| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Calpain-1 catalytic subunit |
|---|
| Ligand | BDBM50073850 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_43477 |
|---|
| Ki | 10±n/a nM |
|---|
| Citation |  Angelastro, MR; Marquart, AL; Mehdi, S; Koehl, JR; Vaz, RJ; Bey, P; Peet, NP The synthesis of ketomethylene pseudopeptide analogues of dipeptide aldehyde inhibitors of calpain. Bioorg Med Chem Lett9:139-40 (1999) [PubMed] Angelastro, MR; Marquart, AL; Mehdi, S; Koehl, JR; Vaz, RJ; Bey, P; Peet, NP The synthesis of ketomethylene pseudopeptide analogues of dipeptide aldehyde inhibitors of calpain. Bioorg Med Chem Lett9:139-40 (1999) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Calpain-1 catalytic subunit |
|---|
| Name: | Calpain-1 catalytic subunit |
|---|
| Synonyms: | CANP | CANX_CHICK | Calcium-activated neutral proteinase | Calpain 1/2 | Calpain-1 catalytic subunit | Calpain-1 large subunit | Mu/M-type |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 80319.95 |
|---|
| Organism: | Gallus gallus |
|---|
| Description: | ChEMBL_43477 |
|---|
| Residue: | 705 |
|---|
| Sequence: | MMPFGGIAARLQRDRLRAEGVGEHNNAVKYLNQDYEALKQECIESGTLFRDPQFPAGPTA
LGFKELGPYSSKTRGVEWKRPSELVDDPQFIVGGATRTDICQGALGDCWLLAAIGSLTLN
EELLHRVVPHGQSFQEDYAGIFHFQIWQFGEWVDVVVDDLLPTKDGELLFVHSAECTEFW
SALLEKAYAKLNGCYESLSGGSTTEGFEDFTGGVAEMYDLKRAPRNMGHIIRKALERGSL
LGCSIDITSAFDMEAVTFKKLVKGHAYSVTAFKDVNYRGQQEQLIRIRNPWGQVEWTGAW
SDGSSEWDNIDPSDREELQLKMEDGEFWMSFRDFMREFSRLEICNLTPDALTKDELSRWH
TQVFEGTWRRGSTAGGCRNNPATFWINPQFKIKLLEEDDDPGDDEVACSFLVALMQKHRR
RERRVGGDMHTIGFAVYEVPEEAQGSQNVHLKKDFFLRNQSRARSETFINLREVSNQIRL
PPGEYIVVPSTFEPHKEADFILRVFTEKQSDTAELDEEISADLADEEEITEDDIEDGFKN
MFQQLAGEDMEISVFELKTILNRVIARHKDLKTDGFSLDSCRNMVNLMDKDGSARLGLVE
FQILWNKIRSWLTIFRQYDLDKSGTMSSYEMRMALESAGFKLNNKLHQVVVARYADAETG
VDFDNFVCCLVKLETMFRFFHSMDRDGTGTAVMNLAEWLLLTMCG
|
|
|
|---|
| BDBM50073850 |
|---|
| n/a |
|---|
| Name | BDBM50073850 |
|---|
| Synonyms: | (S)-2-((S)-2-Benzyloxycarbonylamino-3-methyl-butyrylamino)-3-phenyl-propionic acid | CHEMBL423112 | MDL-28170 | [(S)-1-(1-Benzyl-2-oxo-ethylcarbamoyl)-2-methyl-propyl]-carbamic acid benzyl ester | [1-(1-Benzyl-2-oxo-ethylcarbamoyl)-2-methyl-propyl]-carbamic acid benzyl ester | [1-(1-Formyl-2-phenyl-ethylcarbamoyl)-2-methyl-propyl]-carbamic acid benzyl ester | benzyl (2S)-3-methyl-1-oxo-1-(1-oxo-3-phenylpropan-2-ylamino)butan-2-ylcarbamate | benzyl (S)-3-methyl-1-oxo-1-(1-oxo-3-phenylpropan-2-ylamino)butan-2-ylcarbamate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H26N2O4 |
|---|
| Mol. Mass. | 382.4528 |
|---|
| SMILES | CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)NC(Cc1ccccc1)C=O |r| |
|---|
| Structure | 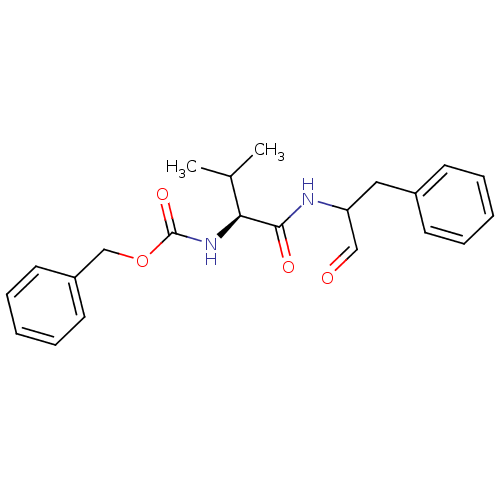
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Angelastro, MR; Marquart, AL; Mehdi, S; Koehl, JR; Vaz, RJ; Bey, P; Peet, NP The synthesis of ketomethylene pseudopeptide analogues of dipeptide aldehyde inhibitors of calpain. Bioorg Med Chem Lett9:139-40 (1999) [PubMed]
Angelastro, MR; Marquart, AL; Mehdi, S; Koehl, JR; Vaz, RJ; Bey, P; Peet, NP The synthesis of ketomethylene pseudopeptide analogues of dipeptide aldehyde inhibitors of calpain. Bioorg Med Chem Lett9:139-40 (1999) [PubMed]