| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor X |
|---|
| Ligand | BDBM50060747 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_48806 (CHEMBL662831) |
|---|
| Ki | 52000±n/a nM |
|---|
| Citation |  Feng, DM; Gardell, SJ; Lewis, SD; Bock, MG; Chen, Z; Freidinger, RM; Naylor-Olsen, AM; Ramjit, HG; Woltmann, R; Baskin, EP; Lynch, JJ; Lucas, R; Shafer, JA; Dancheck, KB; Chen, IW; Mao, SS; Krueger, JA; Hare, TR; Mulichak, AM; Vacca, JP Discovery of a novel, selective, and orally bioavailable class of thrombin inhibitors incorporating aminopyridyl moieties at the P1 position. J Med Chem40:3726-33 (1997) [PubMed] Article Feng, DM; Gardell, SJ; Lewis, SD; Bock, MG; Chen, Z; Freidinger, RM; Naylor-Olsen, AM; Ramjit, HG; Woltmann, R; Baskin, EP; Lynch, JJ; Lucas, R; Shafer, JA; Dancheck, KB; Chen, IW; Mao, SS; Krueger, JA; Hare, TR; Mulichak, AM; Vacca, JP Discovery of a novel, selective, and orally bioavailable class of thrombin inhibitors incorporating aminopyridyl moieties at the P1 position. J Med Chem40:3726-33 (1997) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor X |
|---|
| Name: | Coagulation factor X |
|---|
| Synonyms: | Activated coagulation factor X (FXa) | Activated factor Xa heavy chain | Coagulation factor X precursor | Coagulation factor Xa | F10 | FA10_HUMAN | Factor X heavy chain | Factor X light chain | Factor Xa | Stuart factor | Stuart-Prower factor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 54726.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 488 |
|---|
| Sequence: | MGRPLHLVLLSASLAGLLLLGESLFIRREQANNILARVTRANSFLEEMKKGHLERECMEE
TCSYEEAREVFEDSDKTNEFWNKYKDGDQCETSPCQNQGKCKDGLGEYTCTCLEGFEGKN
CELFTRKLCSLDNGDCDQFCHEEQNSVVCSCARGYTLADNGKACIPTGPYPCGKQTLERR
KRSVAQATSSSGEAPDSITWKPYDAADLDPTENPFDLLDFNQTQPERGDNNLTRIVGGQE
CKDGECPWQALLINEENEGFCGGTILSEFYILTAAHCLYQAKRFKVRVGDRNTEQEEGGE
AVHEVEVVIKHNRFTKETYDFDIAVLRLKTPITFRMNVAPACLPERDWAESTLMTQKTGI
VSGFGRTHEKGRQSTRLKMLEVPYVDRNSCKLSSSFIITQNMFCAGYDTKQEDACQGDSG
GPHVTRFKDTYFVTGIVSWGEGCARKGKYGIYTKVTAFLKWIDRSMKTRGLPKAKSHAPE
VITSSPLK
|
|
|
|---|
| BDBM50060747 |
|---|
| n/a |
|---|
| Name | BDBM50060747 |
|---|
| Synonyms: | (S)-1-[(R)-3-(3,4-Dichloro-phenyl)-2-phenylmethanesulfonylamino-propionyl]-pyrrolidine-2-carboxylic acid (6-amino-2,4-dimethyl-pyridin-3-ylmethyl)-amide | CHEMBL338788 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H33Cl2N5O4S |
|---|
| Mol. Mass. | 618.574 |
|---|
| SMILES | Cc1cc(N)nc(C)c1CNC(=O)[C@@H]1CCCN1C(=O)[C@@H](Cc1ccc(Cl)c(Cl)c1)NS(=O)(=O)Cc1ccccc1 |
|---|
| Structure | 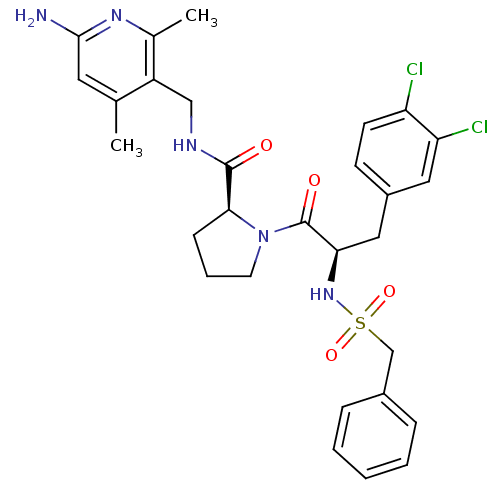
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Feng, DM; Gardell, SJ; Lewis, SD; Bock, MG; Chen, Z; Freidinger, RM; Naylor-Olsen, AM; Ramjit, HG; Woltmann, R; Baskin, EP; Lynch, JJ; Lucas, R; Shafer, JA; Dancheck, KB; Chen, IW; Mao, SS; Krueger, JA; Hare, TR; Mulichak, AM; Vacca, JP Discovery of a novel, selective, and orally bioavailable class of thrombin inhibitors incorporating aminopyridyl moieties at the P1 position. J Med Chem40:3726-33 (1997) [PubMed] Article
Feng, DM; Gardell, SJ; Lewis, SD; Bock, MG; Chen, Z; Freidinger, RM; Naylor-Olsen, AM; Ramjit, HG; Woltmann, R; Baskin, EP; Lynch, JJ; Lucas, R; Shafer, JA; Dancheck, KB; Chen, IW; Mao, SS; Krueger, JA; Hare, TR; Mulichak, AM; Vacca, JP Discovery of a novel, selective, and orally bioavailable class of thrombin inhibitors incorporating aminopyridyl moieties at the P1 position. J Med Chem40:3726-33 (1997) [PubMed] Article