| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Plasminogen |
|---|
| Ligand | BDBM50066336 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_155236 (CHEMBL764726) |
|---|
| Ki | 691000±n/a nM |
|---|
| Citation |  Tucker, TJ; Brady, SF; Lumma, WC; Lewis, SD; Gardell, SJ; Naylor-Olsen, AM; Yan, Y; Sisko, JT; Stauffer, KJ; Lucas, BJ; Lynch, JJ; Cook, JJ; Stranieri, MT; Holahan, MA; Lyle, EA; Baskin, EP; Chen, IW; Dancheck, KB; Krueger, JA; Cooper, CM; Vacca, JP Design and synthesis of a series of potent and orally bioavailable noncovalent thrombin inhibitors that utilize nonbasic groups in the P1 position. J Med Chem41:3210-9 (1998) [PubMed] Article Tucker, TJ; Brady, SF; Lumma, WC; Lewis, SD; Gardell, SJ; Naylor-Olsen, AM; Yan, Y; Sisko, JT; Stauffer, KJ; Lucas, BJ; Lynch, JJ; Cook, JJ; Stranieri, MT; Holahan, MA; Lyle, EA; Baskin, EP; Chen, IW; Dancheck, KB; Krueger, JA; Cooper, CM; Vacca, JP Design and synthesis of a series of potent and orally bioavailable noncovalent thrombin inhibitors that utilize nonbasic groups in the P1 position. J Med Chem41:3210-9 (1998) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Plasminogen |
|---|
| Name: | Plasminogen |
|---|
| Synonyms: | Activation peptide | Angiostatin | PLG | PLMN_HUMAN | Plasmin | Plasmin heavy chain A | Plasmin heavy chain A, short form | Plasmin light chain B |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 90579.18 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 810 |
|---|
| Sequence: | MEHKEVVLLLLLFLKSGQGEPLDDYVNTQGASLFSVTKKQLGAGSIEECAAKCEEDEEFT
CRAFQYHSKEQQCVIMAENRKSSIIIRMRDVVLFEKKVYLSECKTGNGKNYRGTMSKTKN
GITCQKWSSTSPHRPRFSPATHPSEGLEENYCRNPDNDPQGPWCYTTDPEKRYDYCDILE
CEEECMHCSGENYDGKISKTMSGLECQAWDSQSPHAHGYIPSKFPNKNLKKNYCRNPDRE
LRPWCFTTDPNKRWELCDIPRCTTPPPSSGPTYQCLKGTGENYRGNVAVTVSGHTCQHWS
AQTPHTHNRTPENFPCKNLDENYCRNPDGKRAPWCHTTNSQVRWEYCKIPSCDSSPVSTE
QLAPTAPPELTPVVQDCYHGDGQSYRGTSSTTTTGKKCQSWSSMTPHRHQKTPENYPNAG
LTMNYCRNPDADKGPWCFTTDPSVRWEYCNLKKCSGTEASVVAPPPVVLLPDVETPSEED
CMFGNGKGYRGKRATTVTGTPCQDWAAQEPHRHSIFTPETNPRAGLEKNYCRNPDGDVGG
PWCYTTNPRKLYDYCDVPQCAAPSFDCGKPQVEPKKCPGRVVGGCVAHPHSWPWQVSLRT
RFGMHFCGGTLISPEWVLTAAHCLEKSPRPSSYKVILGAHQEVNLEPHVQEIEVSRLFLE
PTRKDIALLKLSSPAVITDKVIPACLPSPNYVVADRTECFITGWGETQGTFGAGLLKEAQ
LPVIENKVCNRYEFLNGRVQSTELCAGHLAGGTDSCQGDSGGPLVCFEKDKYILQGVTSW
GLGCARPNKPGVYVRVSRFVTWIEGVMRNN
|
|
|
|---|
| BDBM50066336 |
|---|
| n/a |
|---|
| Name | BDBM50066336 |
|---|
| Synonyms: | (S)-1-((R)-2-Amino-2-cyclohexyl-acetyl)-pyrrolidine-2-carboxylic acid 2,5-dichloro-benzylamide | (S)-1-(2-Amino-2-cyclohexyl-acetyl)-pyrrolidine-2-carboxylic acid 2,5-dichloro-benzylamide | CHEMBL42495 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H27Cl2N3O2 |
|---|
| Mol. Mass. | 412.353 |
|---|
| SMILES | N[C@H](C1CCCCC1)C(=O)N1CCC[C@H]1C(=O)NCc1cc(Cl)ccc1Cl |
|---|
| Structure | 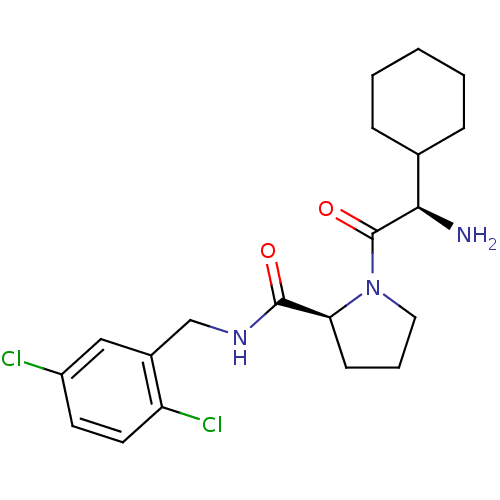
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tucker, TJ; Brady, SF; Lumma, WC; Lewis, SD; Gardell, SJ; Naylor-Olsen, AM; Yan, Y; Sisko, JT; Stauffer, KJ; Lucas, BJ; Lynch, JJ; Cook, JJ; Stranieri, MT; Holahan, MA; Lyle, EA; Baskin, EP; Chen, IW; Dancheck, KB; Krueger, JA; Cooper, CM; Vacca, JP Design and synthesis of a series of potent and orally bioavailable noncovalent thrombin inhibitors that utilize nonbasic groups in the P1 position. J Med Chem41:3210-9 (1998) [PubMed] Article
Tucker, TJ; Brady, SF; Lumma, WC; Lewis, SD; Gardell, SJ; Naylor-Olsen, AM; Yan, Y; Sisko, JT; Stauffer, KJ; Lucas, BJ; Lynch, JJ; Cook, JJ; Stranieri, MT; Holahan, MA; Lyle, EA; Baskin, EP; Chen, IW; Dancheck, KB; Krueger, JA; Cooper, CM; Vacca, JP Design and synthesis of a series of potent and orally bioavailable noncovalent thrombin inhibitors that utilize nonbasic groups in the P1 position. J Med Chem41:3210-9 (1998) [PubMed] Article