| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM50095028 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_2274 |
|---|
| Ki | 2.7±n/a nM |
|---|
| Citation |  Smith, AL; Stevenson, GI; Lewis, S; Patel, S; Castro, JL Solid-phase synthesis of 2,3-disubstituted indoles: discovery of a novel, high-affinity, selective h5-HT2A antagonist. Bioorg Med Chem Lett10:2693-6 (2000) [PubMed] Smith, AL; Stevenson, GI; Lewis, S; Patel, S; Castro, JL Solid-phase synthesis of 2,3-disubstituted indoles: discovery of a novel, high-affinity, selective h5-HT2A antagonist. Bioorg Med Chem Lett10:2693-6 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2 | 5-HT-2A | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT-2A) | 5-hydroxytryptamine receptor 2A (5HT-2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_HUMAN | HTR2 | HTR2A | Serotonin receptor 2A |
|---|
| Type: | undefined |
|---|
| Mol. Mass.: | 52607.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P28223 |
|---|
| Residue: | 471 |
|---|
| Sequence: | MDILCEENTSLSSTTNSLMQLNDDTRLYSNDFNSGEANTSDAFNWTVDSENRTNLSCEGC
LSPSCLSLLHLQEKNWSALLTAVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAVWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VSFFIPLTIMVITYFLTIKSLQKEATLCVSDLGTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYTGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNEDVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENKKPLQLILVNTIPALAYK
SSQLQMGQKKNSKQDAKTTDNDCSMVALGKQHSEEASKDNSDGVNEKVSCV
|
|
|
|---|
| BDBM50095028 |
|---|
| n/a |
|---|
| Name | BDBM50095028 |
|---|
| Synonyms: | 2-Phenyl-3-(2-piperidin-1-yl-ethyl)-1H-indole | CHEMBL91205 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H24N2 |
|---|
| Mol. Mass. | 304.4287 |
|---|
| SMILES | C(Cc1c([nH]c2ccccc12)-c1ccccc1)N1CCCCC1 |
|---|
| Structure | 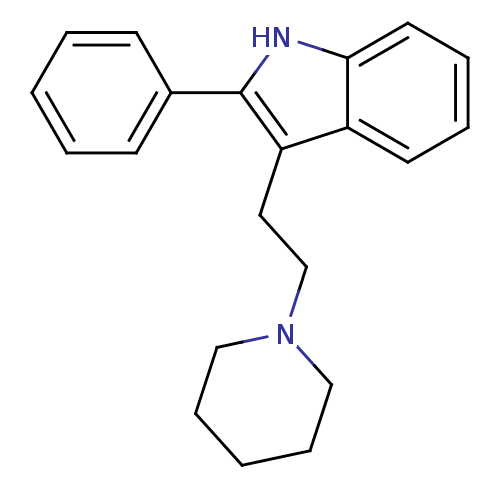
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Smith, AL; Stevenson, GI; Lewis, S; Patel, S; Castro, JL Solid-phase synthesis of 2,3-disubstituted indoles: discovery of a novel, high-affinity, selective h5-HT2A antagonist. Bioorg Med Chem Lett10:2693-6 (2000) [PubMed]
Smith, AL; Stevenson, GI; Lewis, S; Patel, S; Castro, JL Solid-phase synthesis of 2,3-disubstituted indoles: discovery of a novel, high-affinity, selective h5-HT2A antagonist. Bioorg Med Chem Lett10:2693-6 (2000) [PubMed]