| Reaction Details |
|---|
 | Report a problem with these data |
| Target | C-C chemokine receptor type 5 |
|---|
| Ligand | BDBM50104940 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_39638 (CHEMBL649944) |
|---|
| Ki | 1.1±n/a nM |
|---|
| Citation |  Palani, A; Shapiro, S; Clader, JW; Greenlee, WJ; Cox, K; Strizki, J; Endres, M; Baroudy, BM Discovery of 4-[(Z)-(4-bromophenyl)- (ethoxyimino)methyl]-1'-[(2,4-dimethyl-3- pyridinyl)carbonyl]-4'-methyl-1,4'- bipiperidine N-oxide (SCH 351125): an orally bioavailable human CCR5 antagonist for the treatment of HIV infection. J Med Chem44:3339-42 (2001) [PubMed] Palani, A; Shapiro, S; Clader, JW; Greenlee, WJ; Cox, K; Strizki, J; Endres, M; Baroudy, BM Discovery of 4-[(Z)-(4-bromophenyl)- (ethoxyimino)methyl]-1'-[(2,4-dimethyl-3- pyridinyl)carbonyl]-4'-methyl-1,4'- bipiperidine N-oxide (SCH 351125): an orally bioavailable human CCR5 antagonist for the treatment of HIV infection. J Med Chem44:3339-42 (2001) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| C-C chemokine receptor type 5 |
|---|
| Name: | C-C chemokine receptor type 5 |
|---|
| Synonyms: | C-C CKR-5 | C-C chemokine receptor type 5 | CC-CKR-5 | CCR-5 | CCR5 | CCR5/mu opioid receptor complex | CCR5_HUMAN | CD_antigen=CD195 | CHEMR13 | CMKBR5 | HIV-1 fusion coreceptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 40540.21 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P51681 |
|---|
| Residue: | 352 |
|---|
| Sequence: | MDYQVSSPIYDINYYTSEPCQKINVKQIAARLLPPLYSLVFIFGFVGNMLVILILINCKR
LKSMTDIYLLNLAISDLFFLLTVPFWAHYAAAQWDFGNTMCQLLTGLYFIGFFSGIFFII
LLTIDRYLAVVHAVFALKARTVTFGVVTSVITWVVAVFASLPGIIFTRSQKEGLHYTCSS
HFPYSQYQFWKNFQTLKIVILGLVLPLLVMVICYSGILKTLLRCRNEKKRHRAVRLIFTI
MIVYFLFWAPYNIVLLLNTFQEFFGLNNCSSSNRLDQAMQVTETLGMTHCCINPIIYAFV
GEKFRNYLLVFFQKHIAKRFCKCCSIFQQEAPERASSVYTRSTGEQEISVGL
|
|
|
|---|
| BDBM50104940 |
|---|
| n/a |
|---|
| Name | BDBM50104940 |
|---|
| Synonyms: | CHEMBL323172 | {4-[(4-Bromo-phenyl)-methoxyimino-methyl]-4'-methyl-[1,4']bipiperidinyl-1'-yl}-(2,4-dimethyl-pyridin-3-yl)-methanone |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H35BrN4O2 |
|---|
| Mol. Mass. | 527.496 |
|---|
| SMILES | CO\N=C(\C1CCN(CC1)C1(C)CCN(CC1)C(=O)c1c(C)ccnc1C)c1ccc(Br)cc1 |
|---|
| Structure | 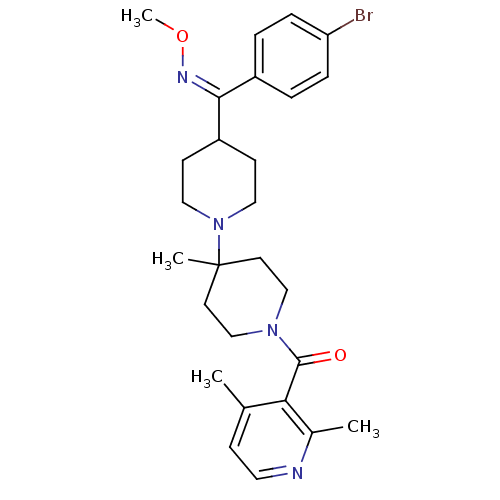
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Palani, A; Shapiro, S; Clader, JW; Greenlee, WJ; Cox, K; Strizki, J; Endres, M; Baroudy, BM Discovery of 4-[(Z)-(4-bromophenyl)- (ethoxyimino)methyl]-1'-[(2,4-dimethyl-3- pyridinyl)carbonyl]-4'-methyl-1,4'- bipiperidine N-oxide (SCH 351125): an orally bioavailable human CCR5 antagonist for the treatment of HIV infection. J Med Chem44:3339-42 (2001) [PubMed]
Palani, A; Shapiro, S; Clader, JW; Greenlee, WJ; Cox, K; Strizki, J; Endres, M; Baroudy, BM Discovery of 4-[(Z)-(4-bromophenyl)- (ethoxyimino)methyl]-1'-[(2,4-dimethyl-3- pyridinyl)carbonyl]-4'-methyl-1,4'- bipiperidine N-oxide (SCH 351125): an orally bioavailable human CCR5 antagonist for the treatment of HIV infection. J Med Chem44:3339-42 (2001) [PubMed]