| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1A |
|---|
| Ligand | BDBM50077588 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1415 (CHEMBL616203) |
|---|
| Ki | 0.400000±n/a nM |
|---|
| Citation |  Mewshaw, RE; Webb, MB; Marquis, KL; McGaughey, GB; Shi, X; Wasik, T; Scerni, R; Brennan, JA; Andree, TH New generation dopaminergic agents. 6. Structure-activity relationship studies of a series of 4-(aminoethoxy)indole and 4-(aminoethoxy)indolone derivatives based on the newly discovered 3-hydroxyphenoxyethylamine D2 template. J Med Chem42:2007-20 (1999) [PubMed] Article Mewshaw, RE; Webb, MB; Marquis, KL; McGaughey, GB; Shi, X; Wasik, T; Scerni, R; Brennan, JA; Andree, TH New generation dopaminergic agents. 6. Structure-activity relationship studies of a series of 4-(aminoethoxy)indole and 4-(aminoethoxy)indolone derivatives based on the newly discovered 3-hydroxyphenoxyethylamine D2 template. J Med Chem42:2007-20 (1999) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 1A |
|---|
| Name: | 5-hydroxytryptamine receptor 1A |
|---|
| Synonyms: | 5-HT-1A | 5-HT1 | 5-HT1A | 5-Hydroxytryptamine receptor 1A (5-HT1A) | 5-hydroxytryptamine receptor 1A (5HT1A) | 5HT1A_RAT | 5ht1a | G-21 | Htr1a | Serotonin 1 (5-HT1) receptor | Serotonin 1a (5-HT1a) receptor/Adrenergic receptor alpha-1 | Serotonin receptor 1A |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 46445.29 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Binding assays were performed using rat hippocampal membranes. |
|---|
| Residue: | 422 |
|---|
| Sequence: | MDVFSFGQGNNTTASQEPFGTGGNVTSISDVTFSYQVITSLLLGTLIFCAVLGNACVVAA
IALERSLQNVANYLIGSLAVTDLMVSVLVLPMAALYQVLNKWTLGQVTCDLFIALDVLCC
TSSILHLCAIALDRYWAITDPIDYVNKRTPRRAAALISLTWLIGFLISIPPMLGWRTPED
RSDPDACTISKDHGYTIYSTFGAFYIPLLLMLVLYGRIFRAARFRIRKTVRKVEKKGAGT
SLGTSSAPPPKKSLNGQPGSGDWRRCAENRAVGTPCTNGAVRQGDDEATLEVIEVHRVGN
SKEHLPLPSESGSNSYAPACLERKNERNAEAKRKMALARERKTVKTLGIIMGTFILCWLP
FFIVALVLPFCESSCHMPALLGAIINWLGYSNSLLNPVIYAYFNKDFQNAFKKIIKCKFC
RR
|
|
|
|---|
| BDBM50077588 |
|---|
| n/a |
|---|
| Name | BDBM50077588 |
|---|
| Synonyms: | CHEMBL59737 | [2-(1H-Indol-4-yloxy)-ethyl]-(4-phenyl-butyl)-amine |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H24N2O |
|---|
| Mol. Mass. | 308.4174 |
|---|
| SMILES | C(CCc1ccccc1)CNCCOc1cccc2[nH]ccc12 |
|---|
| Structure | 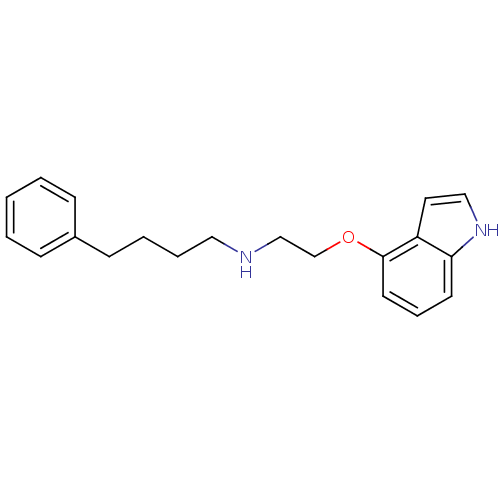
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Mewshaw, RE; Webb, MB; Marquis, KL; McGaughey, GB; Shi, X; Wasik, T; Scerni, R; Brennan, JA; Andree, TH New generation dopaminergic agents. 6. Structure-activity relationship studies of a series of 4-(aminoethoxy)indole and 4-(aminoethoxy)indolone derivatives based on the newly discovered 3-hydroxyphenoxyethylamine D2 template. J Med Chem42:2007-20 (1999) [PubMed] Article
Mewshaw, RE; Webb, MB; Marquis, KL; McGaughey, GB; Shi, X; Wasik, T; Scerni, R; Brennan, JA; Andree, TH New generation dopaminergic agents. 6. Structure-activity relationship studies of a series of 4-(aminoethoxy)indole and 4-(aminoethoxy)indolone derivatives based on the newly discovered 3-hydroxyphenoxyethylamine D2 template. J Med Chem42:2007-20 (1999) [PubMed] Article