| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-1B adrenergic receptor |
|---|
| Ligand | BDBM50082843 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_34459 (CHEMBL651991) |
|---|
| Ki | 16±n/a nM |
|---|
| Citation |  Lagu, B; Tian, D; Nagarathnam, D; Marzabadi, MR; Wong, WC; Miao, SW; Zhang, F; Sun, W; Chiu, G; Fang, J; Forray, C; Chang, RS; Ransom, RW; Chen, TB; O'Malley, S; Zhang, K; Vyas, KP; Gluchowski, C Design and synthesis of novel alpha(1)(a) adrenoceptor-selective antagonists. 3. Approaches to eliminate opioid agonist metabolites by using substituted phenylpiperazine side chains. J Med Chem42:4794-803 (1999) [PubMed] Lagu, B; Tian, D; Nagarathnam, D; Marzabadi, MR; Wong, WC; Miao, SW; Zhang, F; Sun, W; Chiu, G; Fang, J; Forray, C; Chang, RS; Ransom, RW; Chen, TB; O'Malley, S; Zhang, K; Vyas, KP; Gluchowski, C Design and synthesis of novel alpha(1)(a) adrenoceptor-selective antagonists. 3. Approaches to eliminate opioid agonist metabolites by using substituted phenylpiperazine side chains. J Med Chem42:4794-803 (1999) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-1B adrenergic receptor |
|---|
| Name: | Alpha-1B adrenergic receptor |
|---|
| Synonyms: | ADA1B_HUMAN | ADRA1B | Adrenergic alpha1B | Adrenergic receptor | Adrenergic receptor alpha | Alpha 1B-adrenoceptor | Alpha 1B-adrenoreceptor | Alpha-1B adrenergic receptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 56862.13 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P35368 |
|---|
| Residue: | 520 |
|---|
| Sequence: | MNPDLDTGHNTSAPAHWGELKNANFTGPNQTSSNSTLPQLDITRAISVGLVLGAFILFAI
VGNILVILSVACNRHLRTPTNYFIVNLAMADLLLSFTVLPFSAALEVLGYWVLGRIFCDI
WAAVDVLCCTASILSLCAISIDRYIGVRYSLQYPTLVTRRKAILALLSVWVLSTVISIGP
LLGWKEPAPNDDKECGVTEEPFYALFSSLGSFYIPLAVILVMYCRVYIVAKRTTKNLEAG
VMKEMSNSKELTLRIHSKNFHEDTLSSTKAKGHNPRSSIAVKLFKFSREKKAAKTLGIVV
GMFILCWLPFFIALPLGSLFSTLKPPDAVFKVVFWLGYFNSCLNPIIYPCSSKEFKRAFV
RILGCQCRGRGRRRRRRRRRLGGCAYTYRPWTRGGSLERSQSRKDSLDDSGSCLSGSQRT
LPSASPSPGYLGRGAPPPVELCAFPEWKAPGALLSLPAPEPPGRRGRHDSGPLFTFKLLT
EPESPGTDGGASNGGCEAAADVANGQPGFKSNMPLAPGQF
|
|
|
|---|
| BDBM50082843 |
|---|
| n/a |
|---|
| Name | BDBM50082843 |
|---|
| Synonyms: | 4-(3,4-Difluoro-phenyl)-3-{3-[4-(2-hydroxy-phenyl)-piperazin-1-yl]-propylcarbamoyl}-6-methyl-2-oxo-1,2,3,4-tetrahydro-pyrimidine-5-carboxylic acid methyl ester | CHEMBL141821 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H31F2N5O5 |
|---|
| Mol. Mass. | 543.5623 |
|---|
| SMILES | COC(=O)C1=C(C)NC(=O)N(C1c1ccc(F)c(F)c1)C(=O)NCCCN1CCN(CC1)c1ccccc1O |c:4| |
|---|
| Structure | 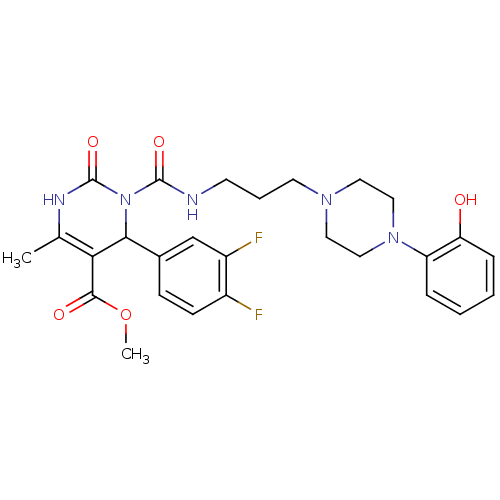
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lagu, B; Tian, D; Nagarathnam, D; Marzabadi, MR; Wong, WC; Miao, SW; Zhang, F; Sun, W; Chiu, G; Fang, J; Forray, C; Chang, RS; Ransom, RW; Chen, TB; O'Malley, S; Zhang, K; Vyas, KP; Gluchowski, C Design and synthesis of novel alpha(1)(a) adrenoceptor-selective antagonists. 3. Approaches to eliminate opioid agonist metabolites by using substituted phenylpiperazine side chains. J Med Chem42:4794-803 (1999) [PubMed]
Lagu, B; Tian, D; Nagarathnam, D; Marzabadi, MR; Wong, WC; Miao, SW; Zhang, F; Sun, W; Chiu, G; Fang, J; Forray, C; Chang, RS; Ransom, RW; Chen, TB; O'Malley, S; Zhang, K; Vyas, KP; Gluchowski, C Design and synthesis of novel alpha(1)(a) adrenoceptor-selective antagonists. 3. Approaches to eliminate opioid agonist metabolites by using substituted phenylpiperazine side chains. J Med Chem42:4794-803 (1999) [PubMed]