| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-1B adrenergic receptor |
|---|
| Ligand | BDBM50090018 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_34456 (CHEMBL651988) |
|---|
| Ki | 290±n/a nM |
|---|
| Citation |  Barrow, JC; Nantermet, PG; Selnick, HG; Glass, KL; Rittle, KE; Gilbert, KF; Steele, TG; Homnick, CF; Freidinger, RM; Ransom, RW; Kling, P; Reiss, D; Broten, TP; Schorn, TW; Chang, RS; O'Malley, SS; Olah, TV; Ellis, JD; Barrish, A; Kassahun, K; Leppert, P; Nagarathnam, D; Forray, C In vitro and in vivo evaluation of dihydropyrimidinone C-5 amides as potent and selective alpha(1A) receptor antagonists for the treatment of benign prostatic hyperplasia. J Med Chem43:2703-18 (2000) [PubMed] Barrow, JC; Nantermet, PG; Selnick, HG; Glass, KL; Rittle, KE; Gilbert, KF; Steele, TG; Homnick, CF; Freidinger, RM; Ransom, RW; Kling, P; Reiss, D; Broten, TP; Schorn, TW; Chang, RS; O'Malley, SS; Olah, TV; Ellis, JD; Barrish, A; Kassahun, K; Leppert, P; Nagarathnam, D; Forray, C In vitro and in vivo evaluation of dihydropyrimidinone C-5 amides as potent and selective alpha(1A) receptor antagonists for the treatment of benign prostatic hyperplasia. J Med Chem43:2703-18 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-1B adrenergic receptor |
|---|
| Name: | Alpha-1B adrenergic receptor |
|---|
| Synonyms: | ADA1B_HUMAN | ADRA1B | Adrenergic alpha1B | Adrenergic receptor | Adrenergic receptor alpha | Alpha 1B-adrenoceptor | Alpha 1B-adrenoreceptor | Alpha-1B adrenergic receptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 56862.13 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P35368 |
|---|
| Residue: | 520 |
|---|
| Sequence: | MNPDLDTGHNTSAPAHWGELKNANFTGPNQTSSNSTLPQLDITRAISVGLVLGAFILFAI
VGNILVILSVACNRHLRTPTNYFIVNLAMADLLLSFTVLPFSAALEVLGYWVLGRIFCDI
WAAVDVLCCTASILSLCAISIDRYIGVRYSLQYPTLVTRRKAILALLSVWVLSTVISIGP
LLGWKEPAPNDDKECGVTEEPFYALFSSLGSFYIPLAVILVMYCRVYIVAKRTTKNLEAG
VMKEMSNSKELTLRIHSKNFHEDTLSSTKAKGHNPRSSIAVKLFKFSREKKAAKTLGIVV
GMFILCWLPFFIALPLGSLFSTLKPPDAVFKVVFWLGYFNSCLNPIIYPCSSKEFKRAFV
RILGCQCRGRGRRRRRRRRRLGGCAYTYRPWTRGGSLERSQSRKDSLDDSGSCLSGSQRT
LPSASPSPGYLGRGAPPPVELCAFPEWKAPGALLSLPAPEPPGRRGRHDSGPLFTFKLLT
EPESPGTDGGASNGGCEAAADVANGQPGFKSNMPLAPGQF
|
|
|
|---|
| BDBM50090018 |
|---|
| n/a |
|---|
| Name | BDBM50090018 |
|---|
| Synonyms: | 4-(3,4-Difluoro-phenyl)-2-oxo-1,2,3,4-tetrahydro-pyrimidine-5-carboxylic acid {3-[4-(2-cyano-phenyl)-piperidin-1-yl]-propyl}-amide | CHEMBL90652 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H27F2N5O2 |
|---|
| Mol. Mass. | 479.5217 |
|---|
| SMILES | Fc1ccc(cc1F)[C@H]1NC(=O)N=CC1C(=O)NCCCN1CCC(CC1)c1ccccc1C#N |c:13| |
|---|
| Structure | 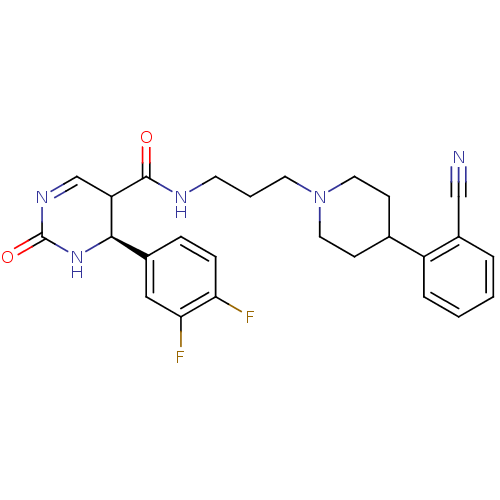
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Barrow, JC; Nantermet, PG; Selnick, HG; Glass, KL; Rittle, KE; Gilbert, KF; Steele, TG; Homnick, CF; Freidinger, RM; Ransom, RW; Kling, P; Reiss, D; Broten, TP; Schorn, TW; Chang, RS; O'Malley, SS; Olah, TV; Ellis, JD; Barrish, A; Kassahun, K; Leppert, P; Nagarathnam, D; Forray, C In vitro and in vivo evaluation of dihydropyrimidinone C-5 amides as potent and selective alpha(1A) receptor antagonists for the treatment of benign prostatic hyperplasia. J Med Chem43:2703-18 (2000) [PubMed]
Barrow, JC; Nantermet, PG; Selnick, HG; Glass, KL; Rittle, KE; Gilbert, KF; Steele, TG; Homnick, CF; Freidinger, RM; Ransom, RW; Kling, P; Reiss, D; Broten, TP; Schorn, TW; Chang, RS; O'Malley, SS; Olah, TV; Ellis, JD; Barrish, A; Kassahun, K; Leppert, P; Nagarathnam, D; Forray, C In vitro and in vivo evaluation of dihydropyrimidinone C-5 amides as potent and selective alpha(1A) receptor antagonists for the treatment of benign prostatic hyperplasia. J Med Chem43:2703-18 (2000) [PubMed]