| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prothrombin |
|---|
| Ligand | BDBM50069152 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_209097 (CHEMBL811699) |
|---|
| Ki | 4500±n/a nM |
|---|
| Citation |  Duncia, JV; Santella, JB; Higley, CA; VanAtten, MK; Weber, PC; Alexander, RS; Kettner, CA; Pruitt, JR; Liauw, AY; Quan, ML; Knabb, RM; Wexler, RR Pyrazoles, 1,2,4-triazoles, and tetrazoles as surrogates for cis-amide bonds in boronate ester thrombin inhibitors. Bioorg Med Chem Lett8:775-80 (1999) [PubMed] Duncia, JV; Santella, JB; Higley, CA; VanAtten, MK; Weber, PC; Alexander, RS; Kettner, CA; Pruitt, JR; Liauw, AY; Quan, ML; Knabb, RM; Wexler, RR Pyrazoles, 1,2,4-triazoles, and tetrazoles as surrogates for cis-amide bonds in boronate ester thrombin inhibitors. Bioorg Med Chem Lett8:775-80 (1999) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prothrombin |
|---|
| Name: | Prothrombin |
|---|
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 70029.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00734 |
|---|
| Residue: | 622 |
|---|
| Sequence: | MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLEREC
VEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHV
NITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQE
CSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASA
QAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETG
DGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYI
DGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTEN
DLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHP
VCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDST
RIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKY
GFYTHVFRLKKWIQKVIDQFGE
|
|
|
|---|
| BDBM50069152 |
|---|
| n/a |
|---|
| Name | BDBM50069152 |
|---|
| Synonyms: | CHEMBL2373350 | N-[(R)-5-Amino-1-((1S,2S,6R,8S)-2,9,9-trimethyl-3,5-dioxa-4-bora-tricyclo[6.1.1.0*2,6*]dec-4-yl)-pentyl]-2-(5-benzyl-3-methylsulfanylmethyl-[1,2,4]triazol-1-yl)-acetamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H42BN5O3S |
|---|
| Mol. Mass. | 539.541 |
|---|
| SMILES | [H][C@@]12C[C@@]([H])(C1(C)C)[C@]1(C)OB(O[C@]1([H])C2)[C@H](CCCCN)NC(=O)Cn1nc(CSC)nc1Cc1ccccc1 |
|---|
| Structure | 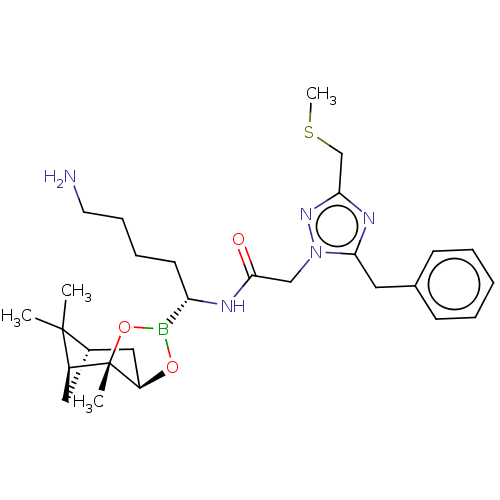
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Duncia, JV; Santella, JB; Higley, CA; VanAtten, MK; Weber, PC; Alexander, RS; Kettner, CA; Pruitt, JR; Liauw, AY; Quan, ML; Knabb, RM; Wexler, RR Pyrazoles, 1,2,4-triazoles, and tetrazoles as surrogates for cis-amide bonds in boronate ester thrombin inhibitors. Bioorg Med Chem Lett8:775-80 (1999) [PubMed]
Duncia, JV; Santella, JB; Higley, CA; VanAtten, MK; Weber, PC; Alexander, RS; Kettner, CA; Pruitt, JR; Liauw, AY; Quan, ML; Knabb, RM; Wexler, RR Pyrazoles, 1,2,4-triazoles, and tetrazoles as surrogates for cis-amide bonds in boronate ester thrombin inhibitors. Bioorg Med Chem Lett8:775-80 (1999) [PubMed]