| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1B |
|---|
| Ligand | BDBM50005835 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1357 (CHEMBL616430) |
|---|
| Ki | 9.6±n/a nM |
|---|
| Citation |  Xu, YC; Schaus, JM; Walker, C; Krushinski, J; Adham, N; Zgombick, JM; Liang, SX; Kohlman, DT; Audia, JE N-Methyl-5-tert-butyltryptamine: A novel, highly potent 5-HT1D receptor agonist. J Med Chem42:526-31 (1999) [PubMed] Article Xu, YC; Schaus, JM; Walker, C; Krushinski, J; Adham, N; Zgombick, JM; Liang, SX; Kohlman, DT; Audia, JE N-Methyl-5-tert-butyltryptamine: A novel, highly potent 5-HT1D receptor agonist. J Med Chem42:526-31 (1999) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 1B |
|---|
| Name: | 5-hydroxytryptamine receptor 1B |
|---|
| Synonyms: | 5-HT-1B | 5-HT-1D-beta | 5-HT1B | 5-hydroxytryptamine receptor 1B (5-HT1B) | 5HT1B_HUMAN | HTR1B | HTR1DB | S12 | Serotonin (5-HT) receptor | Serotonin 1D beta receptor | Serotonin Receptor 1B |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 43579.17 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Receptor binding assays were performed using human clone stably expressed in CHO cells |
|---|
| Residue: | 390 |
|---|
| Sequence: | MEEPGAQCAPPPPAGSETWVPQANLSSAPSQNCSAKDYIYQDSISLPWKVLLVMLLALIT
LATTLSNAFVIATVYRTRKLHTPANYLIASLAVTDLLVSILVMPISTMYTVTGRWTLGQV
VCDFWLSSDITCCTASILHLCVIALDRYWAITDAVEYSAKRTPKRAAVMIALVWVFSISI
SLPPFFWRQAKAEEEVSECVVNTDHILYTVYSTVGAFYFPTLLLIALYGRIYVEARSRIL
KQTPNRTGKRLTRAQLITDSPGSTSSVTSINSRVPDVPSESGSPVYVNQVKVRVSDALLE
KKKLMAARERKATKTLGIILGAFIVCWLPFFIISLVMPICKDACWFHLAIFDFFTWLGYL
NSLINPIIYTMSNEDFKQAFHKLIRFKCTS
|
|
|
|---|
| BDBM50005835 |
|---|
| n/a |
|---|
| Name | BDBM50005835 |
|---|
| Synonyms: | (3-[2-(dimethylamino)ethyl]-1H-indol-5-yl)-N-methylmethanesulfonamide | 1-[3-(2-dimethylaminoethyl)-1H-indol-5-yl]-N-methyl-methanesulfonamide | 1-{3-[2-(dimethylamino)ethyl]-1H-indol-5-yl}-N-methylmethanesulfonamide | 3-(2-(dimethylamino)ethyl)-N-methyl-1H-indole-5-methanesulfonamide | 3-[2-(dimethylamino)ethyl]-N-methylindole-5-methanesulfonamide | CHEMBL128 | SUMATRIPTAN | Sumatran | Sumax |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H21N3O2S |
|---|
| Mol. Mass. | 295.4 |
|---|
| SMILES | CNS(=O)(=O)Cc1ccc2[nH]cc(CCN(C)C)c2c1 |
|---|
| Structure | 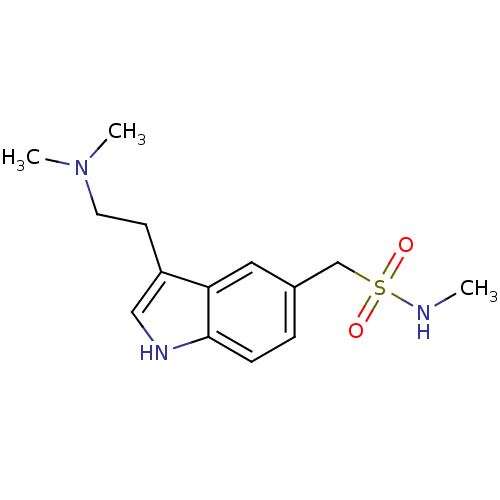
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Xu, YC; Schaus, JM; Walker, C; Krushinski, J; Adham, N; Zgombick, JM; Liang, SX; Kohlman, DT; Audia, JE N-Methyl-5-tert-butyltryptamine: A novel, highly potent 5-HT1D receptor agonist. J Med Chem42:526-31 (1999) [PubMed] Article
Xu, YC; Schaus, JM; Walker, C; Krushinski, J; Adham, N; Zgombick, JM; Liang, SX; Kohlman, DT; Audia, JE N-Methyl-5-tert-butyltryptamine: A novel, highly potent 5-HT1D receptor agonist. J Med Chem42:526-31 (1999) [PubMed] Article