| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Platelet-activating factor receptor |
|---|
| Ligand | BDBM50000714 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_158343 (CHEMBL768487) |
|---|
| IC50 | 94±n/a nM |
|---|
| Citation |  Cai, X; Scannell, RT; Yaeger, D; Hussoin, MS; Killian, DB; Qian, C; Eckman, J; Hwang, SB; Libertine-Garahan, L; Yeh, CG; Ip, SH; Shen, TY (+/-)-trans-2-[3-methoxy-4-(4-chlorophenylthioethoxy)-5-(N-methyl-N- hydroxyureidyl)methylphenyl]-5-(3,4, 5-trimethoxyphenyl)tetrahydrofuran (CMI-392), a potent dual 5-lipoxygenase inhibitor and platelet-activating factor receptor antagonist. J Med Chem41:1970-9 (1998) [PubMed] Article Cai, X; Scannell, RT; Yaeger, D; Hussoin, MS; Killian, DB; Qian, C; Eckman, J; Hwang, SB; Libertine-Garahan, L; Yeh, CG; Ip, SH; Shen, TY (+/-)-trans-2-[3-methoxy-4-(4-chlorophenylthioethoxy)-5-(N-methyl-N- hydroxyureidyl)methylphenyl]-5-(3,4, 5-trimethoxyphenyl)tetrahydrofuran (CMI-392), a potent dual 5-lipoxygenase inhibitor and platelet-activating factor receptor antagonist. J Med Chem41:1970-9 (1998) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Platelet-activating factor receptor |
|---|
| Name: | Platelet-activating factor receptor |
|---|
| Synonyms: | PAF Platelet activating factor | PAF-R | PAFR | PTAFR | PTAFR_HUMAN | Platelet activating factor receptor | Platelet-activating factor receptor |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 39219.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | PAF Platelet activating factor PTAFR HUMAN::P25105 |
|---|
| Residue: | 342 |
|---|
| Sequence: | MEPHDSSHMDSEFRYTLFPIVYSIIFVLGVIANGYVLWVFARLYPCKKFNEIKIFMVNLT
MADMLFLITLPLWIVYYQNQGNWILPKFLCNVAGCLFFINTYCSVAFLGVITYNRFQAVT
RPIKTAQANTRKRGISLSLVIWVAIVGAASYFLILDSTNTVPDSAGSGNVTRCFEHYEKG
SVPVLIIHIFIVFSFFLVFLIILFCNLVIIRTLLMQPVQQQRNAEVKRRALWMVCTVLAV
FIICFVPHHVVQLPWTLAELGFQDSKFHQAINDAHQVTLCLLSTNCVLDPVIYCFLTKKF
RKHLTEKFYSMRSSRKCSRATTDTVTEVVVPFNQIPGNSLKN
|
|
|
|---|
| BDBM50000714 |
|---|
| n/a |
|---|
| Name | BDBM50000714 |
|---|
| Synonyms: | 3-[4-(2-Chloro-phenyl)-9-methyl-6H-1-thia-5,7,8,9a-tetraaza-cyclopenta[e]azulen-2-yl]-1-morpholin-4-yl-propan-1-one | 3-[4-(2-Chloro-phenyl)-9-methyl-6H-1-thia-5,7,8,9a-tetraaza-cyclopenta[e]azulen-2-yl]-1-morpholin-4-yl-propan-1-one(WEB 2086) | 3-[4-(2-Chloro-phenyl)-9-methyl-6H-1-thia-5,7,8,9a-tetraaza-cyclopenta[e]azulen-2-yl]-1-morpholin-4-yl-propan-1-one(WEB2086) | Apafant | CHEMBL280164 | WEB2086 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H22ClN5O2S |
|---|
| Mol. Mass. | 455.96 |
|---|
| SMILES | Cc1nnc2CN=C(c3cc(CCC(=O)N4CCOCC4)sc3-n12)c1ccccc1Cl |c:6| |
|---|
| Structure | 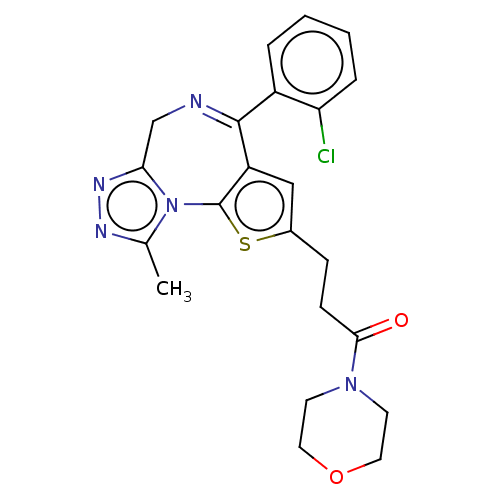
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Cai, X; Scannell, RT; Yaeger, D; Hussoin, MS; Killian, DB; Qian, C; Eckman, J; Hwang, SB; Libertine-Garahan, L; Yeh, CG; Ip, SH; Shen, TY (+/-)-trans-2-[3-methoxy-4-(4-chlorophenylthioethoxy)-5-(N-methyl-N- hydroxyureidyl)methylphenyl]-5-(3,4, 5-trimethoxyphenyl)tetrahydrofuran (CMI-392), a potent dual 5-lipoxygenase inhibitor and platelet-activating factor receptor antagonist. J Med Chem41:1970-9 (1998) [PubMed] Article
Cai, X; Scannell, RT; Yaeger, D; Hussoin, MS; Killian, DB; Qian, C; Eckman, J; Hwang, SB; Libertine-Garahan, L; Yeh, CG; Ip, SH; Shen, TY (+/-)-trans-2-[3-methoxy-4-(4-chlorophenylthioethoxy)-5-(N-methyl-N- hydroxyureidyl)methylphenyl]-5-(3,4, 5-trimethoxyphenyl)tetrahydrofuran (CMI-392), a potent dual 5-lipoxygenase inhibitor and platelet-activating factor receptor antagonist. J Med Chem41:1970-9 (1998) [PubMed] Article