | BDBM69602 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_33586 (CHEMBL647137) |
|---|
| Ki | 0.62±n/a nM |
|---|
| Citation |  Quaglia, W; Pigini, M; Tayebati, SK; Piergentili, A; Giannella, M; Leonardi, A; Taddei, C; Melchiorre, C Synthesis, absolute configuration, and biological profile of the enantiomers of trans-[2-(2,6-dimethoxyphenoxy)ethyl] [(3-p-tolyl-2,3-dihydro-1,4-benzodioxin-2-yl)methyl]amine (mephendioxan), a potent competitive alpha 1A-adrenoreceptor antagonist. J Med Chem39:2253-8 (1996) [PubMed] Article Quaglia, W; Pigini, M; Tayebati, SK; Piergentili, A; Giannella, M; Leonardi, A; Taddei, C; Melchiorre, C Synthesis, absolute configuration, and biological profile of the enantiomers of trans-[2-(2,6-dimethoxyphenoxy)ethyl] [(3-p-tolyl-2,3-dihydro-1,4-benzodioxin-2-yl)methyl]amine (mephendioxan), a potent competitive alpha 1A-adrenoreceptor antagonist. J Med Chem39:2253-8 (1996) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-1A adrenergic receptor |
|---|
| Name: | Alpha-1A adrenergic receptor |
|---|
| Synonyms: | ADA1A_BOVIN | ADRA1A | ADRA1C | Alpha 1A-adrenoceptor | Alpha 1A-adrenoreceptor | Alpha-1C adrenergic receptor | Cerebral cortex alpha adrenergic receptor | adrenergic Alpha1 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 51492.08 |
|---|
| Organism: | CALF |
|---|
| Description: | adrenergic Alpha1 0 CALF |
|---|
| Residue: | 466 |
|---|
| Sequence: | MVFLSGNASDSSNCTHPPPPVNISKAILLGVILGGLILFGVLGNILVILSVACHRHLHSV
THYYIVNLAVADLLLTSTVLPFSAIFEILGYWAFGRVFCNVWAAVDVLCCTASIMGLCII
SIDRYIGVSYPLRYPTIVTQKRGLMALLCVWALSLVISIGPLFGWRQPAPEDETICQINE
EPGYVLFSALGSFYVPLTIILVMYCRVYVVAKRESRGLKSGLKTDKSDSEQVTLRIHRKN
AQVGGSGVTSAKNKTHFSVRLLKFSREKKAAKTLGIVVGCFVLCWLPFFLVMPIGSFFPD
FRPSETVFKIAFWLGYLNSCINPIIYPCSSQEFKKAFQNVLRIQCLRRKQSSKHTLGYTL
HAPSHVLEGQHKDLVRIPVGSAETFYKISKTDGVCEWKIFSSLPRGSARMAVARDPSACT
TARVRSKSFLQVCCCLGPSTPSHGENHQIPTIKIHTISLSENGEEV
|
|
|
|---|
| BDBM69602 |
|---|
| n/a |
|---|
| Name | BDBM69602 |
|---|
| Synonyms: | 2,3-dihydro-1,4-benzodioxin-3-ylmethyl-[2-(2,6-dimethoxyphenoxy)ethyl]amine;hydrochloride | CHEMBL25554 | MLS000859914 | N-(2,3-dihydro-1,4-benzodioxin-3-ylmethyl)-2-(2,6-dimethoxyphenoxy)ethanamine;hydrochloride | SMR000326775 | WB-4101 | WB-4101 hydrochloride | cid_11957505 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H23NO5 |
|---|
| Mol. Mass. | 345.3896 |
|---|
| SMILES | COc1cccc(OC)c1OCCNCC1COc2ccccc2O1 |
|---|
| Structure | 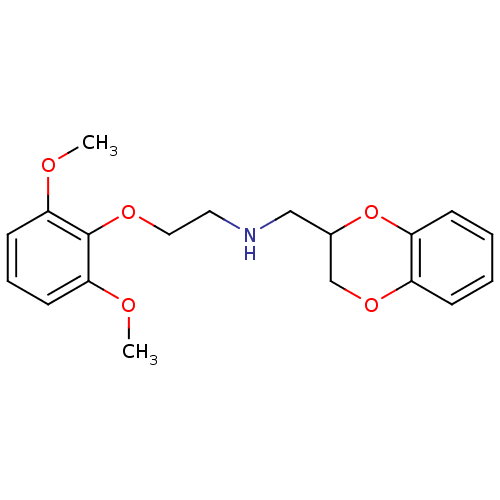
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Quaglia, W; Pigini, M; Tayebati, SK; Piergentili, A; Giannella, M; Leonardi, A; Taddei, C; Melchiorre, C Synthesis, absolute configuration, and biological profile of the enantiomers of trans-[2-(2,6-dimethoxyphenoxy)ethyl] [(3-p-tolyl-2,3-dihydro-1,4-benzodioxin-2-yl)methyl]amine (mephendioxan), a potent competitive alpha 1A-adrenoreceptor antagonist. J Med Chem39:2253-8 (1996) [PubMed] Article
Quaglia, W; Pigini, M; Tayebati, SK; Piergentili, A; Giannella, M; Leonardi, A; Taddei, C; Melchiorre, C Synthesis, absolute configuration, and biological profile of the enantiomers of trans-[2-(2,6-dimethoxyphenoxy)ethyl] [(3-p-tolyl-2,3-dihydro-1,4-benzodioxin-2-yl)methyl]amine (mephendioxan), a potent competitive alpha 1A-adrenoreceptor antagonist. J Med Chem39:2253-8 (1996) [PubMed] Article