| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-1B adrenergic receptor |
|---|
| Ligand | BDBM50060964 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_30103 (CHEMBL642030) |
|---|
| Ki | 0.2±n/a nM |
|---|
| Citation |  Meyer, MD; Altenbach, RJ; Basha, FZ; Carroll, WA; Condon, S; Elmore, SW; Kerwin, JF; Sippy, KB; Tietje, K; Wendt, MD; Hancock, AA; Brune, ME; Buckner, SA; Drizin, I Structure-activity studies for a novel series of tricyclic substituted hexahydrobenz[e]isoindole alpha(1A) adrenoceptor antagonists as potential agents for the symptomatic treatment of benign prostatic hyperplasia (BPH). J Med Chem43:1586-603 (2000) [PubMed] Meyer, MD; Altenbach, RJ; Basha, FZ; Carroll, WA; Condon, S; Elmore, SW; Kerwin, JF; Sippy, KB; Tietje, K; Wendt, MD; Hancock, AA; Brune, ME; Buckner, SA; Drizin, I Structure-activity studies for a novel series of tricyclic substituted hexahydrobenz[e]isoindole alpha(1A) adrenoceptor antagonists as potential agents for the symptomatic treatment of benign prostatic hyperplasia (BPH). J Med Chem43:1586-603 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-1B adrenergic receptor |
|---|
| Name: | Alpha-1B adrenergic receptor |
|---|
| Synonyms: | ADA1B_MESAU | ADRA1B | Adrenergic receptor alpha | Alpha-1b adrenergic receptor | adrenergic Alpha1B |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 56514.13 |
|---|
| Organism: | C.H.O. |
|---|
| Description: | adrenergic Alpha1B ADRA1B C.H.O.::P18841 |
|---|
| Residue: | 515 |
|---|
| Sequence: | MNPDLDTGHNTSAPAQWGELKDANFTGPNQTSSNSTLPQLDVTRAISVGLVLGAFILFAI
VGNILVILSVACNRHLRTPTNYFIVNLAIADLLLSFTVLPFSATLEVLGYWVLGRIFCDI
WAAVDVLCCTASILSLCAISIDRYIGVRYSLQYPTLVTRRKAILALLSVWVLSTVISIGP
LLGWKEPAPNDDKECGVTEEPFYALFSSLGSFYIPLAVILVMYCRVYIVAKRTTKNLEAG
VMKEMSNSKELTLRIHSKNFHEDTLSSTKAKGHNPRSSIAVKLFKFSREKKAAKTLGIVV
GMFILCWLPFFIALPLGSLFSTLKPPDAVFKVVFWLGYFNSCLNPIIYPCSSKEFKRAFM
RILGCQCRSGRRRRRRRRLGACAYTYRPWTRGGSLERSQSRKDSLDDSGSCMSGSQRTLP
SASPSPGYLGRGAQPPLELCAYPEWKSGALLSLPEPPGRRGRLDSGPLFTFKLLGEPESP
GTEGDASNGGCDATTDLANGQPGFKSNMPLAPGHF
|
|
|
|---|
| BDBM50060964 |
|---|
| n/a |
|---|
| Name | BDBM50060964 |
|---|
| Synonyms: | (R)-5-(2-((2-(2-ethoxyphenoxy)ethyl)amino)propyl)-2-methoxybenzenesulfonamide | 5-[(2R)-2-{[2-(2-ethoxyphenoxy)ethyl]amino}propyl]-2-methoxybenzenesulfonamide | 5-[2-[2-(2-ethoxyphenoxy)ethylamino]propyl]-2-methoxy-benzenesulfonamide | CHEMBL836 | TAMSULOSIN |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H28N2O5S |
|---|
| Mol. Mass. | 408.512 |
|---|
| SMILES | CCOc1ccccc1OCCN[C@H](C)Cc1ccc(OC)c(c1)S(N)(=O)=O |
|---|
| Structure | 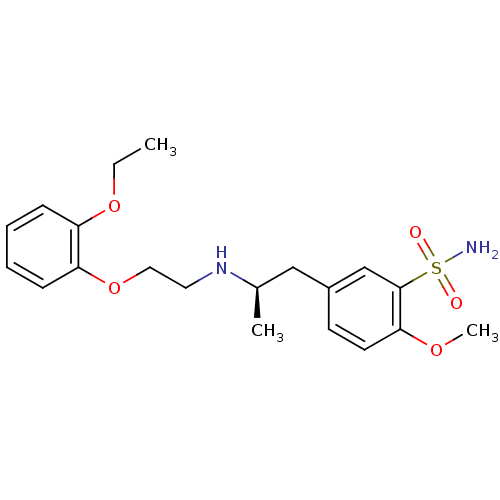
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Meyer, MD; Altenbach, RJ; Basha, FZ; Carroll, WA; Condon, S; Elmore, SW; Kerwin, JF; Sippy, KB; Tietje, K; Wendt, MD; Hancock, AA; Brune, ME; Buckner, SA; Drizin, I Structure-activity studies for a novel series of tricyclic substituted hexahydrobenz[e]isoindole alpha(1A) adrenoceptor antagonists as potential agents for the symptomatic treatment of benign prostatic hyperplasia (BPH). J Med Chem43:1586-603 (2000) [PubMed]
Meyer, MD; Altenbach, RJ; Basha, FZ; Carroll, WA; Condon, S; Elmore, SW; Kerwin, JF; Sippy, KB; Tietje, K; Wendt, MD; Hancock, AA; Brune, ME; Buckner, SA; Drizin, I Structure-activity studies for a novel series of tricyclic substituted hexahydrobenz[e]isoindole alpha(1A) adrenoceptor antagonists as potential agents for the symptomatic treatment of benign prostatic hyperplasia (BPH). J Med Chem43:1586-603 (2000) [PubMed]