| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Bifunctional purine biosynthesis protein ATIC |
|---|
| Ligand | BDBM50089572 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_32812 |
|---|
| Ki | 17000±n/a nM |
|---|
| Citation |  Boger, DL; Marsilje, TH; Castro, RA; Hedrick, MP; Jin, Q; Baker, SJ; Shim, JH; Benkovic, SJ Design, synthesis, and biological evaluation of fluoronitrophenyl substituted folate analogues as potential inhibitors of GAR transformylase and AICAR transformylase. Bioorg Med Chem Lett10:1471-5 (2000) [PubMed] Boger, DL; Marsilje, TH; Castro, RA; Hedrick, MP; Jin, Q; Baker, SJ; Shim, JH; Benkovic, SJ Design, synthesis, and biological evaluation of fluoronitrophenyl substituted folate analogues as potential inhibitors of GAR transformylase and AICAR transformylase. Bioorg Med Chem Lett10:1471-5 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Bifunctional purine biosynthesis protein ATIC |
|---|
| Name: | Bifunctional purine biosynthesis protein ATIC |
|---|
| Synonyms: | 5-aminoimidazole-4-carboxamide ribonucleotide formyltransferase | 5-aminoimidazole-4-carboxamide-ribonucleotide transformylase | AICAR Tfase | AICAR transformylase | ATIC | Aminoimidazole carboxamide ribonucleotide transformylase (AICAR Tfase) | Bifunctional purine biosynthesis protein PURH | IMP Cyclohydrolase (IMPCH) | IMP cyclohydrolase | IMP synthetase | Inosinicase | PUR9_HUMAN | PURH | Phosphoribosylaminoimidazolecarboxamide formyltransferase | Thymidylate synthase/GAR transformylase/AICAR transformylase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 64616.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P31939 |
|---|
| Residue: | 592 |
|---|
| Sequence: | MAPGQLALFSVSDKTGLVEFARNLTALGLNLVASGGTAKALRDAGLAVRDVSELTGFPEM
LGGRVKTLHPAVHAGILARNIPEDNADMARLDFNLIRVVACNLYPFVKTVASPGVTVEEA
VEQIDIGGVTLLRAAAKNHARVTVVCEPEDYVVVSTEMQSSESKDTSLETRRQLALKAFT
HTAQYDEAISDYFRKQYSKGVSQMPLRYGMNPHQTPAQLYTLQPKLPITVLNGAPGFINL
CDALNAWQLVKELKEALGIPAAASFKHVSPAGAAVGIPLSEDEAKVCMVYDLYKTLTPIS
AAYARARGADRMSSFGDFVALSDVCDVPTAKIISREVSDGIIAPGYEEEALTILSKKKNG
NYCVLQMDQSYKPDENEVRTLFGLHLSQKRNNGVVDKSLFSNVVTKNKDLPESALRDLIV
ATIAVKYTQSNSVCYAKNGQVIGIGAGQQSRIHCTRLAGDKANYWWLRHHPQVLSMKFKT
GVKRAEISNAIDQYVTGTIGEDEDLIKWKALFEEVPELLTEAEKKEWVEKLTEVSISSDA
FFPFRDNVDRAKRSGVAYIAAPSGSAADKVVIEACDELGIILAHTNLRLFHH
|
|
|
|---|
| BDBM50089572 |
|---|
| n/a |
|---|
| Name | BDBM50089572 |
|---|
| Synonyms: | 2-{4-[(2-Amino-4-oxo-3,4-dihydro-quinazolin-6-ylmethyl)-(4-fluoro-3-nitro-benzyl)-amino]-benzoylamino}-pentanedioic acid | CHEMBL286459 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H25FN6O8 |
|---|
| Mol. Mass. | 592.5319 |
|---|
| SMILES | Nc1nc2ccc(CN(Cc3ccc(F)c(c3)[N+]([O-])=O)c3ccc(cc3)C(=O)NC(CCC(O)=O)C(O)=O)cc2c(=O)[nH]1 |
|---|
| Structure | 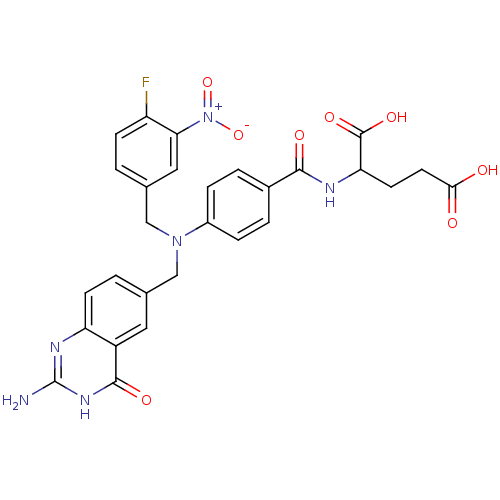
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Boger, DL; Marsilje, TH; Castro, RA; Hedrick, MP; Jin, Q; Baker, SJ; Shim, JH; Benkovic, SJ Design, synthesis, and biological evaluation of fluoronitrophenyl substituted folate analogues as potential inhibitors of GAR transformylase and AICAR transformylase. Bioorg Med Chem Lett10:1471-5 (2000) [PubMed]
Boger, DL; Marsilje, TH; Castro, RA; Hedrick, MP; Jin, Q; Baker, SJ; Shim, JH; Benkovic, SJ Design, synthesis, and biological evaluation of fluoronitrophenyl substituted folate analogues as potential inhibitors of GAR transformylase and AICAR transformylase. Bioorg Med Chem Lett10:1471-5 (2000) [PubMed]