| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Reverse transcriptase/RNaseH |
|---|
| Ligand | BDBM50070532 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_196516 (CHEMBL798330) |
|---|
| IC50 | 240±n/a nM |
|---|
| Citation |  Sahlberg, C; Noréen, R; Engelhardt, P; Högberg, M; Kangasmetsä, J; Vrang, L; Zhang, H Synthesis and anti-HIV activities of urea-PETT analogs belonging to a new class of potent non-nucleoside HIV-1 reverse transcriptase inhibitors. Bioorg Med Chem Lett8:1511-6 (1999) [PubMed] Sahlberg, C; Noréen, R; Engelhardt, P; Högberg, M; Kangasmetsä, J; Vrang, L; Zhang, H Synthesis and anti-HIV activities of urea-PETT analogs belonging to a new class of potent non-nucleoside HIV-1 reverse transcriptase inhibitors. Bioorg Med Chem Lett8:1511-6 (1999) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Reverse transcriptase/RNaseH |
|---|
| Name: | Reverse transcriptase/RNaseH |
|---|
| Synonyms: | HIV-1 Reverse Transcriptase RNase H | Human immunodeficiency virus type 1 reverse transcriptase | Reverse transcriptase/RNaseH |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 65229.15 |
|---|
| Organism: | Human immunodeficiency virus 1 |
|---|
| Description: | ChEMBL_1473730 |
|---|
| Residue: | 566 |
|---|
| Sequence: | PISPIETVPVKLKPGMDGPKVKQWPLTEEKIKALVEICTEMEKEGKISKIGPENPYNTPV
FAIKKKDSTKWRKLVDFRELNKRTQDFWEVQLGIPHPAGLKKRKSVTVLDVGDAYFSVPL
DEDFRKYTAFTIPSINNETPGIRYQYNVLPQGWKGSPAIFQSSMTKILEPFRKQNPDIVI
YQYMDDLYVGSDLEIGQHRTKIEELRQHLLRWGLTTPDKKHQKEPPFLWMGYELHPDKWT
VQPIVLPEKDSWTVNDIQKLVGKLNWASQIYPGIRVRQLCKLLRGTKALTEVIPLTEEAE
LELAENREILKEPVHGVYYDPSKDLIAEIQKQGQGQWTYQIYQEPFKNLRTGKYARMRGA
HTNDVKQLTEAVQKITTESIVIWGKTPKFKLPIQKETWETWWTEYWQATWIPEWEFVNTP
PLVKLWYQLEKEPIVGAETFYVDGAANRETKLGKAGYVTNRGRQKVVTLTDTTNQKTELQ
AIYLALQDSGLEVNIVTDSQYALGIIQAQPDQSESELVNQIIEQLIKKEKVYLAWVPAHK
GIGGNEQVDKLVSAGIRKVLFLDGID
|
|
|
|---|
| BDBM50070532 |
|---|
| n/a |
|---|
| Name | BDBM50070532 |
|---|
| Synonyms: | 1-(5-Bromo-pyridin-2-yl)-3-[(1R,2R)-2-(3-ethoxy-2-fluoro-6-methoxy-phenyl)-cyclopropyl]-urea | CHEMBL37834 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H19BrFN3O3 |
|---|
| Mol. Mass. | 424.264 |
|---|
| SMILES | CCOc1ccc(OC)c([C@H]2C[C@H]2NC(=O)Nc2ccc(Br)cn2)c1F |
|---|
| Structure | 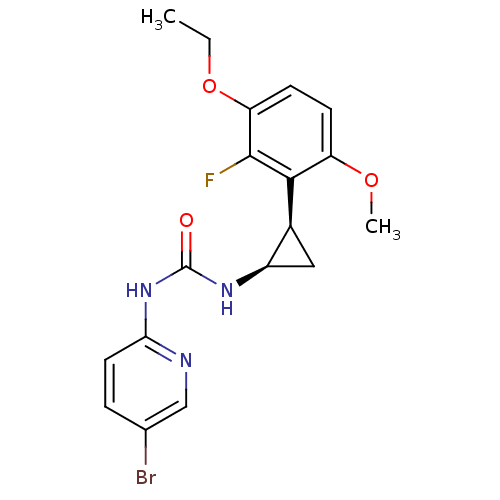
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sahlberg, C; Noréen, R; Engelhardt, P; Högberg, M; Kangasmetsä, J; Vrang, L; Zhang, H Synthesis and anti-HIV activities of urea-PETT analogs belonging to a new class of potent non-nucleoside HIV-1 reverse transcriptase inhibitors. Bioorg Med Chem Lett8:1511-6 (1999) [PubMed]
Sahlberg, C; Noréen, R; Engelhardt, P; Högberg, M; Kangasmetsä, J; Vrang, L; Zhang, H Synthesis and anti-HIV activities of urea-PETT analogs belonging to a new class of potent non-nucleoside HIV-1 reverse transcriptase inhibitors. Bioorg Med Chem Lett8:1511-6 (1999) [PubMed]