

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Somatostatin receptor type 2 | ||
| Ligand | BDBM50075279 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEBML_200682 | ||
| Ki | 7.3±n/a nM | ||
| Citation |  Pasternak, A; Pan, Y; Marino, D; Sanderson, PE; Mosley, R; Rohrer, SP; Birzin, ET; Huskey, SE; Jacks, T; Schleim, KD; Cheng, K; Schaeffer, JM; Patchett, AA; Yang, L Potent, orally bioavailable somatostatin agonists: good absorption achieved by urea backbone cyclization. Bioorg Med Chem Lett9:491-6 (1999) [PubMed] Pasternak, A; Pan, Y; Marino, D; Sanderson, PE; Mosley, R; Rohrer, SP; Birzin, ET; Huskey, SE; Jacks, T; Schleim, KD; Cheng, K; Schaeffer, JM; Patchett, AA; Yang, L Potent, orally bioavailable somatostatin agonists: good absorption achieved by urea backbone cyclization. Bioorg Med Chem Lett9:491-6 (1999) [PubMed] | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Somatostatin receptor type 2 | |||
| Name: | Somatostatin receptor type 2 | ||
| Synonyms: | SOMATOSTATIN SST2 | SRIF-1 | SS-2-R | SS2-R | SS2R | SSR2_HUMAN | SSTR2 | Somatostatin receptor type 2 (SSTR2) | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 41344.94 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P30874 | ||
| Residue: | 369 | ||
| Sequence: |
| ||
| BDBM50075279 | |||
| n/a | |||
| Name | BDBM50075279 | ||
| Synonyms: | (2R,3S)-N-(5-Amino-[1,3]dioxan-2-ylmethyl)-2-[3-(3,4-dichloro-benzyl)-2-oxo-imidazolidin-1-yl]-3-(1H-indol-3-yl)-butyramide | CHEMBL147092 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C27H31Cl2N5O4 | ||
| Mol. Mass. | 560.472 | ||
| SMILES | C[C@H]([C@@H](N1CCN(Cc2ccc(Cl)c(Cl)c2)C1=O)C(=O)NC[C@H]1OC[C@H](N)CO1)c1c[nH]c2ccccc12 |wU:2.2,22.23,wD:25.27,1.0,(-.86,3.13,;-.83,1.59,;.5,.83,;1.83,1.6,;1.82,3.14,;4.49,3.17,;4.49,1.63,;5.84,.86,;7.16,1.65,;8.5,.88,;9.83,1.66,;9.81,3.2,;11.14,3.98,;8.47,3.96,;8.45,5.5,;7.15,3.17,;3.16,.84,;3.18,-.7,;.5,-.71,;2.05,-.71,;-.27,-2.04,;.47,-3.37,;-.3,-4.7,;.47,-6.04,;-.32,-7.37,;-1.86,-7.36,;-2.65,-8.68,;-2.61,-6,;-1.84,-4.69,;-2.16,.81,;-2.16,-.72,;-3.49,-1.5,;-4.82,-.73,;-6.15,-1.5,;-7.48,-.73,;-7.48,.81,;-6.15,1.58,;-4.82,.81,)| | ||
| Structure | 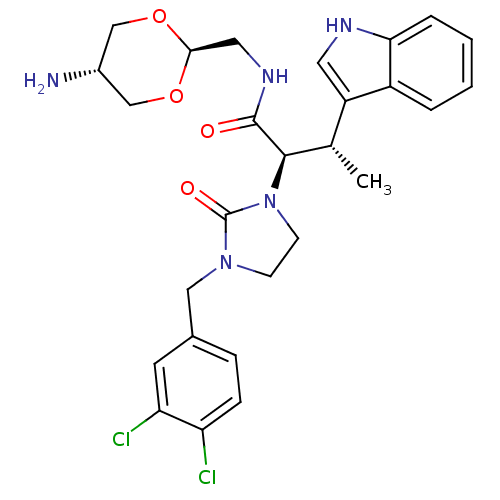
| ||