| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-2C adrenergic receptor |
|---|
| Ligand | BDBM50068818 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_33528 (CHEMBL876746) |
|---|
| Ki | 2244±n/a nM |
|---|
| Citation |  Nagarathnam, D; Wetzel, JM; Miao, SW; Marzabadi, MR; Chiu, G; Wong, WC; Hong, X; Fang, J; Forray, C; Branchek, TA; Heydorn, WE; Chang, RS; Broten, T; Schorn, TW; Gluchowski, C Design and synthesis of novel alpha1a adrenoceptor-selective dihydropyridine antagonists for the treatment of benign prostatic hyperplasia. J Med Chem41:5320-33 (1999) [PubMed] Article Nagarathnam, D; Wetzel, JM; Miao, SW; Marzabadi, MR; Chiu, G; Wong, WC; Hong, X; Fang, J; Forray, C; Branchek, TA; Heydorn, WE; Chang, RS; Broten, T; Schorn, TW; Gluchowski, C Design and synthesis of novel alpha1a adrenoceptor-selective dihydropyridine antagonists for the treatment of benign prostatic hyperplasia. J Med Chem41:5320-33 (1999) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-2C adrenergic receptor |
|---|
| Name: | Alpha-2C adrenergic receptor |
|---|
| Synonyms: | ADA2C_HUMAN | ADRA2C | ADRA2L2 | ADRA2RL2 | Adrenergic alpha2C | Adrenergic receptor | Adrenergic receptor alpha | Adrenergic, alpha-2C-, receptor | Alpha-2 adrenergic receptor subtype C4 | Alpha-2C adrenoceptor | Alpha-2C adrenoreceptor | adrenergic, alpha-2C-, receptor [Homo sapiens] |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 49552.32 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P18825 |
|---|
| Residue: | 462 |
|---|
| Sequence: | MASPALAAALAVAAAAGPNASGAGERGSGGVANASGASWGPPRGQYSAGAVAGLAAVVGF
LIVFTVVGNVLVVIAVLTSRALRAPQNLFLVSLASADILVATLVMPFSLANELMAYWYFG
QVWCGVYLALDVLFCTSSIVHLCAISLDRYWSVTQAVEYNLKRTPRRVKATIVAVWLISA
VISFPPLVSLYRQPDGAAYPQCGLNDETWYILSSCIGSFFAPCLIMGLVYARIYRVAKLR
TRTLSEKRAPVGPDGASPTTENGLGAAAGAGENGHCAPPPADVEPDESSAAAERRRRRGA
LRRGGRRRAGAEGGAGGADGQGAGPGAAESGALTASRSPGPGGRLSRASSRSVEFFLSRR
RRARSSVCRRKVAQAREKRFTFVLAVVMGVFVLCWFPFFFSYSLYGICREACQVPGPLFK
FFFWIGYCNSSLNPVIYTVFNQDFRRSFKHILFRRRRRGFRQ
|
|
|
|---|
| BDBM50068818 |
|---|
| n/a |
|---|
| Name | BDBM50068818 |
|---|
| Synonyms: | 1-(3-{[2,6-Dimethyl-5-methylcarbamoyl-4-(4-nitro-phenyl)-1,4-dihydro-pyridine-3-carbonyl]-amino}-propyl)-4-phenyl-piperidine-4-carboxylic acid methyl ester | CHEMBL147077 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H39N5O6 |
|---|
| Mol. Mass. | 589.682 |
|---|
| SMILES | CNC(=O)C1C(C(C(=O)NCCCN2CCC(CC2)(C(=O)OC)c2ccccc2)=C(C)N=C1C)c1ccc(cc1)[N+]([O-])=O |c:33,t:30| |
|---|
| Structure | 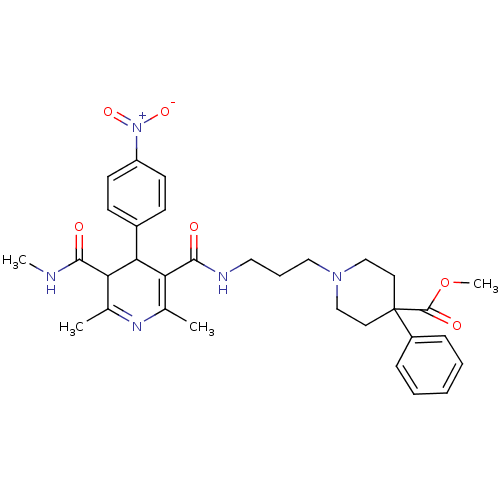
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nagarathnam, D; Wetzel, JM; Miao, SW; Marzabadi, MR; Chiu, G; Wong, WC; Hong, X; Fang, J; Forray, C; Branchek, TA; Heydorn, WE; Chang, RS; Broten, T; Schorn, TW; Gluchowski, C Design and synthesis of novel alpha1a adrenoceptor-selective dihydropyridine antagonists for the treatment of benign prostatic hyperplasia. J Med Chem41:5320-33 (1999) [PubMed] Article
Nagarathnam, D; Wetzel, JM; Miao, SW; Marzabadi, MR; Chiu, G; Wong, WC; Hong, X; Fang, J; Forray, C; Branchek, TA; Heydorn, WE; Chang, RS; Broten, T; Schorn, TW; Gluchowski, C Design and synthesis of novel alpha1a adrenoceptor-selective dihydropyridine antagonists for the treatment of benign prostatic hyperplasia. J Med Chem41:5320-33 (1999) [PubMed] Article