| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Peroxisome proliferator-activated receptor alpha |
|---|
| Ligand | BDBM50103945 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_153237 (CHEMBL764649) |
|---|
| EC50 | 400±n/a nM |
|---|
| Citation |  Liu, KG; Smith, JS; Ayscue, AH; Henke, BR; Lambert, MH; Leesnitzer, LM; Plunket, KD; Willson, TM; Sternbach, DD Identification of a series of oxadiazole-substituted alpha-isopropoxy phenylpropanoic acids with activity on PPARalpha, PPARgamma, and PPARdelta. Bioorg Med Chem Lett11:2385-8 (2001) [PubMed] Liu, KG; Smith, JS; Ayscue, AH; Henke, BR; Lambert, MH; Leesnitzer, LM; Plunket, KD; Willson, TM; Sternbach, DD Identification of a series of oxadiazole-substituted alpha-isopropoxy phenylpropanoic acids with activity on PPARalpha, PPARgamma, and PPARdelta. Bioorg Med Chem Lett11:2385-8 (2001) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Peroxisome proliferator-activated receptor alpha |
|---|
| Name: | Peroxisome proliferator-activated receptor alpha |
|---|
| Synonyms: | NR1C1 | Nuclear receptor subfamily 1 group C member 1 | PPAR | PPAR alpha/gamma | PPAR-alpha | PPARA | PPARA_HUMAN | Peroxisome Proliferator-Activated Receptor alpha | Peroxisome proliferator-activated receptor | Peroxisome proliferator-activated receptor alpha (PPAR alpha) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 52222.08 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q07869 |
|---|
| Residue: | 468 |
|---|
| Sequence: | MVDTESPLCPLSPLEAGDLESPLSEEFLQEMGNIQEISQSIGEDSSGSFGFTEYQYLGSC
PGSDGSVITDTLSPASSPSSVTYPVVPGSVDESPSGALNIECRICGDKASGYHYGVHACE
GCKGFFRRTIRLKLVYDKCDRSCKIQKKNRNKCQYCRFHKCLSVGMSHNAIRFGRMPRSE
KAKLKAEILTCEHDIEDSETADLKSLAKRIYEAYLKNFNMNKVKARVILSGKASNNPPFV
IHDMETLCMAEKTLVAKLVANGIQNKEAEVRIFHCCQCTSVETVTELTEFAKAIPGFANL
DLNDQVTLLKYGVYEAIFAMLSSVMNKDGMLVAYGNGFITREFLKSLRKPFCDIMEPKFD
FAMKFNALELDDSDISLFVAAIICCGDRPGLLNVGHIEKMQEGIVHVLRLHLQSNHPDDI
FLFPKLLQKMADLRQLVTEHAQLVQIIKKTESDAALHPLLQEIYRDMY
|
|
|
|---|
| BDBM50103945 |
|---|
| n/a |
|---|
| Name | BDBM50103945 |
|---|
| Synonyms: | 2-Isopropoxy-3-(4-{2-[5-(2-trifluoromethyl-phenyl)-[1,2,4]oxadiazol-3-yl]-ethoxy}-phenyl)-propionic acid | 2-isopropoxy-3-(4-(2-(5-(2-(trifluoromethyl)phenyl)-1,2,4-oxadiazol-3-yl)ethoxy)phenyl)propanoic acid | CHEMBL433316 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H23F3N2O5 |
|---|
| Mol. Mass. | 464.4343 |
|---|
| SMILES | CC(C)OC(Cc1ccc(OCCc2noc(n2)-c2ccccc2C(F)(F)F)cc1)C(O)=O |
|---|
| Structure | 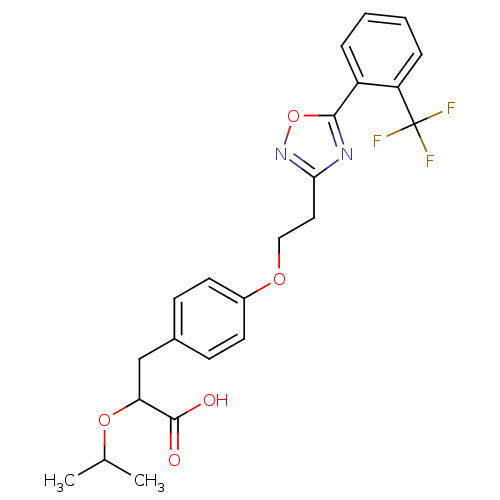
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Liu, KG; Smith, JS; Ayscue, AH; Henke, BR; Lambert, MH; Leesnitzer, LM; Plunket, KD; Willson, TM; Sternbach, DD Identification of a series of oxadiazole-substituted alpha-isopropoxy phenylpropanoic acids with activity on PPARalpha, PPARgamma, and PPARdelta. Bioorg Med Chem Lett11:2385-8 (2001) [PubMed]
Liu, KG; Smith, JS; Ayscue, AH; Henke, BR; Lambert, MH; Leesnitzer, LM; Plunket, KD; Willson, TM; Sternbach, DD Identification of a series of oxadiazole-substituted alpha-isopropoxy phenylpropanoic acids with activity on PPARalpha, PPARgamma, and PPARdelta. Bioorg Med Chem Lett11:2385-8 (2001) [PubMed]